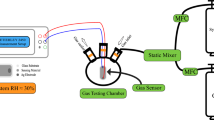
Abstract
Amperometric mode of electrochemical gas sensor requires several systematic approaches to optimize compositions of electrodes and electrolyte materials when both the electrodes are exposed to same gas environment. Using Gd-doped ceria electrolyte, Ni2+ doped CuO active electrode and fixed inactive electrode La0.5Sr0.5CoO3 electrochemical sensors were investigated for 3–40 ppm ammonia in base gas (5%O2 + N2). Highest sensitivity of 225 μA/decade at 550 °C was obtained for sensor with 15 mol% of Gd-doped ceria (GDC 15) and 2 mol% of Ni2+ doped CuO as electrolyte and active electrode, respectively, when studied in the range 300–650 °C. Among all the electrolyte compositions, GDC 15 showed highest electrical conductivity (0.84 × 10–2 S/cm at 550 °C), and thus, highest sensitivity was correlated at a particular temperature. The vacancy trap mechanism was found to influence electrical conductivity of electrolyte. The same sensor configuration showed highest potentiometric sensitivity of 116 mV/decade at 550 °C. High sensitivity to ppm level NH3, selectivity, short-term and long-term stability are some of the promising results of the developed sensor for exhaust gas applications.
Graphical abstract















Similar content being viewed by others
References
Miura N, Sato T, Anggraini SA et al (2014) A review of mixed-potential type zirconia-based gas sensors. Ionics (Kiel) 20:901–925. https://doi.org/10.1007/s11581-014-1140-1
Chen XJ, Khor KA, Chan SH, Yu LG (2002) Influence of microstructure on the ionic conductivity of yttria-stabilized zirconia electrolyte. Mater Sci Eng A 335:246–252. https://doi.org/10.1016/S0921-5093(01)01935-9
Accardo G, Ferone C, Cioffi R et al (2016) ITSOFCs by sol-gel route. J Appl Biomater Funct Mater 14:35–41. https://doi.org/10.5301/jabfm.5000265
Liu Y, Parisi J, Sun X, Lei Y (2014) Solid-state gas sensors for high temperature applications-a review. J Mater Chem A 2:9919–9943. https://doi.org/10.1039/c3ta15008a
Pasierb P, Rekas M (2009) Solid-state potentiometric gas sensors-current status and future trends. J Solid State Electrochem 13:3–25. https://doi.org/10.1007/s10008-008-0556-9
Xiong L, Compton RG (2014) Amperometric gas detection: a review. Int J Electrochem Sci 9:7152–7181
Stetter JR, Li J (2008) Amperometric gas sensors-a review. Chem Rev 108:352–366. https://doi.org/10.1021/cr0681039
Zha S, Moore A, Abernathy H, Liu M (2004) GDC-based low-temperature SOFCs powered by hydrocarbon fuels. J Electrochem Soc 151:A1128. https://doi.org/10.1149/1.1764566
Arabac A, Öksüzömer MF (2012) Preparation and characterization of 10 mol% Gd doped CeO 2 (GDC) electrolyte for SOFC applications. Ceram Int 38:6509–6515. https://doi.org/10.1016/j.ceramint.2012.05.030
Kuppusami P, Muthukkumaran K, Divakar R et al (2007) Nano-ionic thin films of gadolinia-doped ceria prepared by pulsed laser ablation. Ionics (Kiel) 13:343–348. https://doi.org/10.1007/s11581-007-0122-y
Nithya S, Sharan R, Roy M et al (2019) Ni doping in CuO: a highly sensitive electrode for sensing ammonia in ppm level using lanthanum gallate based electrolyte. Mater Res Bull 118:110478. https://doi.org/10.1016/j.materresbull.2019.05.003
Olofsson G, Wallenberg LR, Andersson A (2005) Selective catalytic oxidation of ammonia to nitrogen at low temperature on Pt/CuO/Al2O3. J Catal 230:1–13. https://doi.org/10.1016/j.jcat.2004.11.023
Cui X, Zhou J, Ye Z et al (2010) Selective catalytic oxidation of ammonia to nitrogen over mesoporous CuO/RuO2 synthesized by co-nanocasting-replication method. J Catal 270:310–317. https://doi.org/10.1016/j.jcat.2010.01.005
Hung C (2006) Selective catalytic oxidation of ammonia to nitrogen on CuO-CeO2 bimetallic oxide catalysts. 6:150–169
Su YM, Kung FC, Su TL et al (2011) Material characteristics and electric properties of SiOx-doped GDC electrolytes. J Chin Inst Eng Trans Chin Inst Eng A 34:31–38. https://doi.org/10.1080/02533839.2011.555465
Dutta A, Patra S, Bedekar V et al (2009) Nanocrystalline gadolinium doped ceria: combustion synthesis and electrical characterization. J Nanosci nanotechnol. https://doi.org/10.1166/jnn.2009.dk26
Shim JO, Jeong DW, Jang WJ et al (2014) Deoxygenation of oleic acid over Ce(1–x)Zr(x)O2 catalysts in hydrogen environment. Renew Energy 65:36–40. https://doi.org/10.1016/j.renene.2013.07.008
Kosaka T, Sato K (2010) Free volumes associated with sintering in gadolinium doped ceria solid solutions. J Nanomater. https://doi.org/10.1155/2010/935146
Acharya SA, Gaikwad VM, D’Souza SW, Barman SR (2014) Gd/Sm dopant-modified oxidation state and defect generation in nano-ceria. Solid State Ion 260:21–29. https://doi.org/10.1016/j.ssi.2014.03.008
Tian C, Chan SW (1996) Ionic conductivities of doped CeO2 thin films as related to their microstructure. Mater Res Soc Symp-Proc 411:277–282. https://doi.org/10.1557/PROC-411-277
Tian C, Chan S-W (1996) Grain boundary conductivity and microstructure study of 4% Y2O3 doped CeO2 thin films. MRS Online Proc Library Arch. https://doi.org/10.1557/PROC-453-555
Tian C, Chan S-W (2002) Electrical conductivities of (CeO2)1–x(Y2O3)x thin films. J Am Ceram Soc 85(9):2222–2229. https://doi.org/10.1111/j.1151-2916.2002.tb00439.x
Kudo T, Obayashi H (1976) Mixed electrical conduction in the fluorite-type Cel−xGdxO2–x/2. J Electrochem Soc 123(3):415. https://doi.org/10.1149/1.2132840
Tianshu Z, Hing P, Huang H, Kilner J (2002) Ionic conductivity in the CeO2-Gd2O3 system (0.05 ≤ Gd/Ce ≤ 0.4) prepared by oxalate coprecipitation. Solid State Ion 148:567–573. https://doi.org/10.1016/S0167-2738(02)00121-2
Zhou XD, Huebner W, Kosacki I, Anderson HU (2002) Microstructure and grain-boundary effect on electrical properties of gadolinium-doped ceria. J Am Ceram Soc 85:1757–1762. https://doi.org/10.1111/j.1151-2916.2002.tb00349.x
Zhan Z, Wen T-L, Tu H, Lu Z-Y (2001) AC impedance investigation of samarium-doped ceria. J Electrochem Soc 148:A427. https://doi.org/10.1149/1.1359198
Sharan R, Roy M, Dutta A (2020) Sensing behaviour investigation of amperometric electrochemical NH 3 sensor: optimization of lanthanum gallate based electrolyte composition. J Electrochem Soc 167:117508. https://doi.org/10.1149/1945-7111/aba641
Elumalai P, Plashnitsa V, Fujio Y, Miura N (2019) High temperature mixed-potential-type ammonia sensor using stabilized zirconia and oxide-based sensing electrode. ECS Trans 16:247–255. https://doi.org/10.1149/1.2981126
Li X, Wang C, Wang B et al (2016) Effects of sintering temperature on the NH3 sensing properties of Mg2Cu0.25Fe1O3.75 electrode for YSZ-based potentiometric NH3 sensor. Ceram Int 42:2214–2220. https://doi.org/10.1016/j.ceramint.2015.10.013
Yang B, Wang C, Xiao R et al (2019) High NH3 selectivity of NiFe2O4 sensing electrode for potentiometric sensor at elevated temperature. Anal Chim Acta 1089:165–173. https://doi.org/10.1016/j.aca.2019.09.006
Wang C, Xu J, Yang B et al (2018) Effect of MgO doping on the BiVO4 sensing electrode performance for YSZ-based potentiometric ammonia sensor. Solid State Electron 147:19–25. https://doi.org/10.1016/j.sse.2018.05.002
Zhang J, Zhang C, Xia J, et al (2017) Mixed-potential NH3 sensor based on Ce0.8Gd0.2O1.9 solid electrolyte. Sens Actuators B Chem https://doi.org/10.1016/j.snb.2017.04.035
Moos R, Müller R, Plog C et al (2002) Selective ammonia exhaust gas sensor for automotive applications. Sens Actuators, B Chem 83:181–189. https://doi.org/10.1016/S0925-4005(01)01038-3
Tsui L, Benavidez A, Palanisamy P et al (2018) Electrochimica Acta Automatic signal decoding and sensor stability of a 3-electrode mixed- potential sensor for NO x/NH 3 quanti fi cation. Electrochim Acta 283:141–148. https://doi.org/10.1016/j.electacta.2018.06.133
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors' declares that they have no conflict of interest.
Additional information
Handling Editor: Christopher Blanford.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nithya, S., Dutta, A. Electrochemical sensing of trace level NH3: active electrode and electrolyte optimizations. J Mater Sci 56, 6269–6285 (2021). https://doi.org/10.1007/s10853-020-05654-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-020-05654-8