Abstract
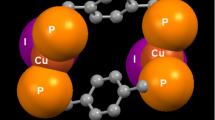
Two new bis(β-diketone) ligands based on triphenylamine have been prepared and crystallized. Treatment of 4,4′-diformyltriphenylamine with two phospholenes (2,2,2-trimethoxy-4,5-dimethyl-1,3,2-dioxaphospholene and 2,2,2-trimethoxy-4,5-diethyl-1,3,2-dioxaphospholene) afforded the bis(β-diketones) triphenylaminebis(2,4-pentanedione) (tpbaH2, 1); and triphenylaminebis(3,5-heptanedione) (tpbprH2, 2) as white solids. X-ray analysis of 1 and 2 shows that they are in the enol form. Also, their triphenylamine moieties have a chiral “propeller” shape; the centrosymmetric structures contain equal numbers of the two enantiomeric propellers. Reaction of 1 and 2 with [Cu(NH3)4]2+(aq) yielded dark-green solids. The Cu complex of 1 was insoluble in common solvents, but that of 2, Cu4(tpbpr)4 (3), is soluble in dichloromethane and chloroform. 3 is assigned the cyclic tetrameric structure Cu4(tpbpr)4 based on ESI–MS and microanalytical data. This is similar to the copper(II) β-diketonate molecular squares reported previously from our group. Molecular modeling indicates that Cu4(tpbpr)4 has Cu···Cu distances of ca. 21 Å, as compared to ca. 14 Å in the previous molecular squares.
Graphical Abstract
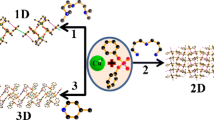
Crystal structures of two new bis(β-diketones) are reported, which are likely to form large supramolecular structures on reaction with metal ions such as Cu2+.










Similar content being viewed by others
Data availability
The X-ray datasets generated and analyzed during the current study are available via the Cambridge Structural Database (https://www.ccdc.cam.ac.uk/structures/), using the CCDC deposition numbers in Table 1.
References
Hwang S, Moorefield CN, Fronczek FR, Lukoyanova O, Echegoyen L, Newkome GR (2005) Construction of triangular metallomacrocycles: [M3(1,2-bis(2,2′∶6′,2″-terpyridin-4-yl-ethynyl)benzene)3] [M = Ru(II), Fe(II), 2Ru(II)Fe(II)]. Chem Commun 713–715. https://doi.org/10.1039/B409348H
Newkome GR, Cho TJ, Moorefield CN, Baker GR, Cush R, Russo PS (1999) Self- and directed assembly of hexaruthenium macrocycles. Angew Chem Int Ed 38:3717–3721. https://doi.org/10.1002/(SICI)1521-3773(19991216)38:24%3C3717::AID-ANIE3717%3E3.0.CO;2-C
Stang PJ, Olenyuk B (1997) Self-Assembly, symmetry, and molecular architecture: coordination as the motif in the rational design of supramolecular metallacyclic polygons and polyhedra. Acc Chem Res 30:502–518. https://doi.org/10.1021/ar9602011
Uehara K, Kasai K, Mizuno N (2007) Syntheses and characterizations of palladium-based molecular triangle/square compounds and hybrid composites with polyoxometalates. Inorg Chem 46:2563–2570. https://doi.org/10.1021/ic062156r
Berben LA, Faia MC, Crawford NRM, Long JR (2006) Angle-dependent electronic effects in 4,4′-bipyridine-bridged Ru3 triangle and Ru4 square complexes. Inorg Chem 45:6378–6386. https://doi.org/10.1021/ic060570l
Brock AJ, Clegg JK, Li F, Lindoy LF (2018) Recent developments in the metallo-supramolecular chemistry of oligo-β-diketonato ligands. Coord Chem Rev 375:106–133. https://doi.org/10.1016/j.ccr.2017.11.007
Clegg JK, Li F, Lindoy LF (2022) Oligo-β-diketones as versatile ligands for use in metallo-supramolecular chemistry: recent progress and perspectives. Coord Chem Rev 455:214355. https://doi.org/10.1016/j.ccr.2021.214355
Pariya C, Sparrow CC, Back CK, Sandí G, Fronczek FR, Maverick AW (2007) Copper β-diketonate molecular squares and their host-guest reactions. Angew Chem Int Ed 46:6305–6308. https://doi.org/10.1002/anie.200701252
Ramirez F, Bhatia SB, Patwardhan AV, Smith CP (1967) Molecular rearrangements during solvolyses of pentaoxyphosphoranes. Polyketones derived from phthalaldehyde and terephthalaldehyde. J Org Chem 32:3547–3553. https://doi.org/10.1021/jo01286a058
Cherutoi JK, Sandifer JD, Pokharel UR, Fronczek FR, Pakhomova S, Maverick AW (2015) Externally and internally functionalized copper(II) β-diketonate molecular squares. Inorg Chem 54:7791–7802. https://doi.org/10.1021/acs.inorgchem.5b00792
Mallegol T, Gmouh S, Meziane MAA, Blanchard-Desce M, Mongin O (2005) Practical and efficient synthesis of tris(4-formylphenyl)amine, a key building block in materials chemistry. Synthesis (Stuttgart) 1771–1774. https://doi.org/10.1055/s-2005-865336
Ramirez F, Patwardhan AV, Ramanathan N, Desai NB, Greco CV, Heller SR (1965) A new synthesis of α,β-dihydroxy ketones via oxyphosphoranes. Condensation of aliphatic α-diketones with aldehydes by means of trialkyl phosphites. P31 and H1 nuclear magnetic resonance spectra. J Am Chem Soc 87:543–548. https://doi.org/10.1021/ja01081a027
Sheldrick GM (2002) SADABS (computer program). University of Göttingen, Germany
Otwinowski Z, Minor W (1997) Processing of X-ray diffraction data collected in oscillation mode, In: Carter CW, Sweet RM (eds) Methods in Enzymology, vol. 276: Macromolecular crystallography, part A. Academic Press, New York, pp 307–326. https://doi.org/10.1016/S0076-6879(97)76066-X
Sheldrick GM (2015) Crystal structure refinement with SHELXL. Acta Crystallogr C: Struct Chem 71:3–8. https://doi.org/10.1107/S2053229614024218
Zhang Y, Maverick AW (2009) Preparation of an isocyano-β-diketone via its metal complexes, by use of metal ions as protecting groups. Inorg Chem 48:10512–10518. https://doi.org/10.1021/ic900202e
Pariya C, Fronczek FR, Maverick AW (2011) Bis(o-phenylenebis(acetylacetonato))-dicopper(II): a strained copper(II) dimer exhibiting a wide range of colors in the solid state. Inorg Chem 50:2748–2753. https://doi.org/10.1021/ic101641r
Pariya C, Marcos YS, Zhang Y, Fronczek FR, Maverick AW (2008) Organosilicon-based multifunctional β-diketones and their rhodium and iridium complexes. Organometallics 27:4318–4324. https://doi.org/10.1021/om701233a
Marcos, Y. PhD Dissertation, Louisiana State University, 2009. https://doi.org/10.31390/gradschool_dissertations.2660
Hamide, H. unpublished work.
Groom CR, Bruno IJ, Lightfoot MP, Ward SC (2016) The Cambridge Structural Database. Acta Crystallogr Sect B 72:171–179. https://doi.org/10.1107/S2052520616003954
Hansen PE (2021) Structural studies of β-diketones and their implications on biological effects. Pharmaceuticals 14:1189. https://doi.org/10.3390/ph14111189
Brock CP, Dunitz JD (1994) Towards a grammar of crystal packing. Chem Mater 6:1118–1127. https://doi.org/10.1021/cm00044a010
Wang J, Liu K, Ma L, Zhan X (2016) Triarylamine: versatile platform for organic, dye-sensitized, and perovskite solar cells. Chem Rev 116:14675–14725. https://doi.org/10.1021/acs.chemrev.6b00432
Reva I, Lapinski L, Chattopadhyay N, Fausto R (2003) Vibrational spectrum and molecular structure of triphenylamine monomer: a combined matrix-isolation FTIR and theoretical study. Phys Chem Chem Phys 5:3844–3850. https://doi.org/10.1039/b306489a
Hayes KS, Nagumo M, Blount JF, Mislow K (1980) Structure, optical resolution, and conformational stability of perchlorotriphenylamine. J Am Chem Soc 102:2773–2776. https://doi.org/10.1021/ja00528a043
Kim T, Mori T, Aida T, Miyajima D (2016) Dynamic propeller conformation for the unprecedentedly high degree of chiral amplification of supramolecular helices. Chem Sci 7:6689–6694. https://doi.org/10.1039/c6sc02814d
Adelizzi B, Filot IAW, Palmans ARA, Meijer EW (2017) Unravelling the pathway complexity in conformationally flexible N-centered triarylamine trisamides. Chem Eur J 23:6103–6110. https://doi.org/10.1002/chem.201603938
Acknowledgements
This research was supported in part by the Office of Basic Energy Sciences, Department of Energy, USA (Grant No. DE-FG02-01ER15267), and by the LSU West Professorship. We thank the Louisiana Board of Regents for support of the X-ray diffractometers.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception, design, material preparation, and data analysis. The first draft of the manuscript was written by JKC. All authors commented on the different versions of the manuscript, and read and approved the final version.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cherutoi, J.K., Fronczek, F.R. & Maverick, A.W. Triphenylamine-Based Bis(β-Diketones) for Construction of Supramolecular Copper(II) Complexes. J Chem Crystallogr 53, 475–482 (2023). https://doi.org/10.1007/s10870-023-00986-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-023-00986-0