Abstract
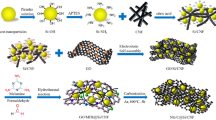
Commercialization of tin dioxide-based anodes for lithium-ion batteries has still not been achieved mainly due to the poor cycling performance caused by the huge volume changes of the electrodes. We herein synthesized a three-dimensional tin dioxide–graphene composite nanofiber (3D SnO2/GNF) membrane via a hydrothermal and electrospinning method assisted by a subsequent calcination process. In this cross-linked three-dimensional network, SnO2 particles are loaded on the graphene crystal structure uniformly, with the aggregation and volume expansion partially inhibited. As a free-standing 3D network, the resultant nanofiber membrane could be used as the anode directly without the addition of the binder and conductive agent. Serving as a binder-free anode material for LIBs, the SnO2/GNF anode exhibits good electrochemical performance with high reversible capacity and excellent cycling stability. More specifically, a high capacity of 763.9 mAh g−1 was obtained at a current density of 100 mA g−1 after 300 cycles. The extraordinary performance could be ascribed to the positive synergistic effect of the nanosized SnO2 particles and graphene.







Similar content being viewed by others
References
Zhang X, Ji L, Toprakci O, Liang Y, Alcoutlabi M (2011) Polym Rev 51:239. doi:10.1080/15583724.2011.593390
Winter M, Besenhard JO (1999) Electrochim Acta 45:31. doi:10.1016/S0013-4686(99)00191-7
P Poizot, S Laruelle, S Grugeon, L Dupont, JM Tarascon (2000) Nature 407: 496. http://www.nature.com/nature/journal/v407/n6803/suppinfo/407496a0_S1.html
Deng D, Kim MG, Lee JY, Cho J (2009) Energy Environ Sci 2:818. doi:10.1039/B823474D
Liu B, Zhang J, Wang X et al (2012) Nano Lett 12:3005. doi:10.1021/nl300794f
Guo XW, Fang XP, Sun Y, Shen LY, Wang ZX, Chen LQ (2013) J Power Sources 226:75. doi:10.1016/j.jpowsour.2012.10.068
Wang Y, Lee JY, Zeng HC (2005) Chem Mater 17:3899. doi:10.1021/cm050724f
Larcher D, Beattie S, Morcrette M, Edstroem K, Jumas JC, Tarascon JM (2007) J Mater Chem 17:3759. doi:10.1039/b705421c
Ding SJ, Chen JS, Qi GG et al (2011) J Am Chem Soc 133:21. doi:10.1021/ja108720w
Ding SJ, Wang ZY, Madhavi S, Lou XW (2011) J Mater Chem 21:13860. doi:10.1039/c1jm11902h
Liu J, Li W, Manthiram A (2010) Chem Commun 46:1437. doi:10.1039/B918501A
Dirican M, Yanilmaz M, Fu K, Lu Y, Kizil H, Zhang X (2014) J Power Sources 264:240. doi:10.1016/j.jpowsour.2014.04.102
Dirican M, Yanilmaz M, Fu K, Lu Y, Kizil H, Zhang XW (2014) J Power Sources 264:240. doi:10.1016/j.jpowsour.2014.04.102
Kong J, Liu Z, Yang Z et al (2012) Nanoscale 4:525. doi:10.1039/C1NR10962F
Liang J, Yu X-Y, Zhou H, Wu HB, Ding S, Lou XW (2014) Angew Chem Int Ed 53:12803. doi:10.1002/anie.201407917
Novoselov KS, Geim AK, Morozov SV et al (2004) Science 306:666. doi:10.1126/science.1102896
Stoller MD, Park SJ, Zhu YW, An JH, Ruoff RS (2008) Nano Lett 8:3498. doi:10.1021/nl802558y
Bolotin KI, Sikes KJ, Jiang Z et al (2008) Solid State Commun 146:351. doi:10.1016/j.ssc.2008.02.024
Yue WB, Lin ZZ, Jiang SH, Yang XJ (2012) J Mater Chem 22:16318. doi:10.1039/c2jm30805c
Ding SJ, Luan DY, Boey FYC, Chen JS, Lou XW (2011) Chem Commun 47:7155. doi:10.1039/c1cc11968k
Paek SM, Yoo E, Honma I (2009) Nano Lett 9:72. doi:10.1021/nl802484w
Li S, Wang YZ, Lai C et al (2014) J Mater Chem A 2:10211. doi:10.1039/c4ta01131g
Liu C, Wang P, Du C et al (2017) J Nanosci Nanotechnol 17:1877. doi:10.1166/jnn.2017.13013
Sher Shah MSA, Lee J, Park AR et al (2017) Electrochim Acta 224:201. doi:10.1016/j.electacta.2016.12.049
Dong Z, Kennedy SJ, Wu Y (2011) J Power Sources 196:4886. doi:10.1016/j.jpowsour.2011.01.090
Lee J, Jo C, Park B et al (2014) Nanoscale 6:10147. doi:10.1039/C4NR01033G
Becerril HA, Mao J, Liu Z, Stoltenberg RM, Bao Z, Chen Y (2008) ACS Nano 2:463. doi:10.1021/nn700375n
Krissanasaeranee M, Supaphol P, Wongkasemjit S (2010) Mater Chem Phys 119:175. doi:10.1016/j.matchemphys.2009.08.040
Kudin KN, Ozbas B, Schniepp HC, Prud’homme RK, Aksay IA, Car R (2008) Nano Lett 8:36. doi:10.1021/nl071822y
Wang D, Li X, Yang J et al (2013) Phys Chem Chem Phys 15:3535. doi:10.1039/C3CP44172E
Shen Z, Hu Y, Chen Y et al (2016) Electrochim Acta 188:661. doi:10.1016/j.electacta.2015.12.062
Chen Y, Hu Y, Shen Z et al (2016) Electrochim Acta 210:53. doi:10.1016/j.electacta.2016.05.086
Sun W, Hu R, Liu H et al (2014) J Power Sources 268:610. doi:10.1016/j.jpowsour.2014.06.039
Han S, Jang B, Kim T, Oh SM, Hyeon T (2005) Adv Func Mater 15:1845. doi:10.1002/adfm.200500243
Park M-S, Wang G-X, Kang Y-M, Wexler D, Dou S-X, Liu H-K (2007) Angew Chem 119:764. doi:10.1002/ange.200603309
Shahid M, Yesibolati N, Reuter MC, Ross FM, Alshareef HN (2014) J Power Sources 263:239. doi:10.1016/j.jpowsour.2014.03.146
Zhou X, Wan L-J, Guo Y-G (2013) Adv Mater 25:2152. doi:10.1002/adma.201300071
Wan Y, Sha Y, Luo S et al (2015) J Power Sources 295:41. doi:10.1016/j.jpowsour.2015.06.125
Acknowledgements
This project is financially supported by the Aviation Science Foundation of China (2008ZH68002), the New Century Talent Support Plan of the Ministry of Education of China (2007NCET-07-0723) and the National Natural Science Foundation of China (60936003).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Wu, Y., Chen, Y., Lin, J. et al. Three-dimensional tin dioxide–graphene composite nanofiber membrane as binder-free anode for high-performance lithium-ion batteries. J Mater Sci 52, 8097–8106 (2017). https://doi.org/10.1007/s10853-017-1017-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-017-1017-6