Abstract
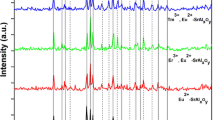
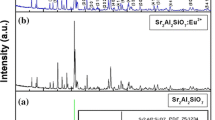
The phosphors of SrAl2O4: Eu2+, RE3+ (RE = Dy3+, Tb3+, Ho3+, La3+, Nd3+, Y3+, Tm3+, Er3+) and SrAl2O4: Eu2+, Dy3+, M2+ (M = Mn2+, Ni2+, Mg2+, Cu2+, Zn2+) were synthesized by co-precipitation combined hydrothermal method, and analyzed by X-ray diffraction (XRD), fluorescence spectrometer and SEM. XRD patterns illustrated that the synthesized products all were the mixed crystal phase of SrAl2O4 and Sr4Al14O25. The diffraction peaks shapes of phosphors SrAl2O4: Eu2+, RE3+ (RE = Tb3+, Ho3+, Y3+) were almost unchanged contrast to the marked changes of phosphors SrAl2O4: Eu2+, RE3+ (RE = Tm3+, Er3+, Nd3+, La3+), which showed co-activator species had a great impact on the crystal field around the emission center (Eu2+). Besides, the peak positions of SrAl2O4: Eu2+, Dy3+, M2+ (M = Mn2+, Ni2+, Mg2+, Cu2+, Zn2+) were unaltered after doping, while the peak intensity of crystal phase Sr4Al14O25 was enhanced after doped by cations (Mn2+, Ni2+, Cu2+, Zn2+), which indicated the doping of transition metal ions would cause the crystal phase transition. The fluorescence analysis results indicated that the luminescent intensity were reduced after being doped by co-activator ions (Dy3+, Tb3+, Ho3+, La3+, Nd3+, Y3+), but enhanced when doped by co-activator ions (Tm3+, Er3+), and the final emission wavelength and intensity sequence were as follows: \(\lambda\)Nd < \(\lambda\)La < \(\lambda\)Er < \(\lambda\)Y < \(\lambda\)Tm < \(\lambda\)Tb < \(\lambda\)Ho < \(\lambda\)Dy, INd < IDy < IY < ITb < IHo < ILa < ITm < IEr. Instead, the fluorescence emission intensity decreased with the increment of the doping concentration of transition metal ions, which perhaps because the transition metal ions doping under the optimal concentration of co-activator could reduce the luminescence intensity of products due to the concentration quenching. The crystal particles of SrAl2O4: Eu2+, Dy3+ presented regular hexagon flake-like morphology with a width of ~ 500 nm and a thickness of ~ 70 nm. The title products could be applied in the safety indicators, emergency lighting danger signals, automobiles, solar cells, etc.







Similar content being viewed by others
References
S. Bishnoi, B. Rajesh, G. Swati, V. Vikesh Jaiswal, M. Sahu, P. Singh, D. Haranath, Mater. Today Proc. 5, 610 (2018)
C. Xu, J. Nie, Z. Xu, X. Liu, J. Qiu, Mater. Lett. 225, 97 (2018)
A.S. Lysenkov, Y.F. Kargin, D.D. Titov, N.V. Petrakova, S.N. Ivicheva, A.I. Zakharov, N.A. Popova, V.V. Zakorzhevskii, I.P. Borovinskaya, I.S. Melnikova, IOP Conf. Ser. Mater. Sci. Eng. 347, 012040 (2018)
W.J. Lai, K.C. Cheng, Fibers Polym. 19, 22 (2018)
N.A.S. Rabaoui, K.O. Rabia, Appl. Phys. A 124, 1 (2018)
K.O.A. Alyamani, L. El Mir, Appl. Phys. A 124, 1 (2018)
T. Aitasalo, A. Durygin, J. Hölsä, M. Lastusaari, J. Niittykoski, A. Suchocki, J. Alloys Compd. 380, 4 (2004)
A. Salehabadi, M. Salavati-Niasari, F. Sarrami, A. Karton, Renew. Energy 114, 1419 (2017)
W. Xie, J. Quan, H. Wu, L. Shao, C. Zou, J. Zhang, X. Shi, Y. Wang, J. Alloys Compd. 514, 97 (2012)
C.C. Ã, W. Li, X. Huang, Z. Wang, X. Chen, X. Qian, R. Guo, Y. Ding, D. Mao, J. Lumin. 130, 347 (2010)
W. Jia, H. Yuan, L. Lu, H. Liu, W.M. Yen, J. Lumin. 76–77, 424 (1998)
X. Hu, H. Yang, T. Guo, D. Shu, W. Shan, G. Li, D. Guo, Ceram. Int. 44, 7535 (2018)
Z. Xue, S. Deng, Y. Liu, B. Lei, Y. Xiao, M. Zheng, J. Rare Earths 31, 241 (2013)
W. Shan, L. Wu, N. Tao, Y. Chen, D. Guo, Ceram. Int. 41, 15034 (2015)
P. Du, E.J. Kim, J.S. Yu, Curr. Appl. Phys. 18, 310 (2018)
R. Zhang, G. Han, L. Zhang, B. Yang, Mater. Chem. Phys. 113, 255 (2009)
P. Du, J.S. Yu, J. Alloys Compd. 653, 468 (2015)
F. Clabau, X. Rocquefelte, S. Jobic, P. Deniard, M.H. Whangbo, A. Garcia, T. Le Mercier, Chem. Mater. 17, 3904 (2005)
H. Du, W. Shan, L. Wang, D. Xu, H. Yin, Y. Chen, D. Guo, J. Lumin. 176, 272 (2016)
D.S. Kshatri, A. Khare, P. Jha, Opt. Int. J. Light Electron. Opt. 124, 2974 (2013)
Acknowledgements
The authors are grateful for the financial support of the National Natural Science Foundation of China (Nos. J1103312, J1210040 and 21341010), the Natural Science Foundation of Hunan Province (No. 11JJ5005), Hunan provincial Science & Technology Department (No. 2016SK2064), Changsha Science & Technology Bureau (Nos. kq1701164 and kq1701029), the Innovative Research Team in University (No. IRT1238), and China Outstanding Engineer Training Plan for Students of Chemical Engineering & Technology in Hunan University (MOE-No.2011-40). We also thank Dr. William Hickey, the U.S. professor of HRM, for the English editing on this paper.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hu, X., Wu, L., Yin, H. et al. Effects of co-activator species and transition metal ions doping on structure and fluorescence properties of strontium aluminate phosphors. J Mater Sci: Mater Electron 30, 3804–3810 (2019). https://doi.org/10.1007/s10854-019-00664-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-019-00664-y