Abstract
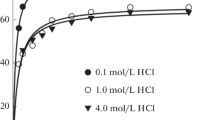
A p-tert-octylcalix[4]arene derivative with four phenyl ketonic side arms has been synthesized as a solvent extraction reagent for precious metal ions in order to compare the extraction behavior with that of the corresponding methyl ketonic derivative of p-tert-octylcalix[4]arene in nitric acid media. The extraction efficiency of the phenyl ketonic derivative for palladium was superior to that of the methyl ketonic one over the whole nitric acid concentration range. Silver and platinum were also moderately extracted by the phenyl ketonic derivative in low nitric acid concentration. The results demonstrated that the extraction behavior of the ligands towards precious metals is related to several structural factors, such as the flexibility of the side arms, the electric charge on the carbonyl oxygen atoms and the lipophilic properties. This work also determined the extraction mechanism of palladium with the phenyl ketonic derivative of p-tert-octylcalix[4]arene based on slope analysis and the peak shift for the FT-IR spectra of the ligand before and after the metal loading. Stripping from the organic phase after forward extraction was performed to separate palladium and silver.









Similar content being viewed by others
References
Alexander, T., Yordanov, D., Max, R.: Solution extraction of transition and post-transition heavy and precious metals by chelate and macrocyclic ligands. Coord. Chem. Rev. 170, 93–124 (1998)
Baba, Y., Eguchi, T., Inoue, K.: Solvent extraction of palladium(II) from aqueous chloride media with 1,2-bis(tert-hexylthio)ethane. Bull. Chem. Soc. Jpn 59, 1321–1325 (1986)
Iwakuma, M., Oshima, T., Baba, Y.: Chemical structure-binding/extractability relationship using new ligands containing sulfur and nitrogen atoms as donor atoms for precious metals. Solv. Extr. Res. Dev. Jpn 15(21), 21–35 (2008)
Koch, K.R.: The reactions of transition metal ions with chlorine(III). Coord. Chem. Rev. 216–217, 449–472 (2001)
Gholivand, M.B., Nozari, N.: Extraction and spectrophotometric determination of trace amount of Pd(II) with 2,2′-dithiodianiline. Talanta 52, 1055–1060 (2000)
Mhaske, A.A., Dhadke, P.M.: Extraction separation studies of Rh, Pt and Pd using Cyanex 921 in toluene-a possible application to recovery from spent catalysts. Hydrometallurgy 61, 143–150 (2001)
Wisniewski, M.: Palladium(II) extraction by pyridine carboxylic acid esters. J. Radioanal. Nucl. Chem. 246(3), 693–696 (2000)
Gutsche, C.D.: Calixarene. Royal Society of Chemistry, Cambridge (1989)
Gutsche, C.D.: Calixarenes Revisited. Royal Society of Chemistry, Cambridge (1998)
Vicens, J., Boehmer, V.: Calixarenes: A Versatile Class of Macrocyclic Compounds. Kluwer Academic Publishers, Amsterdam (1992)
Limetta, G.J., Rogers, R.D., Gopalan, A.S.: Calixarenes for separation. ACS Symposium series 757, American Chemical Society, Washington, 2000
Asfari, Z., Boehmer, V., Harrowfield, J.M., Vicens, J.: Calixarenes 2001. Kluwer, Amsterdam (2001)
Ludwig, R.: Calixarenes in analytical and separation chemistry. Fresenius J. Anal. Chem. 367, 103–128 (2000)
Ohto, K.: Molecular design and metal extraction behavior of calixarene compounds as host extractants. Ion Exch. Solv. Extr. 21, 81–127 (2013)
Roundhill, D.M.: Metal complexes of calixarenes. Prog. Inorg. Chem. 43, 533–592 (1995)
Schwing, M.-J., Arnaud, F., Marques, E.: Cation binding properties of alkyl calixaryl derivatives. A new family of molecular receptors. Pure Appl. Chem. 61(9), 1597–1603 (1989)
Ohto, K., Yano, M., Inoue, K., Yamamoto, T., Goto, M., Nakashio, F., Shinkai, S., Nagasaki, T.: Solvent extraction of trivalent rare earth metal ions with carboxylate derivatives of calixarenes. Anal. Sci. 11(6), 893–902 (1995)
Ohto, K., Shiratsuchi, K., Inoue, K., Goto, M., Nakashio, F., Shinkai, S., Nakashio, T.: Extraction behavior of copper(II) ion by calixarenecarboxylate derivatives preorganized by sodium ion. Solv. Extr. Ion Exc. 14(3), 459–478 (1996)
Marcos, P.M., Ascenso, J.R., Segurado, M.A.P., Pereira, J.L.C.: p-tert-Butyldihomooxacalix[4]arene/p-tert-butylcalix[4]arene: transition and heavy metal cation extraction and transport studies by ketone and ester derivatives. J. Inclu. Phenom. Mol. 42, 281–288 (2002)
Marcos, P.M., Felix, S., Ascenso, J.R., Segurado, M.A.P., Mellah, B., Abidi, R., Bruder, V.H.: A.-Neu, F.: complexation and transport of alkali and alkaline earth metal cations by p-tert-Butyldihomooxacalix[4]arenetetraketone derivatives. Supramol. Chem. 18(2), 147–159 (2006)
Ohto, K., Murakami, E., Shinohara, T., Shiratsuchi, K., Inoue, K., Iwasaki, M.: Selective extraction of silver(I) over palladium(II) with methyl ketonic derivatives of calixarenes from highly concentrated nitric acid. Anal. Chim. Acta 341, 275–283 (1997)
Jayakumar, M., Venkatesan, K.A., Srinivasan, T.G., Vasudeva Rao, P.R.: Studies on the feasibility of electrochemical recovery of palladium from high-level liquid waste. Electrochim. Acta 54, 1083–1088 (2009)
Acknowledgments
This was financially supported by “The Environment Research and Technology Development Fund, No. 3 K-123022, from the Ministry of the Environment, Government of Japan”.
Author information
Authors and Affiliations
Corresponding author
Additional information
The present paper is dedicated to Prof. Jack Harrowfield and Dr. Jacques Vicens on the celebration of their 70th birthday.
Rights and permissions
About this article
Cite this article
Kim, J.Y., Morisada, S., Kawakita, H. et al. Relationship between chemical structure and extraction efficiency toward palladium with ketonic derivatives of p-tert-octylcalix[4]arene in nitric acid media. J Incl Phenom Macrocycl Chem 82, 25–32 (2015). https://doi.org/10.1007/s10847-014-0467-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-014-0467-6