Abstract
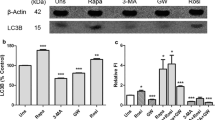
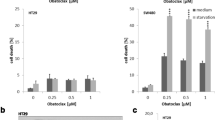
Vesicle-associated membrane protein 8 (VAMP8), a soluble n-ethylmaleimide-sensitive factor receptor protein, acts as an oncogenic gene in the progression of several malignancies. Nevertheless, the roles and mechanisms of VAMP8 in colorectal cancer (CRC) progression remain unknown. The expression and prognostic significance of VAMP8 in CRC samples were analyzed through bioinformatics analyses. Cell proliferation was detected using CCK-8 and EdU incorporation assays and apoptosis was evaluated via flow cytometry. Western blot analysis was conducted to examine the protein expression. Ferroptosis was evaluated by measurement of iron metabolism, lipid peroxidation, and glutathione (GSH) content. VAMP8 was increased in CRC samples relative to normal samples on the basis of GEPIA and HPA databases. CRC patients with high level of VAMP8 had a worse overall survival. VAMP8 depletion led to a suppression of proliferation and promotion of apoptosis in CRC cells. Additionally, VAMP8 knockdown suppressed beclin1 expression and LC3-II/LC3-I ratio, elevated p62 expression, increased Fe2+, labile iron pool, lipid reactive oxygen species, and malondialdehyde levels, and repressed GSH content and glutathione peroxidase activity. Moreover, VAMP8 knockdown inhibited the activation of janus kinase (JAK)/signal transducer and activator of transcription 3 (STAT3) pathway in CRC cells. Mechanistically, activation of the JAK/STAT3 pathway by JAK1 or JAK2 overexpression attenuated VAMP8 silencing-mediated anti-proliferative, pro-apoptotic, anti-autophagic, and pro-ferroptotic effects on CRC cells. In conclusion, VAMP8 knockdown affects the proliferation, apoptosis, autophagy, and ferroptosis by the JAK/STAT3 pathway in CRC cells.






Similar content being viewed by others
Data availability
Data are available from the corresponding author on reasonable request.
References
Amos A, Jiang N, Zong D, Gu J, Zhou J, Yin L et al (2023) Depletion of SOD2 enhances nasopharyngeal carcinoma cell radiosensitivity via ferroptosis induction modulated by DHODH inhibition. BMC Cancer 23(1):117
Anandhan A, Dodson M, Shakya A, Chen J, Liu P, Wei Y et al (2023) NRF2 controls iron homeostasis and ferroptosis through HERC2 and VAMP8. Sci Adv 9(5):eade9585
Antonin W, Holroyd C, Fasshauer D, Pabst S, Von Mollard GF, Jahn R (2000) A SNARE complex mediating fusion of late endosomes defines conserved properties of SNARE structure and function. EMBO J 19(23):6453–6464
Arends MJ (2013) Pathways of colorectal carcinogenesis. Appl Immunohistochem Mol Morphol 21(2):97–102
Behrendorff N, Dolai S, Hong W, Gaisano HY, Thorn P (2011) Vesicle-associated membrane protein 8 (VAMP8) is a SNARE (soluble N-ethylmaleimide-sensitive factor attachment protein receptor) selectively required for sequential granule-to-granule fusion. J Biol Chem 286(34):29627–29634
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68(6):394–424
Cao J, Tang Z, Su Z (2020) Long non-coding RNA LINC01426 facilitates glioblastoma progression via sponging miR-345-3p and upregulation of VAMP8. Cancer Cell Int 20:327
Chen Y, Meng D, Wang H, Sun R, Wang D, Wang S et al (2015) VAMP8 facilitates cellular proliferation and temozolomide resistance in human glioma cells. Neuro Oncol 17(3):407–418
Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F et al (2016) Cancer statistics in China, 2015. CA Cancer J Clin 66(2):115–132
Chen Q, Hao M, Wang L, Li L, Chen Y, Shao X et al (2021a) Prefused lysosomes cluster on autophagosomes regulated by VAMP8. Cell Death Dis 12(10):939
Chen X, Kang R, Kroemer G, Tang D (2021b) Broadening horizons: the role of ferroptosis in cancer. Nat Rev Clin Oncol 18(5):280–296
Chen J, Qin C, Zhou Y, Chen Y, Mao M, Yang J (2022) Metformin may induce ferroptosis by inhibiting autophagy via lncRNA H19 in breast cancer. FEBS Open Bio 12(1):146–153
De Rosa M, Pace U, Rega D, Costabile V, Duraturo F, Izzo P et al (2015) Genetics, diagnosis and management of colorectal cancer (Review). Oncol Rep 34(3):1087–1096
Dinakar YH, Kumar H, Mudavath SL, Jain R, Ajmeer R, Jain V (2022) Role of STAT3 in the initiation, progression, proliferation and metastasis of breast cancer and strategies to deliver JAK and STAT3 inhibitors. Life Sci 309:120996
Du W, Hong J, Wang YC, Zhang YJ, Wang P, Su WY et al (2012) Inhibition of JAK2/STAT3 signalling induces colorectal cancer cell apoptosis via mitochondrial pathway. J Cell Mol Med 16(8):1878–1888
Furuta N, Fujita N, Noda T, Yoshimori T, Amano A (2010) Combinational soluble N-ethylmaleimide-sensitive factor attachment protein receptor proteins VAMP8 and Vti1b mediate fusion of antimicrobial and canonical autophagosomes with lysosomes. Mol Biol Cell 21(6):1001–1010
Hasan N, Hu C (2010) Vesicle-associated membrane protein 2 mediates trafficking of alpha5beta1 integrin to the plasma membrane. Exp Cell Res 316(1):12–23
Jahn R, Scheller RH (2006) SNAREs–engines for membrane fusion. Nat Rev Mol Cell Biol 7(9):631–643
Johnson DE, O’Keefe RA, Grandis JR (2018) Targeting the IL-6/JAK/STAT3 signalling axis in cancer. Nat Rev Clin Oncol 15(4):234–248
Lao VV, Grady WM (2011) Epigenetics and colorectal cancer. Nat Rev Gastroenterol Hepatol 8(12):686–700
Lau SK, Boutros PC, Pintilie M, Blackhall FH, Zhu CQ, Strumpf D et al (2007) Three-gene prognostic classifier for early-stage non small-cell lung cancer. J Clin Oncol 25(35):5562–5569
Lech G, Słotwiński R, Słodkowski M, Krasnodębski IW (2016) Colorectal cancer tumour markers and biomarkers: recent therapeutic advances. World J Gastroenterol 22(5):1745–1755
Li X, Liu J (2023) FANCD2 inhibits ferroptosis by regulating the JAK2/STAT3 pathway in osteosarcoma. BMC Cancer 23(1):179
Li X, He S, Ma B (2020) Autophagy and autophagy-related proteins in cancer. Mol Cancer 19(1):12
Li Y, Zhang Y, Qiu Q, Wang L, Mao H, Hu J et al (2022) Energy-stress-mediated AMPK activation promotes GPX4-dependent ferroptosis through the JAK2/STAT3/p53 axis in renal cancer. Oxid Med Cell Longev 2022:2353115
Li Q, Ding Y, Ou Y, Li M, Jithavech P, Buranasudja V et al (2023) Curcuminoids modulated the IL-6/JAK/STAT3 signaling pathway in LoVo and HT-29 colorectal cancer cells. Curr Pharm Des 29(36):2867–2876
Lin Q, Lai R, Chirieac LR, Li C, Thomazy VA, Grammatikakis I et al (2005) Constitutive activation of JAK3/STAT3 in colon carcinoma tumors and cell lines: inhibition of JAK3/STAT3 signaling induces apoptosis and cell cycle arrest of colon carcinoma cells. Am J Pathol 167(4):969–980
Long J, Yao Z, Sui Y, Fang S (2022) SphK1 promotes cancer progression through activating JAK/STAT pathway and up-regulating s1pr1 expression in colon cancer cells. Anticancer Agents Med Chem 22(2):254–260
Mazzotti F, Cucchetti A, Claassen YHM, Bos A, Bastiaannet E, Ercolani G et al (2019) Years of life lost for older patients after colorectal cancer diagnosis. World J Surg 43(8):2077–2085
Ohnishi Y, Tsuji D, Itoh K (2022) Oxidative stress impairs autophagy via inhibition of lysosomal transport of VAMP8. Biol Pharm Bull 45(11):1609–1615
Peng RW, Abellan E, Fussenegger M (2011) Differential effect of exocytic SNAREs on the production of recombinant proteins in mammalian cells. Biotechnol Bioeng 108(3):611–620
Pennel KAF, Hatthakarnkul P, Wood CS, Lian GY, Al-Badran SSF, Quinn JA et al (2024) JAK/STAT3 represents a therapeutic target for colorectal cancer patients with stromal-rich tumors. J Exp Clin Cancer Res 43(1):64
Pontén F, Jirström K, Uhlen M (2008) The Human Protein Atlas–a tool for pathology. J Pathol 216(4):387–393
Rajabi S, Tahmasvand Z, Maresca M, Hamzeloo-Moghadam M (2024) Gaillardin inhibits autophagy and induces apoptosis in MCF-7 breast cancer cells by regulating JAK/STAT pathway. Mol Biol Rep 51(1):158
Rorsman P, Braun M (2013) Regulation of insulin secretion in human pancreatic islets. Annu Rev Physiol 75:155–179
Scheller RH (2013) In search of the molecular mechanism of intracellular membrane fusion and neurotransmitter release. Nat Med 19(10):1232–1235
Siegel RL, Miller KD (2019) Cancer statistics, 2019. CA Cancer J Clin 69(1):7–34
Stow JL, Murray RZ (2013) Intracellular trafficking and secretion of inflammatory cytokines. Cytokine Growth Factor Rev 24(3):227–239
Sun Y, Zheng Y, Wang C, Liu Y (2018) Glutathione depletion induces ferroptosis, autophagy, and premature cell senescence in retinal pigment epithelial cells. Cell Death Dis 9(7):753
Tang Z, Li C, Kang B, Gao G, Li C, Zhang Z (2017) GEPIA: a web server for cancer and normal gene expression profiling and interactive analyses. Nucleic Acids Res 45(W1):W98-w102
van Tol S, Atkins C, Bharaj P, Johnson KN, Hage A, Freiberg AN et al (2020) VAMP8 contributes to the TRIM6-mediated type I interferon antiviral response during west nile virus infection. J Virol 94(2):e01454-e1519
Wang Y, Wei Z, Pan K, Li J, Chen Q (2020) The function and mechanism of ferroptosis in cancer. Apoptosis 25(11–12):786–798
Wang H, Cheng Y, Mao C, Liu S, Xiao D, Huang J et al (2021) Emerging mechanisms and targeted therapy of ferroptosis in cancer. Mol Ther 29(7):2185–2208
Wang BR, Han JB, Jiang Y, Xu S, Yang R, Kong YG et al (2024) CENPN suppresses autophagy and increases paclitaxel resistance in nasopharyngeal carcinoma cells by inhibiting the CREB-VAMP8 signaling axis. Autophagy 20(2):329–348
Xiong H, Zhang ZG, Tian XQ, Sun DF, Liang QC, Zhang YJ et al (2008) Inhibition of JAK1, 2/STAT3 signaling induces apoptosis, cell cycle arrest, and reduces tumor cell invasion in colorectal cancer cells. Neoplasia 10(3):287–297
Xu J, Zhang J, Mao QF, Wu J, Wang Y (2022) The interaction between autophagy and JAK/STAT3 signaling pathway in tumors. Front Genet 13:880359
Xue X, Jungles K, Onder G, Samhoun J, Gyorffy B, Hardiman KM (2016) HIF-3alpha1 promotes colorectal tumor cell growth by activation of JAK-STAT3 signaling. Oncotarget 7(10):11567–11579
Yang S, Zhou P, Zhang L, Xie X, Zhang Y, Bo K et al (2023) VAMP8 suppresses the metastasis via DDX5/beta-catenin signal pathway in osteosarcoma. Cancer Biol Ther 24(1):2230641
Yuan M, Liao J, Luo J, Cui M, Jin F (2018) Significance of vesicle-associated membrane protein 8 expression in predicting survival in breast cancer. J Breast Cancer 21(4):399–405
Zhao P, Yang L, Lopez JA, Fan J, Burchfield JG, Bai L et al (2009) Variations in the requirement for v-SNAREs in GLUT4 trafficking in adipocytes. J Cell Sci 122(Pt 19):3472–3480
Zhao L, Zhou X, Xie F, Zhang L, Yan H, Huang J et al (2022) Ferroptosis in cancer and cancer immunotherapy. Cancer Commun (Lond) 42(2):88–116
Zhu D, Zhang Y, Lam PP, Dolai S, Liu Y, Cai EP et al (2012) Dual role of VAMP8 in regulating insulin exocytosis and islet β cell growth. Cell Metab 16(2):238–249
Funding
This work was supported by Nanyang Basic and Frontier Research Project (No. JQYJ002) and Science and Technology Plan Project of Wolong District, Nanyang (No. KJ202314).
Author information
Authors and Affiliations
Contributions
Yi Xu and Tianyao Yang conducted the experiments. Qiu Xu and Yan Tang collected and analyzed the data. Qiong Yang designed the study and wrote the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yi Xu and Tianyao Yang are co-first authors.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, Y., Yang, T., Xu, Q. et al. Vesicle-associated membrane protein 8 knockdown exerts anti-proliferative, pro-apoptotic, anti-autophagic, and pro-ferroptotic effects on colorectal cancer cells by inhibition of the JAK/STAT3 pathway. J Bioenerg Biomembr (2024). https://doi.org/10.1007/s10863-024-10019-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10863-024-10019-w