Abstract
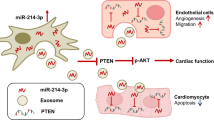
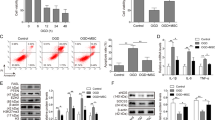
Exosomes of different origins have been found to be protective against ischemic-induced myocardial injury. This study examined the protective effects of circulating exosomes in the mice model of acute myocardial infarction (AMI) and explored the underlying molecular mechanisms. The effects of exosomes on myocardial injury were assessed in the AMI mice model. The in vivo studies showed that circulating exosomes reduced the infarcted size, improved the morphology of heart tissues and also reduced apoptosis of the heart tissues. In addition, the model mice showed an increase in the CD34 + /VEGFR2 + cell population and CD31, CXCR4 and CXCL12 expression after exosomes treatment. MiR-190a-3p was significantly down-regulated in the exosomes derived from the culture medium of hypoxia-treated human cardiomyocytes (HCMs). Further analysis revealed that miR-190a-3p could physically interact with CXCR4/CXCL12 by targeting the respective 3’UTRs. These exosomes could up-regulated CXCR4 and CXCL12 expression in the EPCs; in addition, miR-190a-3p mimics repressed CXCR4/CXCL12 expression in EPCs, while its inhibitor had opposite effects. The in vitro functional assays showed that miR-190a-3p overexpression suppressed the cell viability, proliferation, migration, adhesion and tube formation of EPCs; while miR-190a-3p inhibitor had the opposite effects; exosomes derived from the culture medium of hypoxia-treated HCMs exhibited similar actions of miR-190a-3p inhibitor. Moreover, miR-190a-3p was down-regulated in exosomes from serum in the AMI group when compared to that from sham group. Treatment with exosomes from serum in the AMI group promoted cell proliferation, migration, adhesion and tube formation of EPCs when compared to that in the sham group. More importantly, IT1t attenuated the enhanced effects of miR-190a-3p inhibition on EPC proliferation, migration, adhesion and tube formation. In conclusion, circulating exosomes exerted protective effects on myocardial injury in the AMI mice model, and down-regulation of miR-190a-3p in the circulating exosomes may exert protective effects against myocardial injury. Hypoxia induced the downregulation of miR-190a-3p in the culture medium of HCMs, and the mechanistic investigations indicated that exosomes of hypoxia-conditioned HCM culture medium promoted the cell viability, proliferation, migration, adhesion and tube formation of EPCs via regulating miR-190a-3p/CXCR4/CXCL12 pathway.








Similar content being viewed by others
Data availability
All the data are available upon reasonable request from the corresponding author.
References
Ablin JN, Boguslavski V, Aloush V, Elkayam O, Paran D, Levartovski D et al (2011) Enhanced adhesive properties of endothelial progenitor cells (EPCs) in patients with SLE. Rheumatol Int 31(6):773–778. https://doi.org/10.1007/s00296-010-1377-6
Anderson JL, Morrow DA (2017) Acute Myocardial Infarction. N Engl J Med 376(21):2053–2064. https://doi.org/10.1056/NEJMra1606915
Bajaj A, Sethi A, Rathor P, Suppogu N, Sethi A (2015) Acute Complications of Myocardial Infarction in the Current Era: Diagnosis and Management. J Investig Med 63(7):844–855. https://doi.org/10.1097/jim.0000000000000232
Bianconi V, Sahebkar A, Kovanen P, Bagaglia F, Ricciuti B, Calabrò P et al (2018) Endothelial and cardiac progenitor cells for cardiovascular repair: A controversial paradigm in cell therapy. Pharmacol Ther 181:156–168. https://doi.org/10.1016/j.pharmthera.2017.08.004
Cao C, Wang B, Tang J, Zhao J, Guo J, Guo Q et al (2021) Circulating exosomes repair endothelial cell damage by delivering miR-193a-5p. J Cell Mol Med 25(4):2176–2189. https://doi.org/10.1111/jcmm.16202
Castro-Dominguez Y, Dharmarajan K, McNamara RL (2018) Predicting death after acute myocardial infarction. Trends Cardiovasc Med 28(2):102–109. https://doi.org/10.1016/j.tcm.2017.07.011
Chen G, Xu C, Gillette TG, Huang T, Huang P, Li Q et al (2020) Cardiomyocyte-derived small extracellular vesicles can signal eNOS activation in cardiac microvascular endothelial cells to protect against Ischemia/Reperfusion injury. Theranostics 10(25):11754–11774. https://doi.org/10.7150/thno.43163
Cheng M, Yang J, Zhao X, Zhang E, Zeng Q, Yu Y et al (2019) Circulating myocardial microRNAs from infarcted hearts are carried in exosomes and mobilise bone marrow progenitor cells. Nat Commun 10(1):959. https://doi.org/10.1038/s41467-019-08895-7
Döring Y, Pawig L, Weber C, Noels H (2014) The CXCL12/CXCR4 chemokine ligand/receptor axis in cardiovascular disease. Front Physiol 5:212. https://doi.org/10.3389/fphys.2014.00212
Friedrich EB, Walenta K, Scharlau J, Nickenig G, Werner N (2006) CD34-/CD133+/VEGFR-2+ endothelial progenitor cell subpopulation with potent vasoregenerative capacities. Circ Res 98(3):e20-25. https://doi.org/10.1161/01.res.0000205765.28940.93
Huang P, Wang L, Li Q, Xu J, Xu J, Xiong Y et al (2019) Combinatorial treatment of acute myocardial infarction using stem cells and their derived exosomes resulted in improved heart performance. Stem Cell Res Ther 10(1):300. https://doi.org/10.1186/s13287-019-1353-3
Kalluri R, LeBleu VS (2020) The biology, function, and biomedical applications of exosomes. Science 367(6478). https://doi.org/10.1126/science.aau6977
King TF, McDermott JH (2014) Endothelial progenitor cells and cardiovascular disease. J Stem Cells 9(2):93–106
Lai RC, Arslan F, Lee MM, Sze NS, Choo A, Chen TS et al (2010) Exosome secreted by MSC reduces myocardial ischemia/reperfusion injury. Stem Cell Res 4(3):214–222. https://doi.org/10.1016/j.scr.2009.12.003
Li B, Bai W, Sun P, Zhou B, Hu B, Ying J (2015) The effect of CXCL12 on endothelial progenitor cells: potential target for angiogenesis in intracerebral hemorrhage. J Interferon Cytokine Res 35(1):23–31. https://doi.org/10.1089/jir.2014.0004
Liang C, Liu Y, Xu H, Huang J, Shen Y, Chen F et al (2020) Exosomes of Human Umbilical Cord MSCs Protect Against Hypoxia/Reoxygenation-Induced Pyroptosis of Cardiomyocytes via the miRNA-100-5p/FOXO3/NLRP3 Pathway. Front Bioeng Biotechnol 8:615850. https://doi.org/10.3389/fbioe.2020.615850
Lin F, Zeng Z, Song Y, Li L, Wu Z, Zhang X et al (2019) YBX-1 mediated sorting of miR-133 into hypoxia/reoxygenation-induced EPC-derived exosomes to increase fibroblast angiogenesis and MEndoT. Stem Cell Res Ther 10(1):263. https://doi.org/10.1186/s13287-019-1377-8
Mirra P, Nigro C, Prevenzano I, Procopio T, Leone A, Raciti GA et al (2017) The role of miR-190a in methylglyoxal-induced insulin resistance in endothelial cells. Biochim Biophys Acta Mol Basis Dis 1863(2):440–449. https://doi.org/10.1016/j.bbadis.2016.11.018
Mühlstedt S, Ghadge SK, Duchene J, Qadri F, Järve A, Vilianovich L et al (2016) Cardiomyocyte-derived CXCL12 is not involved in cardiogenesis but plays a crucial role in myocardial infarction. J Mol Med (Berl) 94(9):1005–1014. https://doi.org/10.1007/s00109-016-1432-1
Pegtel DM, Gould SJ (2019) Exosomes. Annu Rev Biochem 88:487–514. https://doi.org/10.1146/annurev-biochem-013118-111902
Rakocevic J, Orlic D, Mitrovic-Ajtic O, Tomasevic M, Dobric M, Zlatic N et al (2017) Endothelial cell markers from clinician’s perspective. Exp Mol Pathol 102(2):303–313. https://doi.org/10.1016/j.yexmp.2017.02.005
Raposo G, Stoorvogel W (2013) Extracellular vesicles: exosomes, microvesicles, and friends. J Cell Biol 200(4):373–383. https://doi.org/10.1083/jcb.201211138
Reed GW, Rossi JE, Cannon CP (2017) Acute myocardial infarction. Lancet 389(10065):197–210. https://doi.org/10.1016/s0140-6736(16)30677-8
Sun B, Meng M, Wei J, Wang S (2020) Long noncoding RNA PVT1 contributes to vascular endothelial cell proliferation via inhibition of miR-190a-5p in diagnostic biomarker evaluation of chronic heart failure. Exp Ther Med 19(5):3348–3354. https://doi.org/10.3892/etm.2020.8599
Thackeray JT, Derlin T, Haghikia A, Napp LC, Wang Y, Ross TL et al (2015) Molecular Imaging of the Chemokine Receptor CXCR4 After Acute Myocardial Infarction. JACC Cardiovasc Imaging 8(12):1417–1426. https://doi.org/10.1016/j.jcmg.2015.09.008
Tu TC, Nagano M, Yamashita T, Hamada H, Ohneda K, Kimura K et al (2016) A Chemokine Receptor, CXCR4, Which Is Regulated by Hypoxia-Inducible Factor 2α, Is Crucial for Functional Endothelial Progenitor Cells Migration to Ischemic Tissue and Wound Repair. Stem Cells Dev 25(3):266–276. https://doi.org/10.1089/scd.2015.0290
Wang X, Chen Y, Zhao Z, Meng Q, Yu Y, Sun J et al (2018) Engineered Exosomes With Ischemic Myocardium-Targeting Peptide for Targeted Therapy in Myocardial Infarction. J Am Heart Assoc 7(15):e008737. https://doi.org/10.1161/jaha.118.008737
White HD, Chew DP (2008) Acute myocardial infarction. Lancet 372(9638):570–584. https://doi.org/10.1016/s0140-6736(08)61237-4
Xiao M, Zeng W, Wang J, Yao F, Peng Z, Liu G et al (2021) Exosomes Protect Against Acute Myocardial Infarction in Rats by Regulating the Renin-Angiotensin System. Stem Cells Dev. https://doi.org/10.1089/scd.2020.0132
Xiong YY, Gong ZT, Tang RJ, Yang YJ (2021) The pivotal roles of exosomes derived from endogenous immune cells and exogenous stem cells in myocardial repair after acute myocardial infarction. Theranostics 11(3):1046–1058. https://doi.org/10.7150/thno.53326
Zhai TY, Cui BH, Zhou Y, Xu XY, Zou L, Lin X et al (2021) Exosomes Released from CaSR-Stimulated PMNs Reduce Ischaemia/Reperfusion Injury. Oxid Med Cell Longev 2021:3010548. https://doi.org/10.1155/2021/3010548
Zhao J, Li X, Hu J, Chen F, Qiao S, Sun X et al (2019) Mesenchymal stromal cell-derived exosomes attenuate myocardial ischaemia-reperfusion injury through miR-182-regulated macrophage polarization. Cardiovasc Res 115(7):1205–1216. https://doi.org/10.1093/cvr/cvz040
Zhao F, Cheng L, Shao Q, Chen Z, Lv X, Li J et al (2020) Characterization of serum small extracellular vesicles and their small RNA contents across humans, rats, and mice. Sci Rep 10(1):4197. https://doi.org/10.1038/s41598-020-61098-9
Zhu W, Sun L, Zhao P, Liu Y, Zhang J, Zhang Y et al (2021) Macrophage migration inhibitory factor facilitates the therapeutic efficacy of mesenchymal stem cells derived exosomes in acute myocardial infarction through upregulating miR-133a-3p. J Nanobiotechnology 19(1):61. https://doi.org/10.1186/s12951-021-00808-5
Funding
This study was supported by Shenzhen Fundamental Research Program (JCYJ20180703145002040) and Academician Shengshou Hu Youth Talent Cultivation Fund (YS-2020–007).
Author information
Authors and Affiliations
Contributions
CJ and TZ conceived the study. CJ and TZ designed the study and wrote the manuscript. CJ, LQ, YL, HZ and XH performed the experiments and analyzed the data. All the authors approved the manuscript for submission.
Corresponding author
Ethics declarations
Ethics approval
All the experimental procedures were approved by the Animal Ethics Committee of Jiangxi Provincial People's Hospital, The First Affiliated Hospital of Nanchang Medical College.
Statement of human and animal rights
Not applicable.
Statement of informed consent
Not applicable.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jiang, Cy., Zhong, Tt., Qiu, Lw. et al. The potential role of circulating exosomes in protecting myocardial injury in acute myocardial infarction via regulating miR-190a-3p/CXCR4/CXCL12 pathway. J Bioenerg Biomembr 54, 175–189 (2022). https://doi.org/10.1007/s10863-022-09944-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10863-022-09944-5