Abstract
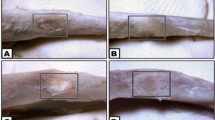
Calcium aluminate cement (CAC) as a biomaterial has been evaluated for its physical, mechanical and biocompatibility properties. Furthermore, the application of CAC for bone repair is due to its composition and coefficient of thermal expansion, which is similar to that of human bone. Thus, the aim of this study was to evaluate compositions of CAC-based blends as substitutes for bone defects. Five compositions of blends (alumina, zirconia, hydroxyapatite, tricalcium phosphate, chitosan), in addition to the base cement consisting of homogeneous CAC were evaluated as a substitute for bone repair. Additionally, the monotypic biofilm formation was assessed. Creation of a monocortical bone defect was performed on the femurs of rats, which were randomly filled with the different materials. The polymethylmethacrylate (PMMA) group was used as a control. All the animals were euthanized 04 weeks after the surgery procedure. Subsequently, computerized microtomography, histological and histomorphometric analyses were performed to verify the bone repair. To evaluate the formation of biofilms, reference strains of Staphylococcus aureus, Streptococcus mutans and Pseudomonas aeruginosa were cultured on the samples, and the biofilm formed was quantified by the MTT method. In the microtomography and histomorphometry results, it was observed that the blends exhibited better results than the control group, with statistically significant differences (p < 0.05) for alumina and zirconia blends. In the biofilm formation, a statistical difference (p < 0.05) in general was observed between the alumina blends and the control group (p < 0.05). It was concluded that CAC-based blends with alumina and zirconia are promising for use in fillings for bone repair.





Similar content being viewed by others
References
Tabata Y. Biomaterial technology for tissue engineering applications. J R Soc Interface. 2009;6:311–24. https://doi.org/10.1098/rsif.2008.0448.focus.
Ozaki W, Buckman SR. Volume maintenance of onlay bone grafts in the craniofacial skeleton: micro-architecture versus embryologic origin. Plast Reconstr Surg. 1998;102:291–9.
Prakasam M, Locs J, Salma-Ancane K, Loca D, Largeteau A, Berzina-Cimdina L. Fabrication, properties and applications of dense hydroxyapatite: a review. J Funct Biomater. 2015;4:1099–140. https://doi.org/10.3390/jfb6041099.
Boger A, Bisig A, Bohner M, Heini P, Schneider E. Variation of the mechanical properties of PMMA to suit osteoporotic cancellous bone. J Biomater Sci Polym Ed. 2008;19:1125–42. https://doi.org/10.1163/156856208785540154.
Cakarer S, Selvi F, Isler SC, Olgac V, Keskin C. Complication of polymethylmethacrylate bone cement in the mandible. Craniofac Surg. 2010;21:1196–8. https://doi.org/10.1097/SCS.0b013e3181e17b4e.
Perni S, Thenault V, Abdo P, Margulis K, Magdassi S, Prokopovich P. Antimicrobial activity of bone cements embedded with organic nanoparticles. Int J Nanomed. 2015;10:6317–29. https://doi.org/10.2147/IJN.S86440.
Parreira RM, Andrade TL, Luz AP, Pandolfellib VC, Oliveira IR. Calcium aluminate cement-based compositions for biomaterial applications. Ceram Int. 2016;42:1173238.
Garcia LFR, Huck C, Scardueli CR, Alberto C, Costa DS. Repair of bone defects filled with new calcium aluminate. J Endod Elsevier. 2015;41:864–70. https://doi.org/10.1016/j.joen.2014.12.029.
Castro-Raucci LM, Teixeira LN, Oliveira IR, Raucci-Neto W, Jacobovitz M, Rosa AL, et al. Osteogenic cell response to calcium aluminate-based cement. Int Endod J. 2017;50:771–9. https://doi.org/10.1111/iej.12682.
Acuña-Gutiérrez IO, Escobedo-Bocardo JC, Almanza-Robles JM, Cortés-Hernández DA, Saldívar-Ramírez MM, Reséndiz-Hernández PJ, Zugasti-Cruz A. Development of LiCl-containing calcium aluminate cement for bone repair and remodeling applications. Mater Sci Eng C Mater Biol Appl. 2017;70:357–63. https://doi.org/10.1016/j.msec.2016.09.022.
Moraes PC, Marques ICS, Basso FG, Rossetto HL, Pires-de-Souza FCP, Costa CAS, Garcia LDFR. Repair of bone defects with chitosan-collagen biomembrane and scaffold containing calcium aluminate cement. Braz Dent J. 2017;28:287–95.
Walsh RM, Woodmansey KF, He J, Svoboda KK, Primus CM, Opperman LA. Histology of NeoMTA plus and Quick-Set2 in contact with pulp and periradicular tissues in a canine model. J Endod. 2018;44:1389–95. https://doi.org/10.1016/j.joen.2018.05.001.
Vasconcellos LMR, Barbara MAM, Deco CP, et al. Healing of normal and osteopenic bone with titanium implant and low-level laser therapy (GaAlAs): a histomorphometric study in rats. Lasers Med Sci. 2014;29:575–80. https://doi.org/10.1007/s10103-013-1326-1.
Vasconcellos LMR, Barbara MAM, da Silva Rovai E, et al. Titanium scaffold osteogenesis in healthy and osteoporotic rats is improved by the use of low-level laser therapy (GaAlAs). Lasers Med Sci. 2016;31:899–905. https://doi.org/10.1007/s10103-017-2167-0.
Kilkenny C, Browne WJ, Cuthill IC, et al. Improving bioscience research reporting: the arrive guidelines for reporting animal research. Animals. 2013;4:35–44. https://doi.org/10.3390/ani4010035.
Connolly JF, Guse R, Tiedeman J, Dehne R. Autologous marrow injection for delayed unions of the tibia: a preliminary report. J Orth Trauma. 1989;4:276–82.
Cankaya AB, Kasapoglu MB, Erdem MA, Kasapoglu C. Effects of polymethylmethacrylate on the stability of screw fixation in mandibular angle fractures: a study on sheep mandibles. Int J Med Sci. 2018;15:1466–71. https://doi.org/10.7150/ijms.26697.
Zhang X, Kang T, Liang P, Tang Y, Quan C. Biological activity of an injectable biphasic calcium phosphate/PMMA bone cement for induced osteogensis in rabbit model. Macromol Biosci. 2018;18. https://doi.org/10.1002/mabi.201700331.
Cuijpers VMJI, Jaroszewicz J, Anil S, Al Farraj Aldosari A, Walboomers XF, Jansen JA. Resolution, sensitivity, and in vivo application of high-resolution computed tomography for titanium-coated polymethyl methacrylate (PMMA) dental implants. Clin Oral Implants Res. 2014;3:359–65. https://doi.org/10.1111/clr.12128.
Puska M, Moritz N, Aho AJ, Vallittu PK. Morphological and mechanical characterization of composite bone cement containing polymethylmethacrylate matrix functionalized with trimethoxysilyl and bioactive glass. J Mech Behav Biomed Mater. 2016;59:11–20. https://doi.org/10.1016/j.jmbbm.2015.12.016.
Cui X, Huang C, Zhang M, Ruan C, Peng S, Li L, et al. Enhanced osteointegration of poly(methylmethacrylate) bone cements by incorporating strontium-containing borate bioactive glass. J R Soc Interface. 2017;14:1–13. https://doi.org/10.1098/rsif.2016.1057.
Cimatti B, Santos MAD, Brassesco MS, Okano LT, Barboza WM, Nogueira-Barbosa MH, et al. Safety, osseointegration, and bone ingrowth analysis of PMMA-based porous cement on animal metaphyseal bone defect model. J Biomed Mater Res B Appl Biomater. 2018;106:649–58. https://doi.org/10.1002/jbm.b.33870.
Sangeetha R, Madheswari D, Priya G. Fabrication of poly (methyl methacrylate)/Ce/Cu substituted apatite/Egg white (Ovalbumin) biocomposite owning adjustable properties: towards bone tissue rejuvenation. J Photochem Photobio B. 2018;16:162–9. https://doi.org/10.1016/j.jphotobiol.2018.08.015
Castro-Raucci LMS, Teixeira LN, Barbosa AFS, Fernandes RR, Raucci-Neto W, Jacobovitz M, et al. Calcium chloride-enriched calcium aluminate cement promotes in vitro osteogenesis. Int Endod J. 2018;51:674–83. https://doi.org/10.1111/iej.12883.
Wu T, Yang S, Shi H, Ye J. Preparation and cytocompatibility of a novel bismuth aluminate/calcium phosphate cement with high radiopacity. J Mater Sci Mater Med 2018;29:149 https://doi.org/10.1007/s10856-018-6154-1.
Huck C, Barud HD, Basso FG, Costa CA, Hebling J, Garcia LD. Cytotoxicity of new calcium aluminate cement (EndoBinder) containing different radiopacifiers. Braz Dent J. 2017;28:57–64. https://doi.org/10.1590/0103-6440201701023.
Venkatesan J, Kim SK. Nano-hydroxyapatite composite biomaterials for bone tissue engineering—a review. J Biomed Nanotechnol. 2014;10:3124–40. https://doi.org/10.1166/jbn.2014.1893.
Chazono M, Tanaka T, Komaki H. Bone formation and bioresorption after implantation of injectable β-tricalcium phosphate granules—hyaluronate complex in rabbit bone defects. J Biomed Mater Res A. 2004;70:542–9.
Bellucci D, Sola A, Cannillo V. Hydroxyapatite and tricalcium phosphate composites with bioactive glass as second phase: state of the art and current applications. J Biomed Mater Res A. 2016;104:1030–56. https://doi.org/10.1002/jbm.a.35619.
Noguchi H, Funayama T, Koda M, Iijima Y, Kumagai H, Ishikawa T, Aiba A, Abe T, Nagashima K, Miura K, Izawa S, Maki S, Furuya T, Yamazaki M. A unidirectional porous beta-tricalcium phosphate material (Affinos®) for reconstruction of bony defects after excision of fibular bone for spinal surgery graft. J Clin Neurosci. 2019;66:71–6. https://doi.org/10.1016/j.jocn.2019.05.021.
Bhardwaj VA, Deepika PC, Basavarajaiah S. Zinc incorporated nano hydroxyapatite: a novel bone graft used for regeneration of intrabony defects. Contemp Clin Dent. 2018;9:427–33. https://doi.org/10.4103/ccd.ccd_192_18.
Rahmati R, Khodabakhshi F. Microstructural evolution and mechanical properties of a friction-stir processed Ti-hydroxyapatite (HA) nanocomposite. J Mech Behav Biomed Mater. 2018;88:127–39. https://doi.org/10.1016/j.jmbbm.2018.08.025.
Geng Z, Wang X, Zhao J, Li Z, Ma L, Zhu S, Liang Y, Cui Z, He H, Yang X. The synergistic effect of strontium-substituted hydroxyapatite and microRNA-21 on improving bone remodeling and osseointegration. Biomater Sci. 2018;22:1–38. https://doi.org/10.1039/c8bm00716k.
Sancilio S, Gallorini M, Di Nisio C, Marsich E, Di Pietro R, Schweikl H, et al. Alginate/hydroxyapatite-based nanocomposite scaffolds for bone tissue engineering improve dental pulp biomineralization and differentiation. Stem Cells Int. 2018;2018:1–13. https://doi.org/10.1155/2018/9643721.
Dion I, Rouais F, Baquey C, Lahaye M, Salmon R, Trut L, et al. Physico-chemistry and cytotoxicity of ceramics. J Mater Sci Mater Med. 1997;5:325–32.
Denry I, Kelly JR. State of the art of zirconia for dental applications. Dent Mater 2008;3:299–307. https://doi.org/10.1016/j.dental.2007.05.007.
Uchida M, Kim H-M, Kokubo T, Miyaji F, Nakamura T. Bonelike apatite formation induced on zirconia gel in a simulated body fluid and its modified solutions. J Am Ceram Soc. 2001;84:2041–44. https://doi.org/10.1111/j.1151-2916.2001.tb00955.x
Mobasherpour I, Solati Hashjin M, Razavi Toosi SS, Darvishi Kamachali R. Effect of the addition ZrO2-Al2O3 on nanocrystalline hydroxyapatite bending strength and fracture toughness. Ceram Int 2009;35:1569–74. https://doi.org/10.1016/j.ceramint.2008.08.017.
Askari E, Mehrali M, Metselaar IH, Kadri NA, Rahman MM. Fabrication and mechanical properties of functionally graded material by electrophoretic deposition. J Mech Behav Biomed Mater. 2012;12:144–50. https://doi.org/10.1016/j.jmbbm.2012.02.029.
Huang XN, Nicholson OS. Mechanical properties and fracture toughness of α-Al2O3-platelet-reinforced γ-PSZ composites at room and high temperatures. J Am Ceram Soc. 1993;76:1294–301. https://doi.org/10.1111/j.1151-2916.1993.tb03754.x.
Shirazi FS, Mehrali M, Oshkour AA, Metselaar HSC, Kadri NA, Abu Osman NA. Mechanical and physical properties of calcium silicate/alumina composite for biomedical engineering applications. J Mech Behav Biomed Mater. 2014;30:168–75. https://doi.org/10.1016/j.jmbbm.2013.10.024.
Dale H, Hallan G, Hallan G, Espehaug B, Havelin LI, Engesaeter LB. Increasing risk of revision due to deep infection after hip arthroplasty. Acta Orthop 2009;80:639–45. https://doi.org/10.3109/17453670903506658.
Widmer AF. New developments in diagnosis and treatment of infection in orthopedic implants. Clin Infect Dis. 2001;33:S94–106. https://doi.org/10.1086/321863.
Tappa KK, Jammalamadaka UM, Mills DK. Design and evaluation of a nanoenhanced anti-infective calcium phosphate bone cements. Conf Proc IEEE Eng Med Biol Soc. 2014;2014:3921–4. https://doi.org/10.1109/EMBC.2014.6944481.
Ikeda S, Uchiyama K, Minegishi Y, Ohno K, Nakamura M, Yoshida K, Fukushima K, Takahira N, Takaso N. Double-layered antibiotic-loaded cement spacer as a novel alternative for managing periprosthetic joint infection: an in vitro study. J Orthop Surg Res. 2018;13:322. https://doi.org/10.1186/s13018-018-1033-5.
Moraes PC, Marques ICS, Basso FG, Rossetto HL, Pires-de-Souza FCP, Costa CAS, Garcia LDFR. Repair of bone defects with chitosan-collagen biomembrane and scaffold containing calcium aluminate cement. Braz Dent J. 2017;28:287–95.
Wang D, Liu Y, Liu Y, Yan L, Zaat SAJ, Wismeijer D, et al. A dual functional bone-defect-filling material with sequential antibacterial and osteoinductive properties for infected bone defect repair. J Biomed Mater Res A. 2019. https://doi.org/10.1002/jbm.a.36744.
Bano I, Arshad M, Yasin T, Ghauri MA, Younus M. Chitosan: a potential biopolymer for wound management. Int J Biol Macromol. 2017;102:380–3. https://doi.org/10.1016/j.ijbiomac.2017.04.047.
Verlee A, Mincke S, Stevens CV. Recent developments in antibacterial and antifungal chitosan and its derivatives. Carbohydr Polym. 2017;164:268–83. https://doi.org/10.1016/j.carbpol.2017.02.001.
Acknowledgements
The authors would like to thank the Foundation for Research Support of the State of Sao Paulo (FAPESP), Brazil, and Coordination of Improvement of Higher Level Personnel (CAPES) for financial support.
Funding
This work was supported by FAPESP, Sao Paulo, Brazil (2016/15032-3 and 2017/22183-0 process), and by a CAPES scholarship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was approved by the Commission of Ethics in the Use of Animals of the Institute of Science and Technology of Unesp—Campus de São José dos Campos (CEUA/Unesp—CSJC—ICT) under number 07/2017 and the procedures were carried out according to the Ethical Principles for Animal Experimentation.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
de Vasconcellos, L.M.R., Camporês, K.L., de Alcântara Abdala, J.M. et al. Biological and microbiological behavior of calcium aluminate cement-based blend for filling of bone defects. J Mater Sci: Mater Med 31, 10 (2020). https://doi.org/10.1007/s10856-019-6348-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10856-019-6348-1