Abstract
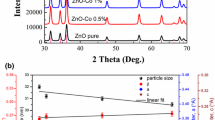
Pristine ZnO and iron-doped ZnO (x = 0, 0.04, 0.08, 0.10, and 0.15) nanoparticles were produced by means of precipitation route. The nanoparticles were subjected to thermogravimetric analysis. Both EDX and XPS were employed to analyze the chemical structure of samples. The XRD pattern shows that the hexagonal structure of iron-doped ZnO nanoparticles is known, and this suggests that Fe2 + can substitute Zn2 + in the lattice of ZnO without the discovery of another precipitated phase. As the amount of Fe material increases, the lattice strain rises from 0.0277 to 0.0379, even though a crystallite size falls from 38 to 28 nm, x is equal 0, and 0.15, respectively. The only factors that significantly affect the shrinking of crystallite size are preparation conditions and structural disorder. In terms of UV–vis absorption spectroscopy, the energy gap, \(E_{g}^{opt}\) value obtained from the derivative of absorbance concerning wavelength and found to be reduced from 3.41 eV when x of around 0 and 3.1 eV when x of around 0.15 with rising Fe content. The photoluminescence spectrometry can produce wide PL peaks because of the numerous recombination faults and sites. The presence of additional peaks from an intrinsic emission is thought to be the cause of the PL's asymmetric spectrum. The M-H measurements demonstrated RT ferromagnetism in the Fe-doped ZnO nanoparticles by vibrating the sample in a magnetometer. Additionally, the coercive field increases from 59.9 G to 85.3 G and the remanent magnetization increases from 3.4 × 10–3 to 21 × 10–3 emu/g when the iron content rises from (x of around 0.02) to (x of around 0.15). By improving the magnetic and optical properties of samples, these findings enable the use of iron-doped ZnO nanoparticles in optoelectronic and spintronic applications.














Similar content being viewed by others
Data availability
The Data will be available when required from the corresponding author.
References
S.J. Pearton et al., ZnO doped with transition metal ions. IEEE Trans. Electron Devices 54(5), 1040–1048 (2007)
A. Moezzi, A.M. McDonagh, M.B. Cortie, Zinc oxide particles: synthesis, properties and applications. Chem. Eng. J. 185, 1–22 (2012)
D. Chu et al., Formation and photocatalytic application of ZnO nanotubes using aqueous solution. Langmuir 26(4), 2811–2815 (2010)
J. Zhang et al., Hierarchically porous ZnO architectures for gas sensor application. Cryst. Growth Des. 9(8), 3532–3537 (2009)
X.J. Liu et al., Intrinsic and extrinsic origins of room temperature ferromagnetism in Ni-doped ZnO films. J. Phys. D Appl. Phys. 42(3), 035004 (2009)
J.R. Neal et al., Room-temperature magneto-optics of ferromagnetic transition-metal-doped ZnO thin films. Phys. Rev. Lett. 96(19), 197208 (2006)
X. Huang et al., Morphology evolution and CL property of Ni-doped zinc oxide nanostructures with room-temperature ferromagnetism. J. Phys. Chem. C 113(11), 4381–4385 (2009)
S. Benramache et al., Study on the correlation between crystallite size and optical gap energy of doped ZnO thin film. J. Nanostruct. Chem. 3(1), 1–6 (2013)
Z.H. Zhang et al., Evidence of intrinsic ferromagnetism in individual dilute magnetic semiconducting nanostructures. Nat. Nanotechnol. 4(8), 523–527 (2009)
N.H. Hong, J. Sakai, V. Brizé, Observation of ferromagnetism at room temperature in ZnO thin films. J. Phys.: Condens. Matter 19(3), 036219 (2007)
R. Bhargava et al., Variation in structural, optical and magnetic properties of Zn1− xCrxO (x= 0.0, 0.10, 0.15, and 0.20) nanoparticles: role of dopant concentration on non-saturation of magnetization. Mater. Chem. Phys. 125(3), 664–671 (2011)
F.A. Taher, E. Abdeltwab, Shape-controlled synthesis of nanostructured Co-doped ZnO thin films and their magnetic properties. CrystEngComm 20(38), 5844–5856 (2018)
A.K. Mishra, D. Das, Investigation on Fe-doped ZnO nanostructures prepared by a chemical route. Mater. Sci. Eng. B 171(1–3), 5–10 (2010)
X.X. Wei et al., Local Fe structure and ferromagnetism in Fe-doped ZnO films. J. Phys.: Condens. Matter 18(31), 7471 (2006)
A. Hewat, W.I.F. David, L. van Eijck, Hugo Rietveld (1932–2016). J. Appl. Crystallog. 49(4), 1394–1395 (2016)
A.F. Al Naim, A. Solieman, E.R. Shaaban, Structural, optical, and magnetic properties of Co-doped ZnO nanocrystalline thin films for spintronic devices. J. Mater. Sci.: Mater. Electron. 31(4), 3613–3621 (2020)
A.C. Galca, G. Socol, V. Craciun, Optical properties of amorphous-like indium zinc oxide and indium gallium zinc oxide thin films. Thin Solid Films 520(14), 4722–4725 (2012)
P.-Y. Kim et al., Structure and properties of IZO transparent conducting thin films deposited by PLD method. J. Korean Phys. Soc. 53(1), 207–211 (2008)
B.D. Cullity, Elements of X-ray diffraction, 2nd edn. (Addition-Weasley, London, 1978)
J.I. Langford, A.J.C. Wilson, J. Appl. Cryst. 11, 102–113 (1978)
M. Ghosh et al., Metal-ion displacement approach for optical recognition of thorium: application of a molybdenum (VI) complex for nanomolar determination and enrichment of Th (IV). ACS Omega 3(11), 16089–16098 (2018)
T. Srinivasulu, K. Saritha, K.T.R. Reddy, Synthesis and characterization of Fe-doped ZnO thin films deposited by chemical spray pyrolysis. Mod. Electron. Mater. 3(2), 76–85 (2017)
X. Gong et al., Engineering high-emissive silicon-doped carbon nanodots towards efficient large-area luminescent solar concentrators. Nano Energy 101, 107617 (2022)
M. El-Hagary et al., The particle size-dependent optical band gap and magnetic properties of Fe-doped CeO2 nanoparticles. Solid State Sci. 91, 15–22 (2019)
T.A. Abdel-Baset et al., Structural and magnetic properties of transition-metal-doped Zn 1–x Fe x O. Nanoscale Res. Lett. 11(1), 1–12 (2016)
A. Popa et al., Interplay between ferromagnetism and photocatalytic activity generated by Fe3+ ions in iron doped ZnO nanoparticles grown on MWCNTs. Physica E: Low Dimens. Syst. Nanostruct. 129, 114581 (2021)
J.J. Beltrán, C.A. Barrero, A. Punnoose, Understanding the role of iron in the magnetism of Fe doped ZnO nanoparticles. Phys. Chem. Chem. Phys. 17(23), 15284–15296 (2015)
M. Zahan, J. Podder, Role of Fe doping on structural and electrical properties of MnO2 nanostructured thin films for glucose sensing performance. Mater. Sci. Semicond. Process. 117, 105109 (2020)
M.M. Ovhal et al., Photoluminescence quenching and enhanced spin relaxation in Fe doped ZnO nanoparticles. Mater. Chem. Phys. 195, 58–66 (2017)
Acknowledgements
The authors would like to express their gratitude to Princess Nourah bint Abdulrahman University Researchers Supporting Project (grant no. PNURSP2022R32), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia. RSM: Conceptualization, Data curation, Writing—original draft. AAEl: Validation, Formal analysis. AAAS: Formal analysis, Writing—review & editing. BMA: review & editing, AA: review & editing, AMA: Investigation, Writing—review & editing. ERS: Supervision, Resources.
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have not disclosed any competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mahmoud, R.S., El-Fadl, A.A., Abu-Sehly, A.A. et al. Structural, optical, and magnetic properties of iron-doped ZnO nanoparticles for optoelectronics. J Mater Sci: Mater Electron 33, 23484–23494 (2022). https://doi.org/10.1007/s10854-022-09108-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-022-09108-6