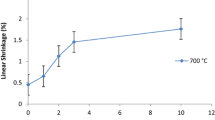
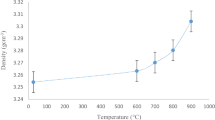
Abstract
This paper presents a comprehensive study on thermal, structural and optical properties of novel willemite glass–ceramics. The precursor glass in the ZnO–SLS glass system was successfully prepared using conventional melt-quenching technique and willemite (Zn2SiO4) glass–ceramics were derived from this precursor glass by a control crystallization process. The effect of heat-treatment temperature on the phase transformation, morphology and size of Zn2SiO4 crystal phase was examined using X-ray diffraction (XRD), field-emission scanning electron microscopy (FESEM) techniques. Furthermore, fourier transform infrared reflection (FTIR) spectroscopy was used to evaluate the Zn2SiO4 crystal structural evolution. The average size of Zn2SiO4 crystallite obtained from calculation of XRD is found to be in the range 30–60 nm, whereas the grain size observed in FESEM is in range of 200–400 nm. The appearance of SiO2, ZnO4 and Zn–O–Si bands detected from FTIR indicate the formation of Zn2SiO4 crystal phase. Besides, the study of the optical band gap has found that optical band gap of the glass–ceramics decreased as the heat treatment temperature increased. The photoluminescence spectra of willemite glass–ceramics exhibit two different emissions around 525 nm (green) and 585 nm (yellow); exhibit a characteristic of broad absorption band around 260 nm. These two different spectra reveal that the luminescence performance of the willemite glass–ceramics was enhanced with the progression of heat treatment temperature due to different located energy levels of the β-Zn2SiO4 and α-Zn2SiO4 crystalline phase. Such luminescent glass–ceramics was expected to find potential applications in phosphors and opto-electronic devices.









Similar content being viewed by others
References
E. Bernardo, L. Esposito, E. Rambaldi, A. Tucci, S. Hreglich, Recycle of waste glass into “glass–ceramic stoneware”. J. Am. Ceram. Soc. 91(7), 2156–2162 (2008)
M. Takesue, H. Hayashi, R.L. Smith, Thermal and chemical methods for producing zinc silicate (willemite): a review. Prog. Cryst. Growth Charact. Mater. 55(3), 98–124 (2009)
I.P. Sahu, D.P. Bisen, N. Brahme, R.K. Tamrakar, Studies on the luminescence behavior of SrCaMgSi2O7: Eu3+ phosphor by solid state reaction method. J. Mater. Sci.: Mater. Electron. 27(2), 1828–1839 (2016)
E. Yousef, M. Hotzel, C. Rüssel, Effect of ZnO and Bi2O3 addition on linear and non-linear optical properties of tellurite glasses. J. Non-Cryst. Solids 353(4), 333–338 (2007)
S. Rosmawati, H.A.A. Sidek, A.T. Zainal, H. Mohd Zobir, Effect of zinc on the physical properties of tellurite glass. J. Appl. Sci. 8, 1956–1961 (2008)
H.A. Abo-Mosallam, H. Darwish, S.M. Salman, Crystallization characteristic and properties of some zinc containing soda lime silicate glasses. J. Mater. Sci.: Mater. Electron. 21(9), 889–896 (2010)
A. Tarafder, A.R. Molla, C. Dey, B. Karmakar, Thermal, structural, and enhanced photoluminescence properties of Eu3+-doped transparent willemite glass–ceramic nanocomposites. J. Am. Ceram. Soc. 96(8), 2424–2431 (2013)
Y. Yu, Y. Wang, D. Chen, P. Huang, E. Ma, F. Bao, Enhanced emissions of Eu3+ by energy transfer from ZnO quantum dots embedded in SiO2 glass. Nanotechnology 19(5), 1–5 (2008)
L. Hou, G. Zuo, Y. Shen, Y. Meng, H. Li, Effects of the replacing content of ZnBr2 on the properties of ZnO–B2O3–SiO2:Mn2+ glass–ceramics. Ceram. Int. 40(8), 13097–13103 (2014)
A.R. Boccaccini, M. Bücker, J. Bossert, K. Marszalek, Glass matrix composites from coal fly ash and waste glass. Waste Manag 17(1), 39–45 (1997)
L. Barbieri, A.C. Bonamartini, I. Lancellotti, Alkaline and alkaline-earth silicate glasses and glass–ceramics from municipal and industrial wastes. J. Eur. Ceram. Soc. 20(14), 2477–2483 (2000)
R.D. Rawlings, J.P. Wu, A.R. Boccaccini, Glass–ceramics: their production from wastes-a review. J. Mater. Sci. 41(3), 733–761 (2006)
S.M. Salman, S.N. Salama, E.A. Mahdy, The effect of strontium oxide replacing calcium oxide on the crystallization and thermal expansion properties of Li2O–CaO–SiO2 glasses. Ceram. Int. 41(1), 137–143 (2015)
J. Sheng, K. Kadono, Y. Utagawa, T. Yazawa, X-ray irradiation on the soda-lime container glass. Appl. Radiat. Isot. 56(4), 621–626 (2002)
E.D. Zanotto, Surface crystallization kinetics in soda-lime-silica glasses. J. Non-Cryst. Solids 129(1), 183–190 (1991)
S. Petrescu, M. Constantinescu, E.M. Anghel, I. Atkinson, M. Olteanu, M. Zaharescu, Structural and physico-chemical characterization of some soda lime zinc alumino-silicate glasses. J. Non-Cryst. Solids 358(23), 3280–3288 (2012)
N. Marinoni, D. D’Alessio, V. Diella, A. Pavese, F. Francescon, Effects of soda-lime-silica waste glass on mullite formation kinetics and micro-structures development in vitreous ceramics. J. Environ. Manag. 124, 100–107 (2013)
B.E. Yekta, P. Alizadeh, L. Rezazadeh, Synthesis of glass–ceramic glazes in the ZnO–Al2O3–SiO2–ZrO2 system. J. Eur. Ceram. Soc. 27(5), 2311–2315 (2007)
A. Tarafder, A.R. Molla, S. Mukhopadhyay, B. Karmakar, Fabrication and enhanced photoluminescence properties of Sm3+-doped ZnO–Al2O3–B2O3–SiO2 glass derived willemite glass–ceramic nanocomposites. Opt. Mater. 36(9), 1463–1470 (2014)
G.Q. Xu, H.T. Xu, Z.X. Zheng, Y.C. Wu, Preparation and characterization of Zn2SiO4:Mn phosphors with hydrothermal methods. J. Lumin. 130(10), 1717–1720 (2010)
M. Takesue, H. Hayashi, R.L. Smith, Thermal and chemical methods for producing zinc silicate (willemite): a review. Prog. Cryst. Growth Charact. Mater. 55(3), 98–124 (2009)
V. Sivakumar, A. Lakshmanan, S. Kalpana, R.S. Rani, R.S. Kumar, M.T. Jose, Low-temperature synthesis of Zn2SiO4:Mn green photoluminescence phosphor. J. Lumin. 132(8), 1917–1920 (2012)
K. Omri, J. El Ghoul, A. Alyamani, C. Barthou, L. El Mir, Luminescence properties of green emission of SiO2/Zn2SiO4:Mn nanocomposite prepared by sol–gel method. Phys. E 53, 48–54 (2013)
Y.C. Kang, I.W. Lenggoro, S.B. Park, K. Okuyama, Photoluminescence characteristics of YAG: Tb phosphor particles with spherical morphology and non-aggregation. J. Phys. Chem. Solids 60(11), 1855–1858 (1999)
R. Stefan, E. Culea, P. Pascuta, The effect of copper ions addition on structural and optical properties of zinc borate glasses. J. Non-Cryst. Solids 358(4), 839–846 (2012)
E.M.A. Khalil, F.H. El Batal, Y.M. Hamdy, H.M. Zidan, M.S. Aziz, A.M. Abdelghany, Infrared absorption spectra of transition metals-doped soda lime silica glasses. Phys. B 405(5), 1294–1300 (2010)
J. Williamson, F.P. Glasser, Crystallisation of zinc silicate liquids and glasses. Phys. Chem. Glasses 5(1), 52–59 (1964)
H. Yang, R.M. Hazen, R.T. Downs, L.W. Finger, Structural change associated with the incommensurate-normal phase transition in akermanite, Ca2MgSi2O7, at high pressure. Phys. Chem. Miner. 24(7), 510–519 (1997)
A.R. Molla, A. Tarafder, B. Karmakar, Synthesis and properties of glasses in the K2O–SiO2–Bi2O3–TiO2 system and bismuth titanate (Bi4Ti3O12) nano glass–ceramics thereof. J. Mater. Sci. 46(9), 2967–2976 (2011)
R. Casasola, J.M. Rincón, M. Romero, Glass–ceramic glazes for ceramic tiles: a review. J. Mater. Sci. 47(2), 553–582 (2012)
N.F. Syamimi, K.A. Matori, W.F. Lim, H.A.A. Sidek, M.H.M. Zaid, Effect of sintering temperature on structural and morphological properties of europium (III) oxide doped willemite. J. Spectrosc. 2014(1), 1–7 (2014)
G.V. Sarrigani, K.A. Matori, W.F. Lim, A. Kharazmi, H.J. Quah, H.R. Bahari, M. Hashim, Structural and optical properties of erbium-doped willemite-based glass–ceramics. Appl. Opt. 54(33), 9925–9929 (2015)
N.A.S. Omar, Y.W. Fen, K.A. Matori, M.H.M. Zaid, M.R. Norhafizah, M. Nurzilla, M.I.M. Zamratul, Synthesis and optical properties of europium doped zinc silicate prepared using low cost solid state reaction method. J. Mater. Sci.: Mater. Electron. 27(2), 1092–1099 (2015)
L. Barbieri, C. Leonelli, T. Manfredini, C. Siligardi, A.B. Corradi, P. Mustarelli, C. Tomasi, Nucleation and crystallization of a lithium aluminosilicate glass. J. Am. Ceram. Soc. 80(12), 3077–3083 (1997)
C.C. Lin, P. Shen, Sol-gel synthesis of zinc orthosilicate. J. Non-Cryst. Solids 171(3), 281–289 (1994)
P.T. Chao, P. Shen, C.C. Lin, Thermal cycle etching of willemite (0001): effects of surface premelting, dislocation outcrops and polygonization. Mater. Sci. Eng. A 335(1), 191–197 (2002)
A. Tarafder, A.R. Molla, C. Dey, B. Karmakar, Thermal, structural, and enhanced photoluminescence properties of Eu3+-doped transparent willemite glass–ceramic nanocomposites. J. Am. Ceram. Soc. 96(8), 2424–2431 (2013)
Z.Y. Li, Z.Q. Zhang, Fragility of photonic band gaps in inverse-opal photonic crystals. Phys. Rev. B 62(3), 1516–1519 (2000)
L.S. El-Deen, M.S. Al Salhi, M.M. Elkholy, IR and UV spectral studies for rare earths-doped tellurite glasses. J. Alloys Compd. 465(1), 333–339 (2008)
P.G. Pavani, K. Sadhana, V.C. Mouli, Optical, physical and structural studies of boro-zinc tellurite glasses. Phys. B 406(6), 1242–1247 (2011)
F. El-Diasty, F.A.A. Wahab, M. Abdel-Baki, Optical band gap studies on lithium aluminum silicate glasses doped with Cr3+ ions. J. Appl. Phys. 100(9), 1–7 (2006)
V. Madhuri, J.S. Kumar, M.S. Rao, S. Cole, Investigations on spectral features of tungsten ions in sodium lead alumino borate glass system. J. Phys. Chem. Solids 78, 70–77 (2015)
S. Hasegawa, M. Kitagawa, Effects of annealing on localized states in amorphous Ge films. Solid State Commun. 27(9), 855–858 (1978)
R.S. Chakradhar, B.M. Nagabhushana, G.T. Chandrappa, K.P. Ramesh, J.L. Rao, Solution combustion derived nanocrystalline Zn2SiO4:Mn phosphors: a spectroscopic view. J. Chem. Phys. 121(20), 10250–10259 (2004)
Acknowledgments
The researchers gratefully acknowledge the financial support for this study from the Malaysian Ministry of Higher Education (MOHE) and Universiti Putra Malaysia through the Fundamental Research Grant Scheme (FRGS) and Inisiatif Putra Berkumpulan (IPB) research grant.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zaid, M.H.M., Matori, K.A., Abdul Aziz, S.H. et al. Synthesis and characterization of low cost willemite based glass–ceramic for opto-electronic applications. J Mater Sci: Mater Electron 27, 11158–11167 (2016). https://doi.org/10.1007/s10854-016-5234-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-016-5234-6