Abstract
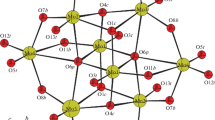
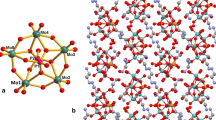
Organic–inorganic hybrid compounds of transition metal mono-substituted molybdophosphates, (H2bipy)2[PNi(Hbipy)Mo11O39]·4H2O (1), (H2bipy)1.5[HPCu(Hbipy)Mo11O39]·4H2O (2) (bipy = 4,4′-bipyridine), were synthesized hydrothermally and characterized by elemental analysis, IR spectroscopy, TGA, fluorescent spectroscopy, powder and single crystal X-ray diffraction methods. In 1 and 2 the transition metal atom (Ni and Cu) occupies one site of twelve molybdenum atoms of Keggin anion and combines a monoprotonated bipyridine molecular cation, Hbipy+, forming a 4,4′-bipyridine–decorated M-substituted molybdophosphate anion (M = Ni and Cu). In 1 two anions form a dimer through hydrogen bonds, in 2 the anions form a chain through hydrogen bonds. The dimers or the chains, H2bipy2+ cations and lattice water molecules are connected by hydrogen bonds, giving supramolecular compounds. The two kinds of bipy molecules, coordinated and uncoordinated, in 1 and 2 give two emission peaks in their photoluminescent spectra.






Similar content being viewed by others
References
P. Souchay, Polyanions and Polycations (Gauthiers-Villars, Paris, 1963), and references therein.
C. L. Hill (2003). Compr. Coord. Chem. II 4, 679–759.
C. M. Tourne and G. F. Tourne (1970). J. Inorg. Nucl. Chem. 32, 3975–3979.
L. C. W. Baker, V. S. Baker, K. Eriks, M. T. Pope, M. Shibata, O. W. Rollins, J. H. Fang, and L. L. Koh (1966). J. Am. Chem. Soc. 88, 2329–2332.
L. C. Baker and J. S. Figgis (1970). J. Am. Chem. Soc. 92, 3794–3797.
H. T. E. Jun, T. J. R. Weakley, and G. B. Jameson (1996). J. Chem. Soc. Dalton Trans. 12, 2537–2540.
E. V. Radkov and R. H. Beer (2000). Inorg. Chim. Acta 297, 191–198.
J. J. Cowan, A. J. Bailey, R. A. Heintz, B. T. Do, K. I. Hardcastle, C. L. Hill, and I. A. Weinstock (2001). Inorg. Chem. 40, 6666–6672.
H. Weiner, H.-J. Lunk, R. Friese, and H. Hart (2005). Inorg. Chem. 44, 7751–7761.
G. G. Gao, F. Y. Li, L. Xu, X. Z. Liu, and Y. Y. Yang (2008). J. Am. Chem. Soc. 130, 10838–10839.
F. M. Santos, P. Brandao, V. Felix, M. R. M. Domingues, J. S. Amaral, V. S. Amaral, H. I. S. Nogueira, and A. M. V. Cavaleiro (2012). Dalton Trans. 41, 12145–12155.
S. Reinoso, P. Vitoria, L. San Felices, and J. M. Gutiérrez-Zorrilla (2013). Eur. J. Inorg. Chem. 2013, 1644–1648.
L. A. Combs-Walker and C. L. Hill (1991). Inorg. Chem. 30, 4016–4026.
L. Lisnard, A. Dolbecq, P. Mialane, J. Marrot, and F. Sécheresse (2004). Inorg. Chim. Acta 357, 845–852.
J.-L. Liang, H.-J. Zhang, Y.-K. Lu, H.-L. Guo, J.-C. Zhao, M.-M. Wu, Y.-Q. Liu, and C.-G. Liu (2014). Inorg. Chem. Commun. 45, 135–139.
S. Ogo, M. Miyamoto, Y. Ide, T. Sano, and M. Sadakane (2012). Dalton Trans. 41, 9901–9907.
X. Wei, M. H. Dickman, and M. T. Pope (1998). J. Am. Chem. Soc. 120, 10254–10255.
J. R. Galán-Mascarós, C. Giménez-Saiz, S. Triki, and C. J. Gómez-Garcí (1995). Angew. Chem. Int. Ed. Engl. 34, 1460–1462.
Y. Lu, Y. Xu, E. Wang, Y. Li, L. Wang, C. Hu, and L. Xu (2004). J. Solid State Chem. 177, 2210–2215.
Y. Lu, Y. Xu, E. Wang, J. Lü, C. Hu, and L. Xu (2004). Cryst. Growth Des. 5, 257–260.
L. S. Felices, P. Vitoria, J. M. Gutiérrez-Zorrilla, L. Lezama, and S. Reinoso (2006). Inorg. Chem. 45, 7748–7757.
Q.-Z. Zhang and C.-Z. Lu (2006). Z. Anorg. Allg. Chem. 632, 330–334.
D. K. Lyon, W. K. Miller, T. Novet, P. J. Domaille, E. Evitt, D. C. Johnson, and R. G. Finke (1991). J. Am. Chem. Soc. 113, 7209–7221.
A. M. Khenkin and C. L. Hill (1993). J. Am. Chem. Soc. 115, 8178–8186.
J. Wang, L. Yan, G. Li, X. Wang, Y. Ding, and J. Suo (2005). Tetrahedron Lett. 46, 7023–7027.
N. V. Maksimchuk, M. N. Timofeeva, M. S. Melgunov, A. N. Shmakov, Y. A. Chesalov, D. N. Dybtsev, V. P. Fedin, and O. A. Kholdeeva (2008). J. Catal. 257, 315–323.
D. Kumar, E. Derat, A. M. Khenkin, R. Neumann, and S. Shaik (2005). J. Am. Chem. Soc. 117, 17712–17718.
A. M. Khenkin, D. Kumar, S. Shaik, and R. Neumann (2006). J. Am. Chem. Soc. 128, 15451–15460.
O. A. Kholdeeva, M. N. Timofeeva, G. M. Maksimov, R. I. Maksimolskaya, W. A. Neiwert, and C. L. Hill (2005). Inorg. Chem. 44, 666–672.
X. Z. Liu, G. G. Gao, L. Xu, F. Y. Li, L. Liu, N. Jiang, and Y. Y. Yang (2009). Solid State Sci. 11, 1433–1438.
L. Chen, D. Shi, J. Zhao, Y. Wang, P. Ma, J. Wang, and J. Niu (2011). Cryst. Growth Des. 11, 1913–1923.
S. Reinoso, P. Vitoria, J. M. Gutiérrez-Zorrilla, L. Lezama, L. San Felices, and J. I. Beitia (2005). Inorg. Chem. 44, 9731–9742.
H. I. S. Nogueira, F. A. A. Paz, P. A. F. Teixeira, and J. Klinowski (2006). Chem. Commun. 28, 2953–2955.
S. Reinoso, P. Vitoria, L. S. Felices, L. Lezama, and J. M. Gutiérrez-Zorrilla (2006). Inorg. Chem. 45, 108–118.
Y. B. Huang, J. X. Chen, T. Y. Lan, X. Q. Lu, C. X. Wei, Z. S. Li, and Z. C. Zhang (2006). J. Mol. Struct. 783, 168–175.
C. Pichon, A. Dolbecq, P. Mialane, J. Marrot, E. Rivière, M. Goral, M. Zynek, T. McCormac, S. A. Borshch, E. Zueva, and F. Sécheresse (2008). Chem. Eur. J. 14, 3189–3196.
H. B. Yan, Y. Xu, X. H. Bu, N. K. Goh, L. S. Chia, and G. D. Stucky (2001). J. Chem. Soc. Dalton Trans. 2009–2014.
J. Y. Niu, Z. L. Wang, and J. P. Wang (2004). J. Solid State Chem. 177, 3411–3417.
J.-P. Wang, X.-D. Du, and J.-Y. Niu (2007). J. Solid State Chem. 180, 1347–1352.
G. M. Sheldrick SHELXS97, Program for Crystal Structure Solution (University of Göttingen, Göttingen, 1997).
R. Thouvenot, M. Fournier, and R. Franck (1984). Inorg. Chem. 23, 598–605.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, XM., Shi, T. & Chen, YG. Syntheses, Crystal Structures and Properties of Organic–Inorganic Hybrid Compounds of 4,4′-Bipyridine–Decorated Ni/Cu-Substituted Molybdophosphates. J Clust Sci 27, 457–468 (2016). https://doi.org/10.1007/s10876-015-0940-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-015-0940-4