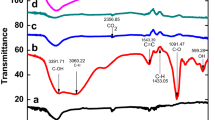
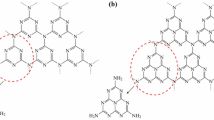
Abstract
Reduced graphene oxide (rGO) was obtained from graphene oxide (GO) by hydrothermal method. In order to easily synthesize hybrid compounds (HB1 and HB2), the carboxyl groups on the surface of rGO were converted to acyl chloride with SOCl2 and rGO-COCl was obtained. To use in the synthesize of the hybrids, two new carboxylic acid substituted multifunctional imine compounds ({(E)-4-((2-hydroxy-4-((1-(p-tolyl)-1 H-1,2,3-triazol-4-yl)methoxy) benzylidene)amino)benzoic acid (5), (E)-4-((2-hydroxy-4-((1-(4-methoxyphenyl)-1 H-1,2,3-triazol-4-yl)methoxy)benzylidene)amino)benzoic acid (6)}) were obtained and characterized by the 1H and 13C(1H)NMR, FT-IR, MALDI-TOF-MS techniques. The hybrids were obtained from the reaction of the imine and rGO-Cl compounds and characterized by the FT-IR, UV–Vis, emission (PL), X-ray diffraction (XRD), SEM-EDX and TEM methods. The chemosensor properties of the hybrids were investigated towards some metal ions (Al3+, Ag+, Co2+, Cr3+, Cu2+, Fe3+, Hg2+, Mn2+, Ni2+, Zn2+, Cd2+, Pb2+). At the short wavelength light, HB1 showed a blue sensor against Ag+, Co2+, Cu2+, Fe3+, Ni2+ and Pb2+ ions, dark green against Al3+, Zn2+ and Cd2+ ions, and purple-colored sensors against Hg2+ ions. HB2 hybrid material exhibited sensor properties against Ag+, Cr3+, Cu2+, Fe3+, Zn2+ and Cd2+ ions. At the long wavelength light, HB1 and HB2 materials showed sensory properties against Ag+, Cr3+, Cu2+, Fe3+, Zn2+ and Cd2+ ions. The hybrid HB1 has selective chemosensing properties against Al3+ and Hg2+ ions at the short wavelength light. In addition, the 1931 CIE (x,y) chromaticity coordinates of the hybrids and imine compounds were investigated and the x, y values of the compounds were found to be close to the National Television Standard Committee (NTSC) standard.












Similar content being viewed by others
Data availability
The data that supports the findings of this study are available in the supplementary material of this article.
References
S. Goenka, V. Sant, S. Sant, J. Controlled Release 173, 75 (2014)
W. Gao, Graphene Oxide (Springer International Publishing, Cham, 2015), pp. 61–95
V. Galstyan, E. Comini, I. Kholmanov, A. Ponzoni, V. Sberveglieri, N. Poli, G. Faglia, G. Sberveglieri, Beilstein J. Nanotechnol. 7, 1421 (2016)
A.P. De Silva, T.S. Moody, G.D. Wright, Analyst 134, 2385 (2009)
V. Bojinov, N. Georgiev, J. Univ. Chem. Technol. Metall. 46, 3 (2011)
H. Jiang, Small 7, 2413 (2011)
A.L. Berhanu, I. Gaurav, A.K. Mohiuddin, J.S. Malik, V. Aulakh, Kumar, K.H. Kim, TrAC - Trends in Analytical Chemistry 116, 74 (2019)
W.S. Hummers, R.E. Offeman, J. Am. Chem. Soc. 80, 1339 (1958)
S.N. Alam, N. Sharma, L. Kumar, Graphene 06, 1 (2017)
X. Mei, X. Meng, F. Wu, E. Physica, Low-Dimensional Syst. Nanostruct. 68, 81 (2015)
A. Koohi, R. Rahimi, R. Zare-Dorabei, S. Zargari, Proc. Int. ECOC 1 (2014)
S. Zhou, H. Liao, M. Liu, G. Feng, B. Fu, R. Li, M. Cheng, Y. Zhao, P. Gong, Bioorg. Med. Chem. 22, 6438 (2014)
R. Kumar, J. Arora, A.K. Prasad, N. Islam, A.K. Verma, Med. Chem. Res. 22, 5624 (2013)
A. Koohi, R. Rahimi, R. Zare-Dorabei, S. Zargari, in Proceedings of The 18th International Electronic Conference on Synthetic Organic Chemistry (MDPI, Basel, Switzerland, 2014), p. a017
M. Bala Murali Krishna, N. Venkatramaiah, R. Venkatesan, D. Narayana Rao, J. Mater. Chem. 22, 3059 (2012)
L. Shahriary, A.a. Athawale, Int. J. Renew. Energy Environ. Eng. 02, 58 (2014)
C. Wang, J. Zhou, L. Chu, RSC Adv. 5, 52466 (2015)
S. Rayati, E. Khodaei, S. Shokoohi, M. Jafarian, B. Elmi, A. Wojtczak, Inorg. Chim. Acta 466, 520 (2017)
S. Omidi, A. Kakanejadifard, F. Azarbani, J. Mol. Liq. 242, 812 (2017)
H. Peng, L. Meng, L. Niu, Q. Lu, (2012)
D. Li, M.B. Müller, S. Gilje, R.B. Kaner, G.G. Wallace, Nat. Nanotechnol. 3, 101 (2008)
H.A.S. Tohamy, B. Anis, M.A. Youssef, A.E.M. Abdallah, M. El-Sakhawy, S. Kamel, Desalination Water Treat. 191, 250 (2020)
M. Karabörk, B.A. Muhammed, M. Tümer, S. Uruş, Phosphorus, Sulfur and Silicon and the Related Elements 197, 133 (2022)
S. Nasrazadani, S. Hassani, Modern Analytical Techniques in Failure Analysis of Aerospace, Chemical, and Oil and Gas Industries (Elsevier Ltd., 2016)
L. Stobinski, B. Lesiak, A. Malolepszy, M. Mazurkiewicz, B. Mierzwa, J. Zemek, P. Jiricek, I. Bieloshapka, J. Electron Spectrosc. Relat. Phenom. 195, 145 (2014)
S. Gurunathan, J.W. Han, J.H. Park, E. Kim, Y.J. Choi, D.N. Kwon, J.H. Kim, Int. J. Nanomed. 10, 6257 (2015)
L. Han, C.M. Liu, S.L. Dong, C.X. Du, X.Y. Zhang, L.H. Li, Y. Wei, Biosens. Bioelectron. 87, 466 (2017)
J. Jose, A.R. Rajamani, S. Anandaram, S.P. Jose, S.C. Peter, P.B. Sreeja, Appl Organomet Chem 33, 5 (2019)
T. Serodre, N.A.P. Oliveira, D.R. Miquita, M.P. Ferreira, A.P. Santos, V.G. Resende, C.A. Furtado, J. Braz. Chem. Soc. 30, 2488 (2019)
M. Bera, P. Chandravati, Gupta, P.K. Maji, J. Nanosci. Nanotechnol. 18, 902 (2017)
V. Selvarani, M.A. Neelakantan, V. Silambarasan, D. Velmurugan, Acta Crystallogr. Sect. E: Struct. Rep. Online 69, o64 (2013)
C. Zhu, S. Guo, Y. Fang, S. Dong, ACS Nano 4, 2429 (2010)
D. Majumdar, N. Baugh, S.K. Bhattacharya, Colloids Surf., A 512, 158 (2017)
S.K. Pradhan, B. Xiao, S. Mishra, A. Killam, A.K. Pradhan, Sci. Rep. 6, 1 (2016)
P. Bansal, A.S. Panwar, D. Bahadur, Int. J. Mater. Mech. Manuf. 2, 18 (2014)
D. Wang, X. Zhang, J.W. Zha, J. Zhao, Z.M. Dang, G.H. Hu, Polymer 54, 1916 (2013)
J. Yang, X. Zhao, X. Shan, H. Fan, L. Yang, Y. Zhang, X. Li, J. Alloys Compd. 556, 1 (2013)
N.M.S. Hidayah, W.W. Liu, C.W. Lai, N.Z. Noriman, C.S. Khe, U. Hashim, H.C. Lee, AIP Conf. Proc. 1892, (2017)
H. Kırpık, M. Kose, J.N. Ballı, Appl. Organomet. Chem. 34, 1 (2020)
Acknowledgements
We are grateful to KSU, Scientific Research Projects Coordination Unit (BAP) (Project number: 2020/3-18D) for the support of this research.
Author information
Authors and Affiliations
Contributions
MB: conceptualization, methodology, software, writing, MT: supervision, SU: data curation.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bal, M., Tümer, M. & Uruş, S. Heterocycled triazole and azomethine substituted multifunctional graphene based hybrid ligands: color and sensor properties. J Mater Sci: Mater Electron 33, 14001–14020 (2022). https://doi.org/10.1007/s10854-022-08331-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-022-08331-5