Abstract
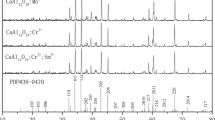
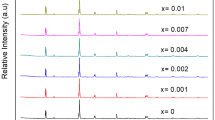
In this study, Bi3+ and Mn4+ co-doped CaAl12O19 phosphors were prepared via high-temperature solid-state reaction method, and their crystal structure, particle morphologies, luminescence properties, charge compensation mechanism and crystal field effect were investigated in detail. A most remarkable tuning effect can be achieved when the Bi3+ concentration is 5% in CaAl12O19:Bi3+, 0.05% Mn4+ and the electric multipolar interaction is inferred to be the main mechanism for concentration quenching effect. It is also found that, as charge compensators rather than sensitizers, Bi3+ ions can effectively tailor the emissions in a wide range through partial reducing Mn4+ to Mn2+. The charge compensation physics presumably involves the shift of Fermi energy level (EF) and variations of valence states of manganese ions after Bi3+ introduction. Moreover, the authors calculated the splitting energy (Δ) and Racah parameters (B and C) for Mn4+ in octahedral sites and Mn2+ in tetrahedral sites by combining excitation spectra with Tanabe-Sugano diagram. The experimental results will be of certain helpfulness in developing phosphors used in white LED.











Similar content being viewed by others
References
R.J. Xie, N. Hirosaki, Sci. Technol. Adv. Mater. 8, 588 (2007)
H.J. Guo, Y.H. Wang, G. Li, J. Liu, P. Feng, D.W. Liu, J. Mater. Chem. C 5, 12090 (2017)
R.J. Xie, H.T. Hintzen, J. Am. Ceram. Soc. 96, 665 (2013)
Y.J. Zhang, H. Zhang, P.P. Dong, Z.Y. Cheng, J.J. Yu, D.S. Li, G.Y. Cao, J. Mater. Sci.-Mater. 28, 86 (2017)
H. Guo, L.L. Sun, J. Liang, B. Li, X.Y. Huang, J. Lumin. 205, 115 (2019)
P.A. Tanner, Chem. Soc. Rev. 42, 5090 (2013)
Z.W. Gao, P.F. Sun, Y.F. Zhong, R.J. Yu, B. Deng, Opt. Laser Technol. 111, 163 (2019)
H.P. Ji, Z.H. Huang, Z.G. Xia, M.S. Molokeev, V.V. Atuchin, M.H. Fang, Y.A. Liu, J. Phys. Chem. C 119, 2038 (2015)
T. Sakthivel, G. Annadurai, R. Vijayakumar, X.Y. Huang, J. Lumin. 205, 129 (2019)
J.L. Zhong, L.X. Yu, X.Q. Man, W. Sun, X. Wu, X.F. Jiang, Z.Y. Gao, Q.H. Guo, Y.X. Zou, Luminescence 33, 1387 (2018)
M. Iwaki, S. Kumagai, S. Konishi, A. Koizumi, T. Hasegawa, K. Uematsu, A. Itadani, K. Toda, M. Sato, J. Alloy. Compd. 776, 1016 (2019)
Y. Chen et al., J. Lumin. 206, 11 (2019)
Q.S. Wu, J.C. Zhou, Dyes Pigments 161, 324 (2019)
X.G. Zhang, Z.P. Zhu, Z.Y. Guo, Z.S. Sun, Y.B. Chen, Chem. Eng. J. 356, 413 (2019)
Z.G. Xia, Y.Y. Zhang, M.S. Molokeev, V.V. Atuchin, Y. Luo, Sci. Rep-UK 3, 3310 (2013)
P.L. Shi, Z.G. Xia, M.S. Molokeev, V.V. Atuchin, Dalton Trans. 43, 9669 (2014)
V.V. Atuchin et al., J. Alloy. Compd. 785, 692 (2019)
M.M. Zhu, Y.X. Pan, M.M. Wu, H.Z. Lian, J. Lin, J. Alloy. Compd. 774, 331 (2019)
U.B. Humayoun, S.B. Kwon, S.K. Sami, D.H. Yoon, J. Alloy. Compd. 776, 594 (2019)
F. Tang, Z.C. Su, H.G. Ye, W.P. Gao, X.Q. Pan, S.J. Xu, Acs Omega 3, 13704 (2018)
J.S. Zhong, X. Chen, D.Q. Chen, M.J. Liu, Y.W. Zhu, X.Y. Li, Z.G. Ji, J. Alloy. Compd. 773, 413 (2019)
Y. Takeda, H. Kato, M. Kobayashi, H. Kobayashi, M. Kakihana, Chem. Lett. 44, 1541 (2015)
Q. Zhou, L. Dolgov, A.M. Srivastava, L. Zhou, Z.L. Wang, J.X. Shi, M.D. Dramicanin, M.G. Brik, M.M. Wu, J. Mater. Chem. C 6, 2652 (2018)
Y.J. Zheng, H.M. Zhang, H.R. Zhang, Z.G. Xia, Y.L. Liu, M.S. Molokeev, B.F. Lei, J. Mater. Chem. C 6, 4217 (2018)
J.R. Du, O.Q. De Clercq, D. Poelman, Ceram. Int. 44, 21613 (2018)
K. Omri, A. Alyamani, L. El Mir, Appl. Phys. A-Mater. 124, 215 (2018)
N. Alonizan, S. Rabaoui, K. Omri, R. Qindeel, Appl. Phys. A-Mater. 124, 710 (2018)
Y. Wakui, Y.J. Shan, K. Tezuka, H. Imoto, M. Ando, Mater. Res. Bull. 90, 51 (2017)
J. Lu, Y.X. Pan, J.G. Wang, X. Chen, S.M. Huang, G.K. Liu, Rsc Adv. 3, 4510 (2013)
L.N. Liem, N. Tran, IOP Conf. Ser.: Mater. Sci. Eng. 343, 012026 (2018)
Y.H. Jin, Y.H. Hu, H.Y. Wu, H. Duan, L. Chen, Y.R. Fu, G.F. Ju, Z.F. Mu, M. He, Chem. Eng. J. 288, 596 (2016)
D.X. Liu, C.G. Ma, P.W. Hu, Z. Li, Y. Tian, P. Su, M.G. Brik, A.M. Srivastava, S. Tanabe, J. Am. Ceram. Soc. 101, 2368 (2018)
Y. Zhao, L. Shi, Y.-J. Han, H.-H. Li, Z.-X. Ji, Z.-W. Zhang, Ceram. Int. 45, 8265 (2019)
Z. Zhou, N. Zhou, M. Xia, M. Yokoyama, H.T. Hintzen, J. Mater. Chem. C 4, 9143 (2016)
L. Wang, Z.X. Dai, R.L. Zhou, B.Y. Qu, X.C. Zeng, Phys. Chem. Chem. Phys. 20, 16992 (2018)
P.A. Rodnyi, P. Dorenbos, G.B. Stryganyuk, A.S. Voloshinovskii, A.S. Potapov, C.W.E. van Eijk, J. Phys.-Condens. Mater. 15, 719 (2003)
M.J. Reisfeld, N.A. Matwiyoff, L.B. Asprey, J. Mol. Spectrosc. 39, 8 (1971)
V. Dordevic, M.G. Brik, A.M. Srivastava, M. Medic, P. Vulic, E. Glais, B. Viana, M.D. Dramicanin, Opt. Mater. 74, 46 (2017)
R. Cao, Y. Ye, Q. Peng, T. Chen, H. Ao, F. Xiao, Z. Luo, P. Liu, J. Electron. Mater. 47, 7537 (2018)
Q. Zhou, L. Dolgov, A.M. Srivastava, L. Zhou, Z. Wang, J. Shi, M.D. Dramicanin, M.G. Brik, M. Wu, J. Mater. Chem. C 6, 2652 (2018)
F. Zhou, D. Aberg, Phys. Rev. B 93, 085123 (2016)
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No.51302059), the Natural Science Foundation of Anhui Province (Grant 1708085ME121) and the Undergraduate Innovative Training Project of Anhui Province (2018CXCYS033).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhao, C., Xu, Y., Wang, L. et al. A remarkably tunable emission from red to yellow to green in Mn4+-activated CaAl12O19 phosphor via co-doping Bi3+. J Mater Sci: Mater Electron 30, 11419–11428 (2019). https://doi.org/10.1007/s10854-019-01491-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-019-01491-x