Abstract
Purpose
The aim of this study is to evaluate the usefulness of T cell receptor excision circle (TREC) and/or kappa-deleting recombination excision circle (KREC) measurements integrated with diagnostic next-generation sequencing (NGS) analysis using a severe combined immunodeficiency (SCID) newborn screening (NBS) program.
Methods
TREC and/or KREC values were measured in 137,484 newborns between April 2017 and December 2021 using EnLite TREC (n = 80,791) or TREC/KREC kits (n = 56,693). For newborns with positive screening results, diagnostic NGS analysis was performed with a 349-gene panel to detect genetic mutations associated with primary immunodeficiencies (PIDs).
Results
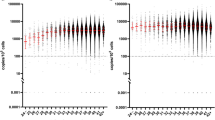
A total of 145 newborns (0.11%) had abnormal TREC and/or KREC values, and a genetic diagnosis was established in 2 patients with SCID (1 in 68,742 newborns) (IL2RG-SCID and reticular dysgenesis) and 10 with non-SCID PIDs with T and/or B cell deficiencies (1 in 13,748 newborns) using NGS analysis. Furthermore, TREC values of 2849 newborns were measured and confirmed the significant correlation between the results of both TREC and TREC/KREC kits (P < 0.001) and naïve T cell counts.
Conclusions
We performed the first large-scale TREC and TREC/KREC NBS programs in Japan. Our NBS programs followed by the diagnostic NGS analysis for newborns with abnormal TREC and/or KREC values are useful for the early identification and rapid molecular evaluation of not only SCID but also different non-SCID PIDs.




Similar content being viewed by others
Data Availability
The data used in this study will be provided to qualified researchers on reasonable request.
References
Heimall J. Severe combined immunodeficiency (SCID): an overview. UpToDate, Post TW (Ed), UpToDate, Waltham, MA.(Accessed on December 12, 2019.)
Amatuni GS, Currier RJ, Church JA, et al. Newborn screening for severe combined immunodeficiency and t-cell lymphopenia in California, 2010–2017. Pediatrics. 2019;143(2):e20182300. https://doi.org/10.1542/peds.2018-2300
Kwan A, Abraham RS, Currier R, Brower A, Andruszewski K, Abbott JK, et al. Newborn screening for severe combined immunodeficiency in 11 screening programs in the United States. JAMA. 2014;312:729–38.
Chan SB, Zhong Y, Lim SCJ, Poh S, Teh KL, Soh JY, et al. Implementation of universal newborn screening for severe combined immunodeficiency in Singapore while continuing routine Bacille-Calmette-Guerin Vaccination given at birth. Front Immunol. 2021;12:794221.
Barbaro M, Ohlsson A, Borte S, Jonsson S, Zetterstrom RH, King J, et al. Newborn screening for severe primary immunodeficiency diseases in Sweden-a 2-year pilot TREC and KREC screening study. J Clin Immunol. 2017;37:51–60.
Currier R, Puck JM. SCID newborn screening: what we’ve learned. J Allergy Clin Immunol. 2021;147:417–26.
Strand J, Gul KA, Erichsen HC, Lundman E, Berge MC, Tromborg AK, et al. Second-tier next generation sequencing integrated in nationwide newborn screening provides rapid molecular diagnostics of severe combined immunodeficiency. Front Immunol. 2020;11:1417.
Tangye SG, Al-Herz W, Bousfiha A, Chatila T, Cunningham-Rundles C, Etzioni A, et al. Human inborn errors of immunity: 2019 update on the classification from the International Union of Immunological Societies Expert Committee. J Clin Immunol. 2020;40:24–64.
Markert ML, Gupton SE, McCarthy EA. Experience with cultured thymus tissue in 105 children. J Allergy Clin Immunol. 2022;149:747–57.
Blom M, Zetterstrom RH, Stray-Pedersen A, Gilmour K, Gennery AR, Puck JM, et al. Recommendations for uniform definitions used in newborn screening for severe combined immunodeficiency. J Allergy Clin Immunol. 2022;149:1428–36.
Kojima D, Wang X, Muramatsu H, Okuno Y, Nishio N, Hama A, et al. Application of extensively targeted next-generation sequencing for the diagnosis of primary immunodeficiencies. J Allergy Clin Immunol. 2016;138:303-5.e3.
Li H, Durbin R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics. 2009;25:1754–60.
Koboldt DC, Zhang Q, Larson DE, Shen D, McLellan MD, Lin L, et al. VarScan 2: somatic mutation and copy number alteration discovery in cancer by exome sequencing. Genome Res. 2012;22:568–76.
Muramatsu H, Okuno Y, Yoshida K, Shiraishi Y, Doisaki S, Narita A, et al. Clinical utility of next-generation sequencing for inherited bone marrow failure syndromes. Genet Med. 2017;19:796–802.
Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015;17:405–24.
Kanda Y. Investigation of the freely available easy-to-use software “EZR” for medical statistics. Bone Marrow Transplant. 2013;48:452–8.
Berg JS, Agrawal PB, Bailey DB, Jr., et al. Newborn sequencing in genomic medicine and public health. Pediatrics. 2017;139(2):e20162252. https://doi.org/10.1542/peds.2016-2252
Adhikari AN, Gallagher RC, Wang Y, Currier RJ, Amatuni G, Bassaganyas L, et al. The role of exome sequencing in newborn screening for inborn errors of metabolism. Nat Med. 2020;26:1392–7.
Blom M, Bredius RGM, Jansen ME, Weijman G, Kemper EA, Vermont CL, et al. Parents’ perspectives and societal acceptance of implementation of newborn screening for SCID in the Netherlands. J Clin Immunol. 2021;41:99–108.
de Felipe B, Olbrich P, Lucenas JM, Delgado-Pecellin C, Pavon-Delgado A, Marquez J, et al. Prospective neonatal screening for severe T- and B-lymphocyte deficiencies in Seville. Pediatr Allergy Immunol. 2016;27:70–7.
Lougaris V, Soresina A, Baronio M, Montin D, Martino S, Signa S, et al. Long-term follow-up of 168 patients with X-linked agammaglobulinemia reveals increased morbidity and mortality. J Allergy Clin Immunol. 2020;146:429–37.
Matamoros Flori N, Mila Llambi J, Espanol Boren T, Raga Borja S, Fontan CG. Primary immunodeficiency syndrome in Spain: first report of the National Registry in Children and Adults. J Clin Immunol. 1997;17:333–9.
Ryser O, Morell A, Hitzig WH. Primary immunodeficiencies in Switzerland: first report of the national registry in adults and children. J Clin Immunol. 1988;8:479–85.
Nakagawa N, Imai K, Kanegane H, Sato H, Yamada M, Kondoh K, et al. Quantification of kappa-deleting recombination excision circles in Guthrie cards for the identification of early B-cell maturation defects. J Allergy Clin Immunol. 2011;128:223-5.e2.
Borte S, von Dobeln U, Fasth A, Wang N, Janzi M, Winiarski J, et al. Neonatal screening for severe primary immunodeficiency diseases using high-throughput triplex real-time PCR. Blood. 2012;119:2552–5.
Speckmann C, Neumann C, Borte S, la Marca G, Sass JO, Wiech E, et al. Delayed-onset adenosine deaminase deficiency: strategies for an early diagnosis. J Allergy Clin Immunol. 2012;130:991–4.
Sharapova SO, Pashchenko OE, Bondarenko AV, Vakhlyarskaya SS, Prokofjeva T, Fedorova AS, et al. Geographical distribution, incidence, malignancies, and outcome of 136 Eastern Slavic patients with Nijmegen Breakage syndrome and NBN founder variant c.657_661del5. Front Immunol. 2020;11:602482.
Flinn AM, Gennery AR. Adenosine deaminase deficiency: a review. Orphanet J Rare Dis. 2018;13:65.
Winkelstein JA, Marino MC, Lederman HM, Jones SM, Sullivan K, Burks AW, et al. X-linked agammaglobulinemia: report on a United States registry of 201 patients. Med (Baltimore). 2006;85:193–202.
Acknowledgements
The authors acknowledge all clinicians, patients, and their families and thank Ms. Yoshie Miura, Ms. Hiroko Ono, and Ms. Chie Amahori for their valuable assistance. The authors acknowledge the Division for Medical Research Engineering, Nagoya University Graduate School of Medicine, for providing technical support.
Funding
This work was supported by a grant from Aichi Rare Disease Network, Nagoya Pediatric Cancer Fund, and Japan Agency for Medical Research and Development (grant/award number: 20gk0110041h0002 to H.M.).
Author information
Authors and Affiliations
Contributions
MW, HM, and Tetsuya Ito performed laboratory works, gathered the clinical information, designed and conducted the research, analyzed data, and wrote the paper. DK and YO performed laboratory work, gathered the clinical information, and analyzed data. S. Kojima and YT directed the research and analyzed the data. S. Kataoka, FN, YS, YN, and IT performed laboratory work and gathered the clinical information. Tsuyoshi Ito, KU, AS, EM, YI, NO, MT, and TT gathered the clinical information. All authors reviewed and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Ethics Approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of the Nagoya University Graduate School of Medicine (Date, 11/10/2016; Approval number, 2016–00258231).
Consent to Participate
Written informed consent was obtained from the parents.
Consent for Publication
The authors affirm that no consent to publish is required for this study.
Conflict of Interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wakamatsu, M., Kojima, D., Muramatsu, H. et al. TREC/KREC Newborn Screening followed by Next-Generation Sequencing for Severe Combined Immunodeficiency in Japan. J Clin Immunol 42, 1696–1707 (2022). https://doi.org/10.1007/s10875-022-01335-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-022-01335-0