Abstract
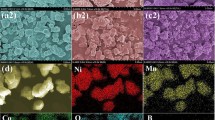
Boron-modified Li(Ni0.8Co0.1Mn0.1)1−xBxO2 cathode materials(NCM811) were successfully prepared by a nano-milling assisted solid-state approach. X-ray diffraction investigations showed that the materials are solid solutions with a layered structure. SEM observations implied that the doped B ions promoted the growth of the target crystal with well-developed facets since it will form liquid phase at lower temperature. The intensity ratio of I(003)/I(104) raised with the increase in Boron doping concentration, until a maximum value of 1.453 was observed at x = 0.01. Further Rietveld refinements revealed that boron ions occupy the crystal lattice in the transition metal slab which helps to promote the lattice ordering by decreasing the Li/Ni ionic mixing. Such B promoted NCM811 cathode materials were confirmed to have an improved diffusion coefficient with a reduced interfacial resistance by subsequent CV and EIS measurements. From the electrochemical test, those B modified NCM811 cathode materials presented enhanced electrochemical performance. Among the synthesized samples, Li(Ni0.8Co0.1Mn0.1)0.99B0.01O2 exhibited the best specific capacity, with 194.7 mAh g−1 and 166.8 mAh g−1 at 0.1C and 5C respectively. The capacity retention at 0.5C was also confirmed as 98.2% after 100 cycles. Such improvement can be explained by the reduced Li/Ni ionic mixing, the increased Li ionic diffusion and the reduced interfacial resistance caused by the promoted growth of the B doped NCM811 crystals. Compared to those NCM811 materials reported elsewhere, the material obtained by this approach showed high potential for future application.






Similar content being viewed by others
References
N. Recham, J.N. Chotard, L. Dupont, C. Delacourt, W. Walker, M. Armand, J.M. Tarascon, Nat. Mater. (2009). https://doi.org/10.1038/nmat2590
Y.K. Sun, S.T. Myung, B.C. Park, J. Prakash, I. Belharouak, K. Amine, Nat. Mater. (2009). https://doi.org/10.1038/nmat2418
Y.K. Sun, Z. Chen, H.J. Noh, D.J. Lee, H.G. Jung, Y. Ren, S. Wang, C.S. Yoon, S.T. Myung, K. Amine, Nat. Mater. (2012). https://doi.org/10.1038/nmat3435
G. Derrien, J. Hassoun, S. Panero, B. Scrosati, Adv. Mater. (2007). https://doi.org/10.1002/adma.200700748
Z.M. Yu, L.C. Zhao, T. Nonferr, Metal. Soc. (2007). https://doi.org/10.1016/S1003-6326(07)60152-6
M.H. Kim, H.S. Shin, D. Shin, Y.K. Sun, J. Power Sources (2006). https://doi.org/10.1016/j.jpowsour.2005.11.083
S.M. Bak, E. Hu, Y. Zhou, X. Yu, S.D. Senanayake, S.J. Cho, K.B. Kim, K.Y. Chung, X.Q. Yang, K.W. Nam, ACS Appl. Mater. Interfaces. (2014). https://doi.org/10.1021/am506712c
K. Min, K. Kim, C. Jung, S.W. Seo, Y.Y. Song, H.S. Lee, J. Shin, E. Cho, J. Power Sources (2016). https://doi.org/10.1016/j.jpowsour.2016.03.017
S. Gao, X. Zhan, Y.T. Cheng, J. Power Sources (2019). https://doi.org/10.1016/j.jpowsour.2018.10.094
Z. Huang, Z. Wang, X. Zheng, H. Guo, X. Li, Q. Jing, Z. Yang, RSC Adv. (2015). https://doi.org/10.1039/c5ra16633k
L. Liu, K. Sun, N. Zhang, T. Yang, J. Solid State Electrochem. (2008). https://doi.org/10.1007/s10008-008-0695-z
K. Min, S.W. Seo, Y.Y. Song, H.S. Lee, E. Cho, Phys. Chem. Chem. Phys. (2017). https://doi.org/10.1039/c6cp06270a
S.W. Woo, S.T. Myung, H. Bang, D.W. Kim, Y.K. Sun, Electrochim. Acta (2009). https://doi.org/10.1016/j.electacta.2009.01.048
M. Eilers-Rethwisch, M. Winter, F.M. Schappacher, J. Power Sources (2018). https://doi.org/10.1016/j.jpowsour.2018.02.080
L.J. Li, X.H. Li, Z.X. Wang, H.J. Guo, P. Yue, W. Chen, L. Wu, J. Alloys Compd. (2010). https://doi.org/10.1016/j.jallcom.2010.07.148
R. Zhao, Z. Yang, J. Liang, D. Lu, C. Liang, X. Guan, A. Gao, H. Chen, J. Alloys Compd. (2016). https://doi.org/10.1016/j.jallcom.2016.07.230
Z. Huang, Z. Wang, Q. Jing, H. Guo, X. Li, Z. Yang, Electrochim. Acta (2016). https://doi.org/10.1016/j.electacta.2016.01.139
X. Li, K. Zhang, M. Wang, Y. Liu, M. Qu, W. Zhao, J. Zheng, Sustain Energy Fuels (2018). https://doi.org/10.1039/c7se00513j
Q. Chen, C. Du, D. Qu, X. Zhang, Z. Tang, RSC Adv. (2015). https://doi.org/10.1039/c5ra14376d
F. Schipper, M. Dixit, D. Kovacheva, M. Talianker, O. Haik, J. Grinblat, E.M. Erickson, C. Ghanty, D.T. Major, B. Markovsky, D. Aurbach, J. Mater. Chem. A (2016). https://doi.org/10.1039/c6ta06740a
C. Qin, J. Cao, J. Chen, G. Dai, T. Wu, Y. Chen, Y. Tang, A. Li, Y. Chen, Dalton Trans. (2016). https://doi.org/10.1039/c6dt01764a
L. Pan, Y. Xia, B. Qiu, H. Zhao, H. Guo, K. Jia, Q. Gu, Z. Liu, J. Power Sources (2016). https://doi.org/10.1016/j.jpowsour.2016.07.064
K. Saravanan, M.V. Reddy, P. Balaya, H. Gong, B.V.R. Chowdari, J.J. Vittal, J. Mater. Chem. (2009). https://doi.org/10.1039/b817242k
G. Arnold, J. Garche, R. Hemmer, S. Ströbele, C. Vogler, M. Wohlfahrt-Mehrens, J. Power Sources (2003). https://doi.org/10.1016/s0378-7753(03)00241-6
R. Dominko, M. Bele, J.M. Goupil, M. Gaberscek, D. Hanzel, I. Arcon, J. Jamnik, Chem. Mater. (2007). https://doi.org/10.1021/cm062843g
Y. Zhou, J. Wang, Y. Hu, R. O’Hayre, Z. Shao, Chem. Commun. (2010). https://doi.org/10.1039/c0cc01721c
N. Recham, L. Dupont, M. Courty, K. Djellab, D. Larcher, M. Armand, J.M. Tarascon, Chem. Mater. (2009). https://doi.org/10.1021/cm803259x
L. Guan, P. Xiao, T.J. Lv, D.Y. Zhang, C.K. Chang, J. Electrochem. Soc. (2017). https://doi.org/10.1149/2.1731713jes
T.J. Lv, L. Guan, P. Xiao, D.Y. Zhang, C.K. Chang, J. Mater. Sci. (2019). https://doi.org/10.1007/s10853-018-03194-w
T. Ohzuku, A. Ueda, M. Nagayama, Y. Iwakoshi, H. Komori, Electrochim. Acta (1993). https://doi.org/10.1016/0013-4686(93)80046-3
X.T. Yin, W.D. Zhou, J. Li, P. Lv, Q. Wang, D. Wang, F.Y. Wu, D. Dastan, H. Garmestani, Z. Shi, S. Ţălu, J. Mater. Sci. (2019). https://doi.org/10.1007/s10854-019-01840-w
D. Dastan, Appl. Phys. A (2017). https://doi.org/10.1007/s00339-017-1309-3
D. Dastan, J. At. Mol. Condens. Nano Phys. 2, 109–119 (2015)
Z.L. Zhang, D.H. Chen, C.K. Chang, RSC Adv. (2017). https://doi.org/10.1039/c7ra10053a
P. Xiao, T.J. Lv, X.P. Chen, C.K. Chang, Sci. Rep. (2017). https://doi.org/10.1038/s41598-017-01657-9
Y. Zhao, L. Peng, B. Liu, G. Yu, Nano Lett. (2014). https://doi.org/10.1021/nl5008568
M.D. Levi, J. Electrochem. Soc. (1999). https://doi.org/10.1149/1.1391759
X. Wu, S.H. Chang, Y.J. Park, K.S. Ryu, J. Power Sources (2004). https://doi.org/10.1016/j.jpowsour.2004.05.043
M. Zhang, H. Zhao, M. Tan, J. Liu, Y. Hu, S. Liu, X. Shu, H. Li, Q. Ran, J. Cai, X. Liu, J. Alloys Compd. (2019). https://doi.org/10.1016/j.jallcom.2018.09.281
M.X. Dong, X.Q. Li, Z.X. Wang, X.H. Li, H.J. Guo, Z.J. Huang, T. Nonferr, Metal. Soc. (2017). https://doi.org/10.1016/S1003-6326(17)60132-8
Funding
The research was supported by Science and Technology Commission of Shanghai Municipality (14520503100 and 201310-JD-B2-009) and Shanghai Municipal Education Commission (15ZZ095).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dong, J., He, H., Zhang, D. et al. Boron improved electrochemical performance of LiNi0.8Co0.1Mn0.1O2 by enhancing the crystal growth with increased lattice ordering. J Mater Sci: Mater Electron 30, 18200–18210 (2019). https://doi.org/10.1007/s10854-019-02174-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-019-02174-3