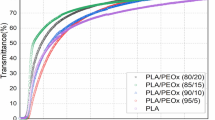
Abstract
Nowadays, due to the rapid development of the times, the study of green controlled release active packaging film has become an integral part of food packaging industry. In order to prepare eco-friendly food green packaging with sustainable release of active ingredients, this study used saturated aqueous solution method to prepare p-methoxybenzaldehyde/β-cyclodextrin inclusion complex, and analyzed the effects of three single factors, namely inclusion temperature, mixing time and inclusion ratio, on the entrapment efficiency and loading capacity. It is found that when the inclusion temperature was 50 °C, mixing time was 4 h and inclusion ratio was 10:1, the entrapment efficiency was 75.18%, the loading capacity could reach 7.52%. Meanwhile, the molecular docking method was used to study molecular interactions, indicating that p-methoxybenzaldehyde can form stable inclusion complexes with β-cyclodextrin. Polylactic acid (PLA) was then used as the base fluid to prepare a p-methoxybenzaldehyde releasable film, which was protected by β-cyclodextrin resulting in a significantly lower release rate than that of added p-methoxybenzaldehyde directly to the film. The inclusion complex was only physical mixing and did not react with PLA, and the inclusion of β-cyclodextrin allowed for effective control of the continuous release of p-methoxybenzaldehyde from the film.
Graphical abstract













Similar content being viewed by others
References
Lacoste, A., Schaich, K.M., Zumbrunnen, D., Yam, K.L.: Advancing controlled release packaging through smart blending. Packag. Technol. Sci. 18, 77–87 (2005). https://doi.org/10.1002/pts.675
Okada, M.: Chemical syntheses of biodegradable polymers. Prog. Polym. Sci. 27, 87–133 (2002). https://doi.org/10.1016/S0079-6700(01)00039-9
Tsuji, H., Ikada, Y.: Properties and morphologies of poly(L-lactide): 1 annealing condition effects on properties and morphologies of poly(L-lactide). Polymer 36, 2709–2716 (1995). https://doi.org/10.1016/0032-3861(95)93647-5
Drumright, R.E., Gruber, P.R., Henton, D.E.: Polylactic acid technology. Adv. Mater. 12, 1841–1846 (2000). https://doi.org/10.1002/1521-4095(200012)12:233.0.CO;2-E
Davis, M.E., Brewster, M.E.: Cyclodextrin-based pharmaceutics: Past, present and future. Nat. Rev. Drug Discovery. 3, 1023–1035 (2004). https://doi.org/10.1038/nrd1576
Valle, E.M.M.D.: Cyclodextrins and their uses: a review. Process Biochem. 39, 1033–1046 (2004). https://doi.org/10.1016/S0032-9592(03)00258-9
Saokham, P., Loftsson, T.: γ-Cyclodextrin. Int. J. Pharm. 516, 278–292 (2017). https://doi.org/10.1016/j.ijpharm.2016.10.062
Gong, L., Li, T.T., Chen, F., Duan, X.W., Yuan, Y.F., Zhang, D.D., Jiang, Y.M.: An inclusion complex of eugenol into β-cyclodextrin: preparation, and physico chemical and antifungal characterization. Food Chem. 196, 324–330 (2016). https://doi.org/10.1016/j.foodchem.2015.09.052
Garcia-Soteloa, D., Silva-Espinozaa, B., Perez-Tellob, M., Olivasc, I., Alvarez-Parrillad, E., González-Aguilara, G.A., Ayala-Zavala, J.F.: Antimicrobial activity and thermal stability of rosemary essential oil: β-cyclodextrin capsules applied in tomato juice. LWT Food Sci. Technol. 111, 837–845 (2019). https://doi.org/10.1016/j.lwt.2019.05.061
Li, Q., Pu, H.Y., Tang, P.X., Tang, B., Sun, Q.M., Li, H.: Propyl gallate/cyclodextrin supramolecular complexes with enhanced solubility and radical scavenging capacity. Food Chem. 245, 1062–1069 (2018). https://doi.org/10.1016/j.foodchem.2017.11.065
Wu, Y.P., Xiao, Y., Yue, Y.X., Zhong, K., Zhao, Y.L., Gao, H.: A deep insight into mechanism for inclusion of 2R, 3R-dihydromyricetin with cyclodextrins and the effect of complexation on antioxidant and lipid-lowering activities. Food Hydrocoll. 103, 105718 (2020). https://doi.org/10.1016/j.foodhyd.2020.105718
Suvarna, V., Gujar, P., Murahari, M.: Complexation of phytochemicals with cyclodextrin derivatives-An insight. Biomed. Pharmacother. 88, 1122–1144 (2017). https://doi.org/10.1016/j.biopha.2017.01.157
Barbieria, N., Sanchez-Contrerasc, A., Canto, A., Cauich-Rodrigueze, J.V., Vargas-Coronadoe, R., Calvo-Irabiend, L.M.: Effect of cyclodextrins and Mexican oregano (Lippia graveolens Kunth) chemotypes on the microencapsulation of essential oil. Ind. Crops Prod. 121, 114–123 (2018). https://doi.org/10.1016/j.indcrop.2018.04.081
Pellicer, J.A., Fortea, M.I., Trabal, J., Rodríguez-López, M.I., Carazo-Díaz, C., Gabaldón, J.A., Núñez-Delicado, E.: Optimization of the microencapsulation of synthetic strawberry flavour with different blends of encapsulating agents using spray drying. Powder Technol. 338, 591–598 (2018). https://doi.org/10.1016/j.powtec.2018.07.080
Żyżelewicz, D., Oracz, J., Kaczmarska, M., Budryn, G., Grzelczyk, J.: Preparation and characterization of inclusion complex of (+)-catechin with β-cyclodextrin. Food Res. Int. 113, 263–268 (2018). https://doi.org/10.1016/j.foodres.2018.07.018
Olga, G., Styliani, C., Ioannis, R.G.: Coencapsulation of ferulic and gallic acid in hp-b-cyclodextrin. Food Chem. 185, 33–40 (2015). https://doi.org/10.1016/j.foodchem.2015.03.058
Zhu, Z.Y., Luo, Y., Liu, Y., Wang, X.T., Liu, F., Guo, M.Z., Wang, Z., Liu, A.J., Zhang, Y.M.: Inclusion of chrysin in β-cyclodextrin and its biological activities. J. Drug Delivery Sci. Technol. 31, 176–186 (2016). https://doi.org/10.1016/j.jddst.2016.01.002
Chen, G., Liu, B.: Cellulose sulfate based film with slow-release antimicrobial properties prepared by incorporation of mustard essential oil and β-cyclodextrin. Food Hydrocoll. 55, 100–107 (2016). https://doi.org/10.1016/j.foodhyd.2015.11.009
Shreaz, S., Bhatia, R., Khan, N., Muralidhar, S., Basir, S.F., Manzoor, N., Khan, L.A.: Exposure of Candida to p-anisaldehyde inhibits its growth and ergosterol biosynthesis. J. Gen. Appl. Microbiol. 57, 129–136 (2011). https://doi.org/10.2323/jgam.57.129
Okamoto, K., Narayama, S., Katsuo, A., Shigematsu, I., Yanase, H.: Biosynthesis of p-anisaldehyde by the white-rot basidiomycete Pleurotus ostreatus. J. Biosci. Bioeng. 93, 207–210 (2002). https://doi.org/10.1016/S1389-1723(02)80015-9
Chen, X.R., Zhang, X.W., Meng, R.Z., Zhao, Z.W., Liu, Z.H., Zhao, X.C., Shi, C., Guo, N.: Efficacy of a combination of nisin and p-Anisaldehyde against Listeria monocytogenes. Food Control 66, 100–106 (2016). https://doi.org/10.1016/j.foodcont.2016.01.025
Shi, C., Zhao, X.C., Meng, R.Z., Liu, Z.J., Zhang, G.N., Guo, N.: Synergistic antimicrobial effects of nisin and p-Anisaldehyde on Staphylococcus aureus in pasteurized milk. LWT Food Sci Technol. 84(222), 230 (2017). https://doi.org/10.1016/j.lwt.2017.05.056
Harish, R., Divakar, S., Srivastava, A., Shivanandappa, T.: Isolation of antioxidant compounds from the methanolic extract of the roots of Decalepis hamiltonii (Wight and Arn.). J. Agric. Food Chem. 53, 7709–7714 (2005). https://doi.org/10.1021/jf051047c
Park, B.S., Lee, K.G., Shibamoto, T., Lee, S.E., Takeoka, G.R.: Antioxidant activity and characterization of volatile constituents of Taheebo (Tabebuia impetiginosa Martius ex DC). J. Agric. Food Chem. 51, 295–300 (2003). https://doi.org/10.1021/jf020811h
Hu, X., Li, N., Heng, T.T., Fang, L., Lu, C.H.: Functionalization of PVDF-based copolymer via photo-induced p-anisaldehyde catalyzed atom transfer radical polymerization. React. Funct. Polym. 150, 104541 (2020). https://doi.org/10.1016/j.reactfunctpolym.2020.104541
Ramot, Y., Haim-Zada, M., Domb, A.J., Nyska, A.: Biocompatibility and safety of PLA and its copolymers. Adv. Drug Delivery Rev. 107, 153–162 (2016). https://doi.org/10.1016/j.addr.2016.03.012
Lai, S.M., Wu, S.H., Lin, G.G., Don, T.M.: Unusual mechanical properties of melt-blended poly(lactic acid)(PLA)/clay nanocomposites. Eur. Polym. J. 52, 193–206 (2014). https://doi.org/10.1016/j.eurpolymj.2013.12.012
Imakura, H., Yamada, Y., Fukazawa, R.: Packaging film, material for keeping freshness of food and freshness-keeping method. JP Pat. Appl. 1992, 13 (1992)
Wen, P., Zhu, D.H., Feng, K., Liu, F.J., Lou, W.Y., Li, N., Zong, M.H., Wu, H.: Fabrication of electrospun polylactic acid nanofilm incorporating cinnamon essential oil/β-cyclodextrin inclusion complex for antimicrobial packaging. Food Chem. 196, 996–1004 (2016). https://doi.org/10.1016/j.foodchem.2015.10.043
Estrada-Villegas, G.M., Martínez-Hernández, R.C., Morales, J., Olayo, R.: Incorporation of ciprofloxacin/beta cyclodextrin inclusion complex to polylactic acid electrospun fibers and modeling of the release behavior. Rev. Mex. Ing. Quim. 18, 737–747 (2019). https://doi.org/10.24275/uam/izt/dcbi/revmexingquim/2019v18n2/Estrada
Chen, J.W., Li, Y.X., Shi, W.Z., Zheng, H., Wang, L., Li, L.: Release of cinnamaldehyde and thymol from PLA/Tilapia fish gelatin-sodium alginate bilayer films to liquid and solid food simulants, and Japanese Sea Bass: a comparative study. Molecules 26, 7140 (2021). https://doi.org/10.3390/molecules26237140
Moradi, S., Barati, A., Tonelli, A.E., Hamedi, H.: Chitosan-based hydrogels loading with thyme oil cyclodextrin inclusion compounds: from preparation to characterization. Eur. Polym. J. 122, 109303 (2020). https://doi.org/10.1016/j.eurpolymj.2019.109303
Xiao, Z.B., Hou, W.J., Kang, Y.X., Niu, Y.W., Kou, X.R.: Encapsulation and sustained release properties of watermelon flavor and its characteristic aroma compounds from γ-cyclodextrin inclusion complexes. Food Hydrocoll. 97, 10520 (2019). https://doi.org/10.1016/j.foodhyd.2019.105202
Brooijmans, N., Kuntz, I.D.: Molecular recognition and docking algorithms. Annu. Rev. Biophys. Biomol. Struct. 32, 335–373 (2003). https://doi.org/10.1146/annurev.biophys.32.110601.142532
Śledź, P., Caflisch, A.: Protein structure-based drug design: from docking to molecular dynamics. Curr. Opin. Struct. Biol. 48, 93–102 (2018). https://doi.org/10.1016/j.sbi.2017.10.010
Zhang, C.L., Liu, J.C., Yang, W.B., Chen, D.L., Jiao, Z.G.: Experimental and molecular docking investigations on the inclusion mechanism of the complex of phloridzin and hydroxypropyl-β-cyclodextrin. Food Chem. 215, 124–128 (2017). https://doi.org/10.1016/j.foodchem.2016.07.155
Morris, G.M., Huey, R., Lindstrom, W., Sanner, M.F., Belew, R.K., Goodsell, D.S., Olson, A.J.: AutoDock4 and AutoDockTools4: automated docking with selective receptor flexibility. J. Comput. Chem. 30, 2785–2791 (2009). https://doi.org/10.1002/jcc.21256
Li, J.Q., Geng, S., Wang, Y., Lv, Y.H., Wang, H.B., Liu, B.G., Liang, G.Z.: The interaction mechanism of oligopeptides containing aromatic rings with β-cyclodextrin and its derivatives. Food Chem. 286, 441–448 (2019). https://doi.org/10.1016/j.foodchem.2019.02.021
Sun, L.N., Lu, L.X., Qiu, X.L., Tang, Y.L.: Development of low-density polyethylene antioxidant active films containing α-tocopherol loaded with MCM-41(Mobil Composition of Matter No. 41) mesoporous silica. Food Control 71, 193–199 (2017). https://doi.org/10.1016/j.foodcont.2016.06.025
Jiang, L.W., Yang, J.D., Wang, Q., Ren, L.L., Zhou, J.: Physicochemical properties of catechin/β-cyclodextrin inclusion complex obtained via co-precipitation. CyTA J. Food. 17, 544–551 (2019). https://doi.org/10.1080/19476337.2019.1612948
Altun, A., Swesi, O.A.A., Alhatab, B.S.S.: Structural and spectroscopic (UV–Vis, IR, Raman, and NMR) characteristics of anisaldehydes that are flavoring food additives: a density functional study in comparison with experiments. J. Mol. Struct. 1128, 590–605 (2017). https://doi.org/10.1016/j.molstruc.2016.09.035
Wang, X.G., Luo, Z.G., Xiao, Z.G.: Preparation, characterization, and thermal stability of β-cyclodextrin/soybean lecithin inclusion complex. Carbohydr. Polym. 101, 1027–1032 (2014). https://doi.org/10.1016/j.carbpol.2013.10.042
Bensouiki, S., Belaib, F., Sindt, M., Rup-Jacques, S., Magri, P., Ikhlef, A., Meniai, A.H.: Synthesis of cyclodextrins-metronidazole inclusion complexes and incorporation of metronidazole - 2-hydroxypropyl-β-cyclodextrin inclusion complex in chitosan nanoparticles. J. Mol. Struct. (2022). https://doi.org/10.1016/j.molstruc.2021.131298
Lin, Y., Huang, R., Sun, X.X., Yu, X., Xiao, Y., Wang, L., Hu, W.Z., Zhong, T.: The p-Anisaldehyde/β-cyclodextrin inclusion complexes as a sustained release agent: characterization, storage stability, antibacterial and antioxidant activity. Food Control 132, 108561 (2022). https://doi.org/10.1016/j.foodcont.2021.108561
Abarca, R.L., Rodríguez, F.J., Guarda, A., Galotto, M.J., Bruna, J.E.: Characterization of beta-cyclodextrin inclusion complexes containing an essential oil component. Food Chem. 196, 968–975 (2016). https://doi.org/10.1016/j.foodchem.2015.10.023
Krishnaswamy, K., Orsat, V., Thangavel, K.: Synthesis and characterization of nano-encapsulated catechin by molecular inclusion with beta-cyclodextrin. J. Food Eng. 111, 255–264 (2012). https://doi.org/10.1016/j.jfoodeng.2012.02.024
Yang, Z.J., Xiao, Z.B., Ji, H.B.: Solid inclusion complex of terpinen-4-ol/ β-cyclodextrin: kinetic release, mechanism and its antibacterial activity. Flavour Fragrance J. 30, 179–187 (2015). https://doi.org/10.1002/ffj.3229
Cinà, V., Russo, M., Lazzara, G., Martino, D.C., Meo, P.L.: Pre- and post-modification of mixed cyclodextrin-calixarene co-polymers: a route towards tenability. Carbohydr. Polym. 157, 1393–1403 (2017). https://doi.org/10.1016/j.carbpol.2016.11.018
Chen, M., Li, Y.H., Li, Y.G., Li, X.L., Zhao, S.Y., Yang, L.J., Liu, X.Y., Zhang, J.Q.: Molecular dynamics simulations and theoretical calculations of cyclodextrin-polydatin inclusion complexes. J. Mol. Struct. 1230, 129840 (2021). https://doi.org/10.1016/j.molstruc.2020.129840
Liu, J.Y., Zhang, S.D., Zhao, X.Y., Lu, Y., Song, M., Wu, S.Z.: Molecular simulation and experimental study on the inclusion of rutin with β-cyclodextrin and its derivative. J. Mol. Struct. 1254, 132359 (2022). https://doi.org/10.1016/j.molstruc.2022.132359
Phaechamud, T., Chitrattha, S.: Pore formation mechanism of porous poly(dl-lactic acid) matrix membrane. Mater. Sci. Eng. 61, 744–752 (2016). https://doi.org/10.1016/j.msec.2016.01.014
Liu, W.L., Yan, C.R., Wei, M., Lei, Y.J., Huang, J.L., Zhang, Y., Zhao, L.M.: Comparison of antibacterial properties of different polylactic acid composite film. Mod. Food Sci. Technol. 36, 175–179 (2020). https://doi.org/10.13982/j.mfst.1673-9078.2020.3.023
Chun, S., Jeon, G.H., Cho, H.J., Ji, Y.I.: Impact of hysterectomy on ovarian reserve in early postoperative period. Korean J. Obstet. Gynecol. 55, 17–21 (2012). https://doi.org/10.5468/KJOG.2012.55.1.17
Acknowledgements
This work was supported by National Key Research and Development Program of China (2016YFD0400701).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, J., Lu, L. & Pan, L. Synthesis of p-methoxybenzaldehyde/β-cyclodextrin inclusion complex and studies of its release properties in polylactic acid film. J Incl Phenom Macrocycl Chem 103, 21–34 (2023). https://doi.org/10.1007/s10847-022-01173-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-022-01173-y