Abstract
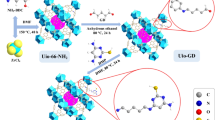
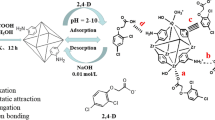
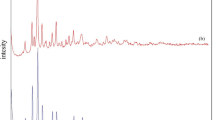
Removal of ciprofloxacin (CIP) from wastewater is of significance to protect the environment. In this study, phosphomolybdic acid (H3PMo12O40) -modified metal–organic framework (UiO-66-NH2) materials were prepared by the solvothermal method for the removal of CIP from wastewater. The adsorption performance of H3PMo12O40@ UiO-66-NH2 on CIP was remarkably superior to that of pure UiO-66-NH2. Several experiments with altered conditions, including CIP concentration, contact time, and pH, were explored to understand the adsorption performance of H3PMo12O40@ UiO-66-NH2 on CIP solutions. The experimental results show that when pH = 7, T = 323 K, the amount of H3PMo12O40 is 0.14 g, which is the best CIP adsorption condition for H3PMo12O40@UiO-66-NH2, and the adsorption amount is 164.28 mg g−1. The adsorption kinetics and isotherms of CIP molecules on the modified UiO-66-NH2 adsorbent showed that they were basically in agreement with the pseudo-second-order kinetic model and Langmuir model, respectively. Thermodynamic parameters indicate that the adsorption process is spontaneous endothermic. After five cycles of experiments, the adsorbent can still remove 81% of the CIP, indicating that it is reproducible and practical for CIP. Hence, the H3PMo12O40@ UiO-66-NH2 has potential application value as an antibiotic’s adsorbent.












Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings of this study are available within the article.
References
Y. Xiang, X. Yang, Z. Xu, W. Hu, Y. Zhou, Z. Wan, Y. Yang, Y. Wei, J. Yang, D.C. Tsang, Sci. Total Environ. 709, 136079 (2020)
B. Jubeh, Z. Breijyeh, R. Karaman, Molecules 25, 2888 (2020)
D.K. Yoo, B.N. Bhadra, S.H. Jhung, J. Hazard. Mater. 403, 123655 (2021)
J.M. Park, S.H. Jhung, Chem. Eng. J. 396, 125224 (2020)
F.M. Mpatani, A.A. Aryee, A.N. Kani, R. Han, Z. Li, E. Dovi, L. Qu, J. Cleaner Prod. 127359 (2021).
I.A. Ricardo, E.A. Alberto, A.H.S. Júnior, D.L.P. Macuvele, N. Padoin, C. Soares, H.G. Riella, M.C.V. Starling, A.G. Trovó, Chem. Eng. J. 130282 (2021).
S.O. Ganiyu, E.D. Van Hullebusch, M. Cretin, G. Esposito, M.A. Oturan, Sep. Purif. Technol. 156, 891–914 (2015)
V. Homem, L. Santos, J. Environ. Manage. 92, 2304–2347 (2011)
N.A. Khan, S.U. Khan, S. Ahmed, I.H. Farooqi, A. Dhingra, A. Hussain, F. Changani, Asian J. Water Environ. Pollut. 16, 81–86 (2019)
I. Cota, F.F. Martinez, Coord. Chem. Rev. 351, 189–204 (2017)
F.E. Che Othman, N. Yusof, J. González-Benito, X. Fan, A.F. Ismail, Polymers 12, 2117 (2020)
B. Maazinejad, O. Mohammadnia, G.A. Ali, A.S. Makhlouf, M.N. Nadagouda, M. Sillanpää, A.M. Asiri, S. Agarwal, V.K. Gupta, H. Sadegh, J. Mol. Liq. 298, 112001 (2020)
Y. Li, X. Yan, X. Hu, R. Feng, M. Zhou, Chem. Eng. J. 375, 122003 (2019)
A. Abd-Elhamid, E.A. Kamoun, A.A. El-Shanshory, H.M. Soliman, H. Aly, J. Mol. Liq. 279, 530–539 (2019)
S. Soni, P. Bajpai, J. Mittal, C. Arora, J. Mol. Liq. 314, 113642 (2020)
K. Zhang, F. Tsai, N. Ma, Y. Xia, H. Liu, X. Zhan, X. Yu, X. Zeng, T. Jiang, D. Shi, Materials 10, 205 (2017)
M. Massoudinejad, M. Ghaderpoori, A. Shahsavani, A. Jafari, B. Kamarehie, A. Ghaderpoury, M.M. Amini, J. Mol. Liq. 255, 263–268 (2018)
D. Sun, L. Ye, Z. Li, Appl. Catal. B-Environ. 164, 428–432 (2015)
X. Zhang, W. Wang, Z. Hu, G. Wang, K. Uvdal, Coord. Chem. Rev. 284, 206–235 (2015)
Y. Jiang, C. Liu, J. Caro, A. Huang, Microporous Mesoporous Mater. 274, 203–211 (2019)
X. Li, S.C. Pillai, L. Wei, Z. Liu, L. Huang, Q. Huang, X. Jia, D. Hou, H. Song, H. Wang, J. Hazard. Mater. 399, 122946 (2020)
S. Subudhi, S. Mansingh, G. Swain, A. Behera, D. Rath, K. Parida, Inorg. Chem. 58, 4921–4934 (2019)
B. Zhang, X. Han, P. Gu, S. Fang, J. Bai, J. Mol. Liq. 238, 316–325 (2017)
G. Wu, J. Ma, S. Li, J. Guan, B. Jiang, L. Wang, J. Li, X. Wang, L. Chen, J. Colloid Interface Sci. 528, 360–371 (2018)
F. Yu, S. Sun, S. Han, J. Zheng, J. Ma, Chem. Eng. J. 285, 588–595 (2016)
C. Liang, X. Zhang, P. Feng, H. Chai, Y. Huang, Chem. Eng. J. 344, 95–104 (2018)
A.K. Kushwaha, N. Gupta, M. Chattopadhyaya, J. Saudi Chem. Soc. 18, 200–207 (2014)
Z. Aghaei, H. Vojoudi, J.B. Ghasemi, S. Bahar, A. Badiei, Microchem J. 157, 104878 (2020)
S. Dadfarnia, A.H. Shabani, S. Moradi, S. Emami, Appl. Surf. Sci. 330, 85–93 (2015)
J. Fu, Q. Xin, X. Wu, Z. Chen, Y. Yan, S. Liu, M. Wang, Q. Xu, J. Colloid Interface Sci. 461, 292–304 (2016)
A. Olajire, A. Giwa, I. Bello, Int. J. Environ. Sci. Technol. 12, 939–950 (2015)
C. Li, X. Wang, D. Meng, L. Zhou, Int. J. Biol. Macromol. 107, 1871–1878 (2018)
E.C. Lima, A. Hosseini-Bandegharaei, J.C. Moreno-Piraján, I. Anastopoulos, J. Mol. Liq. 273, 425–434 (2019)
X. Fang, S. Wu, Y. Wu, W. Yang, Y. Li, J. He, P. Hong, M. Nie, C. Xie, Z. Wu, Appl. Surf. Sci. 518, 146226 (2020)
Q. Chen, Q. He, M. Lv, Y. Xu, H. Yang, X. Liu, F. Wei, Appl. Surf. Sci. 327, 77–85 (2015)
X. He, F. Deng, T. Shen, L. Yang, D. Chen, J. Luo, X. Luo, X. Min, F. Wang, J. Colloid Interface Sci. 539, 223–234 (2019)
Acknowledgements
This work was kindly supported by the National Natural Science Foundation of China (21171053), Natural Science Foundation of Hubei Province, China (2021J089), Natural Science Foundation of Hubei Province Education Committee, China (D20213102), Graduate Student Innovation Research Foundation of Hubei Normal University (20210116).
Author information
Authors and Affiliations
Contributions
HH: conceptualization, methodology, data curation, writing-original draft. QL: investigation, data curation, validation. SYF: software, validation. XJL: software, supervision. YY: data curation, funding acquisition. SJY: writing-review and editing, supervision, funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hu, H., Lv, Q., Feng, SY. et al. Solvothermal synthesis of polyoxometalate-modified UiO-66-NH2 for enhanced removal of ciprofloxacin from aqueous solution. J Mater Sci: Mater Electron 33, 4184–4196 (2022). https://doi.org/10.1007/s10854-021-07614-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-021-07614-7