Abstract
Metal particle agglomeration is known to be one of the factors responsible for the reduction in catalytic activity (even selectivity) when metal particles are supported on carbon. In this study, an approach using functional groups attached to a polymeric support was used to reduce metal agglomeration. Herein, we report on the preparation of polystyrene (PS) (d = 350 nm) and asymmetric polystyrene-b-poly(acrylic acid) (PS-b-PAA) nanospheres (8% and 12% PAA loadings; d = 274 nm and 343 nm) to make cobalt/polymer complexes. The CoxOy nanoparticles were supported on the PS and PS-b-PAA templates (5%, 10% and 15% Co loading), and then the CoxOy/template was coated with resorcinol–formaldehyde (RF). Thermal removal of the templates and annealing of the RF at 600 °C for 2 h gave the CoxOy@HCS materials. At a nominal Co loading of 5%, the PS template produced CoxOy@HCS materials that contained small Co particles (6.1 nm) while at higher Co loadings (10%, 15%) large and irregularly shaped Co nanoparticles (d > 20 nm) were formed. In contrast, PS-b-PAA gave CoxOy@HCS with small, highly dispersed Co nanoparticles (d < 10 nm) at loadings between 5 and 15%. The three polymeric nanospheres and the CoxOy/polymer and CoxOy@HCS materials were all characterized using SEM, TEM, TGA and FTIR spectroscopy. The use of functionalized polymer templates thus allows small Co particles to be easily transferred from the template to the RF to give CoxOy@HCS, even when using high-temperature (600 °C) synthesis conditions.
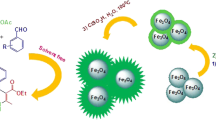
Graphic abstract












Similar content being viewed by others
References
Egeblad K, Rass-Hansen J, Marsden CC, Taarning E, Christensen CH (2009) Heterogeneous catalysis for production of value-added chemicals from biomass. In: Spivey JJ, Dooley KM (eds) Catalysis, vol 21. Royal Society of Chemistry, London, pp 13–50. https://doi.org/10.1039/B712664F
Kalz KF, Kraehnert R, Dvoyashkin M, Dittmeyer R, Gläser R, Krewer U, Reuter K, Grunwaldt J-D (2017) Future challenges in heterogeneous catalysis: understanding catalysts under dynamic reaction conditions. ChemCatChem 9:17–29. https://doi.org/10.1002/cctc.201600996
Peres L, Axet MR, Yi D, Serp P, Soulantica K (2019) Selective hydrogenation of cinnamaldehyde by unsupported and few layer graphene supported platinum concave nanocubes exposing 110 facets stabilized by long-chain amine. Catal Today. https://doi.org/10.1016/j.cattod.2019.05.048
Garces LJ, Hincapie B, Zerger R, Suib SL (2015) The effect of temperature and support on the reduction of cobalt oxide: an in-situ X-ray diffraction study. J Phys Chem C 119(2015):5484–5490. https://doi.org/10.1021/jp5124184
Iglesia E, Soled SL, Fiato RA (1992) Fischer–Tropsch synthesis on cobalt and ruthenium metal dispersion and support effects on reaction rate and selectivity. J Catal 137:212–224. https://doi.org/10.1016/0021-9517(92)90150-G
Lualdi M (2012) Fischer–Tropsch synthesis over cobalt based catalysts for BTL applications. PhD thesis, Department of Chemical Engineering, Stockholm, Sweden
Pomogailo AD, Dzhardimalieva GI (2014) Reduction of metal ions in polymer matrices as a condensation method of nanocomposite synthesis. In: Nanostructure materials preparation via condensation ways. Springer, Dordrecht. https://doi.org/10.1007/978-90-481-2567-8_2
Phaahlamohlaka TN, Kumi DO, Dlamini MW, Jewell LL, Coville NJ (2016) Ruthenium nanoparticles encapsulated inside porous hollow carbon spheres: a novel catalyst for Fischer-Tropsch synthesis. Catal Today 275:76–83. https://doi.org/10.1016/j.cattod.2015.11.034
Dong C, Yu Q, Ye R-P, Su P, Liu J, Wang G-H (2020) Hollow carbon spheres nanoreactors loaded with PdCu nanoparticles: void-confinement effects in liquid-phase hydrogenations. Angew Chem Int Ed. https://doi.org/10.1002/anie.202007297
Nongwe I, Ravat V, Meijboom R, Coville NJ (2016) Pt supported nitrogen doped hollow carbon spheres for the catalysed reduction of cinnamaldehyde. Appl Catal A Gen 517:30–38. https://doi.org/10.1016/j.apcata.2016.02.025
Bin D-S, Chi Z-X, Li Y, Zhang K, Yang X, Sun Y-G, Piao J-Y, Cao A-M, Wan L-J (2017) Controlling the compositional chemistry in single nanoparticles for functional hollow carbon nanospheres. J Am Chem Soc 139:13492–13498. https://doi.org/10.1021/jacs.7b07027
Xu F, Tang Z, Huang S, Chen L, Liang Y, Mai W, Zhong H, Fu R, Wu D (2015) Facile synthesis of ultrahigh-surface-area hollow carbon nanospheres for enhanced adsorption and energy storage. Nat Commun. https://doi.org/10.1038/ncomms8221
Xu F, Ding B, Qiu Y, Dong R, Zhuang W, Xu X, Han H, Yang J, Wei B, Wang H, Kaskel S (2020) Generalized domino-driven synthesis of hollow hybrid carbon spheres with ultrafine metal nitrides/oxides. Matter 3:246–260. https://doi.org/10.1016/j.matt.2020.05.012
Yang L-P, Lin X-J, Zhang X, Zhang W, Cao A-M, Wan L-J (2016) A general synthetic strategy for hollow hybrid microspheres through a progressive-inward-crystallization process. J Am Chem Soc 138:5916–5922. https://doi.org/10.1021/jacs.6b00773
Xu F, Lu Y, Ma J, Huang Z, Su Q, Fu R, Wu D (2017) Facile, general and template-free construction of monodisperse yolk-shell metal@carbon nanospheres. Chem Commun 53:12136–12139. https://doi.org/10.1039/C7CC06502G
Gangatharan PM, Maubane-Nkadimeng MS, Coville NJ (2019) Building carbon structures inside hollow carbon spheres. Sci Rep. https://doi.org/10.1035/s41598-019-46992-1
Phaahlamohlaka TN, Kumi DO, Dlamini MW, Forbes R, Jewell LL, Billing DG, Coville NJ (2017) Effects of Co and Ru intimacy in Fischer–Tropsch catalysis using hollow carbon sphere supports: assesment of the hydrogen spillover processes. ACS Catal 7:1568–1578. https://doi.org/10.1021/acscatal.6b03102
Forster S (2003) Amphilic block copolymers for templating applications. In: Antonietti M (ed) Colloid chemistry I, topics in current chemistry, vol 226. Springer, Berlin, pp 1–28. https://doi.org/10.1007/3-540-36408-0_1
Alexandridis P, Tsianou M (2011) Block copolymer-directed metal nanoparticle morphogenesis and organization. Eur Polym J 47:569–583. https://doi.org/10.1016/j.eurpolymj.2010.10.021
Forster S, Konrad M (2003) From self-organizing polymers to nano- and biomaterials. J Mater Chem 13:2671–2688. https://doi.org/10.1039/B307512P
Yu C-L, Bian F, Zhang S-F, Xu X, Ren P, Wang F-C, Zhang F-A (2018) Preparation of the monodispersed carboxyl-functionalized polymer microspheres with disproportionated rosin moety and adsorption of methylene blue. Adsorp Sci Technol 36:1260–1273. https://doi.org/10.1177/0263617418766774
Cho SM, Song G, Park C, Lee Y, Kang HS, Lee W, Park S, Huh J, Ryu DY, Park C (2018) Surface functionalized nanostructures via position registered supramolecular polymer assembly. Nanoscale 10:6333–6342. https://doi.org/10.1039/C7NR07852H
Martin HJ, White BT, Scanlon CJ, Saito T, Dadmun MD (2017) Tunable synthetic control of soft polymeric nanoparticle morphology. Soft Matter 13:8849–8857. https://doi.org/10.1039/C7SM01533J
Wang Y, Biradar AV, Wang G, Sharma KK, Duncan CT, Rangan S, Asefa T (2010) Controlled synthesis of water-dispersible faceted crystalline copper nanoparticles and their catalytic properties. Chem Eur J 16:10735–10743. https://doi.org/10.1002/chem.201000354
Klingelhofer S, Heitz W, Greiner A, Oestreich S, Forster S, Antonietti M (1997) Preparation of palladium colloids in block copolymer micelles and their use for the catalysis of the heck reaction. J Am Chem Soc 119:10116–10120. https://doi.org/10.1021/ja9714604
Zhang L, Eisenberg A (1995) Multiple morphologies of "crew-cut" polystyre-b-poly(acrylic acid) block copolymers. Science 268:1728–1731. https://doi.org/10.1126/science.268.5218.1728
Fu J, Xu Q, Chen J, Chen Z, Huang X, Tang X (2010) Controlled fabrication of uniform hollow core porous shell carbon spheres by the pyrolysis of core/shell polystyrene/crosslinked polyphosphazene composites. Chem Commun 46:6563–6565. https://doi.org/10.1039/C0CC01185A
Liu J, Qiao SZ, Lui H, Chen J, Orpe A, Zhao D, Lu GQ (2011) Extension of the stober method to the preparation of monodisperse resorcinol-formaldehyde resin polymer and carbon spheres. Angew Chem Int Ed 50:5947–5951. https://doi.org/10.1002/anie.201102011
Luo L, Eisenberg A (2001) Thermodynamic size control of block copolymer vesicles in solution. Langmuir 17:6804–6811. https://doi.org/10.1021/la0104370
Ayres N, Boyes SG, Brittain WJ (2007) Stimuli-responsive polyelectrolyte polymer brushes prepared via atom-transfer radical polymerization. Langmuir 23:182–189. https://doi.org/10.1021/la061526l
Arslan M, Yilmaz G, Yagci Y (2014) Synthesis of polystyrene-b-poly(ethylene glycol) block copolymers by radical exchange reactions of RAFT agents. Des Monomers Polym 17:238–244. https://doi.org/10.1080/15685551.2013.840478
Abood Mabdal-H (2013) Synthesis and characterization of grafted polystyrene with acrylic acid using gamma-irradiation. Aust J Basic Appl Sci 7:746–750
Al-Muhtaseb SA, Ritter JA (2003) Preparation and properties of resorcinol formaldehyde organic and carbon gels. Adv Mater 15:101–114. https://doi.org/10.1002/adma.200390020
Cai W, Gu M, Jin W, Zhou J (2019) CTAB-functionalized C@SiO2 double-shelled hollow microspheres with enhanced and selective adsorption performance for Cr(VI). J Alloys Compd 777:1304–1312. https://doi.org/10.1016/j.jallcom.2018.11.070
Lui X, Li S, Mei J, Lau WM, Mi R, Li Y, Liu H, Liu L (2014) From melamine-resorcinol-formaldehyde to nitrogen doped carbon xerogels with micro and meso-pores for lithium batteries. J Mater Chem A 2:14429–14438. https://doi.org/10.1039/C4TA02928C
Mongwe TH, Matsoso BJ, Mutuma BK, Coville NJ, Maubane MS (2018) Synthesis of chain-like carbon nano-onions by a flame assisted pyrolysis technique using different collecting plates. Diam Relat Mater 90:135–143. https://doi.org/10.1016/j.diamond.2018.10.002
Tang C-W, Wang C-B, Chien S-H (2008) Characterization of cobalt oxides studied by FT-IR, Raman, TPR and TG-MS. Thermochim Acta 473:68–73. https://doi.org/10.1016/j.tca.2008.04.015
Koo J, Kim J, Lee H, Chung H, Lee Y, Yi W, Sohn D (2012) Formation and characterization of poly(acrylic acid) on silica particles irradiated by γ-ray radiation. Macromol Res 20:138–142. https://doi.org/10.1007/s13233-012-0078-2
Todica M, Stefan R, Pop CV, Olar L (2015) IR and Raman investigation of some poly(acrylic) acid gels in aqueous and neutralized state. Acta Phys Pol A 128:128–135. https://doi.org/10.12693/APhysPolA.128.128
Murli C, Song Y (2010) Pressure-induced polymerization of acrylic acid: a Raman spectroscopic study. J Phys Chem B 114:9744–9750. https://doi.org/10.1021/jp1034757
Phaahlamohlaka TN, Dlamini MW, Kumi DO, Forbes R, Jewell LL, Coville NJ (2020) Co inside hollow carbon spheres as a Fischer–Tropsch catalyst: Spillover effects from Ru placed inside and outside the HCSs. Appl Catal A Gen. https://doi.org/10.1016/j.apcata.2020.117617
Molefe T, Forbes RP, Coville NJ (2020) Osmium@Hollow carbon spheres as Fischer–Tropsch synthesis catalysts. Catal Lett. https://doi.org/10.1007/s10562-020-03347-0
Mosiane LR, Matsoso BJ, Makhongoana A, Mutuma BK, Mongwe TH, Coville NJ, Maubane-Nkadimeng MS (2019) Tuning the properties of CVD-grown multiwalled carbon nanotubes by ex situ codoping with boron and nitrogen heteroatoms. J Nanopart Res. https://doi.org/10.1007/s11051-019-4654-7
Boskovic BO, Golovko VB, Cantoro M, Kleinsorge BY, Chuang ATH, Ducati C, Hofmann S, Robertson J, Johnson BFG (2005) Low temperature synthesis of carbon nanofibres on carbon fibre matrices. Carbon 43:2643–2648. https://doi.org/10.1016/j.carbon.2005.04.034
Fu R, Baumann TF, Cronin S, Dresselhaus G, Dresselhaus MS, Satcher JHJ (2005) Formation of graphitic structures in cobalt-nickel doped carbon aerogels. Langmuir 21:2647–2651. https://doi.org/10.1021/la047344d
Chandra S, Bag S, Bhar R, Pramanik P (2011) Effect of transition and non-transition metals during the synthesis of carbon xerogels. Microporous Mesoporous Mater 138:149–156. https://doi.org/10.1016/j.micromeso.2010.09.012
Maldonado-Hodar FJ, Ferro-Garcia MA, Rivera-Utrilla J, Moreno-Castilla C (1999) Synthesis and textural properties of organic aerogels, transition-metal-cotaining organic aerogels and their carbonized derivatives. Carbon 37:1199–1205. https://doi.org/10.1016/S0008-6223(98)00314-5
Liu Z, Li J, Yang Y, Mi JH, Tan XL (2012) Synthesis, characterization and magnetic examination of Fe, Co and Ni doped carbon xerogels. Mat Res Inov 16:362–367. https://doi.org/10.1179/143307512X13463991318033
Zhu D, Huang Y, Cao J, Lee CS, Chen M, Shen Z (2019) Cobalt nanoparticles encapsulated in porous nitrogen-doped carbon: oxygen activation and efficient catalytic removal of formaldehyde at room temperature. Appl Catal B Environ. https://doi.org/10.1016/j.apcatb.2019.117981
Alex AS, Lekshmi AM, Sekkar V, John B, Gouri C, Ilangovan S (2017) Microporous carbon aerogel prepared through ambient pressure drying routes as anode material for lithium ion cells. Polym Adv Technol 28:1945–1950. https://doi.org/10.1002/pat.4085
Mos YM, Vermeulem AC, Buisman CJN, Weijma J (2018) X-Ray Diffraction of iron containing samples: the importance of a suitable configuration. Geomicrobiol J 35:511–517. https://doi.org/10.1080/01490451.2017.1401183
Dlamini MW, Phaahlamohlaka TN, Kumi DO, Forbes R, Jewell LL, Coville NJ (2020) Post doped nitrogen-decorated hollow carbon spheres as a support for Co Fischer–Tropsch catalysts. Catal Today 342:99–110. https://doi.org/10.1016/j.cattod.2019.01.070
Dlamini MW, Kumi DO, Phaahlamohlaka TN, Lyadov AS, Billing DG, Jewell LL, Coville NJ (2015) Carbon spheres prepared by hydrothermal synthesis—a support for bimetallic iron coblat Fischer–Tropsch catalysts. ChemCatChem 7:3000–3011. https://doi.org/10.1002/cctc.201500334
Acknowledgements
This work was supported by the National Research Foundation (NRF), South Africa, the University of the Witwatersrand (Postgraduate Merit Award), HYSA (Hydrogen South Africa) and the DSI-NRF Centre of Excellence in Strong Materials (CoESM). Sincere gratitude goes to Ms Petra Dinham (School of Chemical and Metallurgical Engineering, University of the Witwatersrand) for the SEM images.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Naiqin Zhao.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mente, P., Phaahlamohlaka, T.N., Mashindi, V. et al. Polystyrene-b-poly(acrylic acid) nanospheres for the synthesis of size-controlled cobalt nanoparticles encapsulated inside hollow carbon spheres. J Mater Sci 56, 2113–2128 (2021). https://doi.org/10.1007/s10853-020-05323-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-020-05323-w