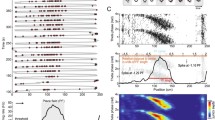
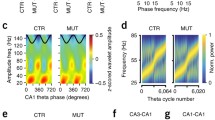
Abstract
Place cells are hippocampal neurons encoding the position of an animal in space. Studies of place cells are essential to understanding the processing of information by neural networks of the brain. An important characteristic of place cell spike trains is phase precession. When an animal is running through the place field, the discharges of the place cells shift from the ascending phase of the theta rhythm through the minimum to the descending phase. The role of excitatory inputs to pyramidal neurons along the Schaffer collaterals and the perforant pathway in phase precession is described, but the role of local interneurons is poorly understood. Our goal is estimating of the contribution of field CA1 interneurons to the phase precession of place cells using mathematical methods. The CA1 field is chosen because it provides the largest set of experimental data required to build and verify the model. Our simulations discover optimal parameters of the excitatory and inhibitory inputs to the pyramidal neuron so that it generates a spike train with the effect of phase precession. The uniform inhibition of pyramidal neurons best explains the effect of phase precession. Among interneurons, axo-axonal neurons make the greatest contribution to the inhibition of pyramidal cells.








Similar content being viewed by others
Data availibility statement
Our code is available on GitHub https://github.com/ivanmysin/Phase_precession.
References
Baker, J. L., & Olds, J. L. (2007). Theta phase precession emerges from a hybrid computational model of a ca3 place cell. Cognitive neurodynamics, 1(3), 237–248. https://doi.org/10.1007/s11571-007-9018-9
Behnel, S., Bradshaw, R., Citro, C., Dalcin, L., Seljebotn, D. S., & Smith, K. (2011). Cython: The best of both worlds. Computing in science and engineering, 13(2), 31–39. https://doi.org/10.1109/MCSE.2010.118
Bezaire, M. J., & Soltesz, I. (2013). Quantitative assessment of ca1 local circuits: knowledge base for interneuron-pyramidal cell connectivity. Hippocampus, 23(9), 751–785. https://doi.org/10.1002/hipo.22141
Burgess, N., & O’Keefe, J. (2011). Models of place and grid cell firing and theta rhythmicity. Current Opinion in Neurobiology, 21(5), 734–744. https://doi.org/10.1016/j.conb.2011.07.002
Buzsáki, G., & Moser, E. I. (2013). Memory, navigation and theta rhythm in the hippocampal-entorhinal system. Nature Neuroscience, 16(2), 130–138. https://doi.org/10.1038/nn.3304
Castro, L., & Aguiar, P. (2012). Phase precession through acceleration of local theta rhythm: a biophysical model for the interaction between place cells and local inhibitory neurons. Journal of Computational Neuroscience, 33(1), 141–150. https://doi.org/10.1007/s10827-011-0378-0
Chance, F. S. (2012). Hippocampal phase precession from dual input components. The Journal of Neuroscience, 32(47), 16693–703. https://doi.org/10.1523/JNEUROSCI.2786-12.2012
Cutsuridis, V., & Hasselmo, M. (2012). Gabaergic contributions to gating, timing, and phase precession of hippocampal neuronal activity during theta oscillations. Hippocampus, 22(7), 1597–1621. https://doi.org/10.1002/hipo.21002
Dudok, B., Szoboszlay, M., Paul, A., Klein, P. M., Liao, Z., Hwaun, E., Szabo, G. G., Geiller, T., Vancura, B., Wang, B.-S., et al. (2021). Recruitment and inhibitory action of hippocampal axo-axonic cells during behavior. Neuron, 109(23), 3838–38508. https://doi.org/10.1016/j.neuron.2021.09.033
Eichenbaum, H. (2014). Time cells in the hippocampus: a new dimension for mapping memories. Nature Reviews. Neuroscience, 15(11), 732–744. https://doi.org/10.1038/nrn3827
Ferguson, K. A., & Campbell, S. A. (2009). A two compartment model of a ca1 pyramidal neuron. Canadian Applied Mathematics Quarterly, 17(2), 293–307.
Fernandez-Lamo, I., Gomez-Dominguez, D., Sanchez-Aguilera, A., Oliva, A., Morales, A.V., Valero, M., Cid, E., Berenyi, A., & Menendez dela Prida, L. (2019). Proximodistal organization of the ca2 hippocampal area. Cell Reports, 26(7), 1734–17466. https://doi.org/10.1016/j.celrep.2019.01.060
Fernández-Ruiz, A., Oliva, A., Nagy, G. A., Maurer, A. P., Berényi, A., & Buzsáki, G. (2017). Entorhinal-ca3 dual-input control of spike timing in the hippocampus by theta-gamma coupling. Neuron, 93(5), 1213–12265. https://doi.org/10.1016/j.neuron.2017.02.017
Geiller, T., Sadeh, S., Rolotti, S. V., Blockus, H., Vancura, B., Negrean, A., Murray, A. J., Rózsa, B., Polleux, F., Clopath, C., & Losonczy, A. (2022). Local circuit amplification of spatial selectivity in the hippocampus. Nature, 601(7891), 105–109. https://doi.org/10.1038/s41586-021-04169-9
Geiller, T., Vancura, B., Terada, S., Troullinou, E., Chavlis, S., Tsagkatakis, G., Tsakalides, P., ócsai, K., Poirazi, P., Rózsa, B. J., et al. (2020). Large-scale 3d two-photon imaging of molecularly identified ca1 interneuron dynamics in behaving mice. Neuron, 108(5), 968–9839. https://doi.org/10.1016/j.neuron.2020.09.013
Geisler, C., Robbe, D., Zugaro, M., Sirota, A., & Buzsáki, G. (2007). Hippocampal place cell assemblies are speed-controlled oscillators. Proceedings of the National Academy of Sciences of the United States of America, 104(19), 8149–8154. https://doi.org/10.1073/pnas.0610121104
Grienberger, C., Milstein, A. D., Bittner, K. C., Romani, S., & Magee, J. C. (2017). Inhibitory suppression of heterogeneously tuned excitation enhances spatial coding in ca1 place cells. Nature Neuroscience, 20(3), 417–426. https://doi.org/10.1038/nn.4486
Hafting, T., Fyhn, M., Bonnevie, T., Moser, M.-B., & Moser, E. I. (2008). Hippocampus-independent phase precession in entorhinal grid cells. Nature, 453(7199), 1248–1252. https://doi.org/10.1038/nature06957
Harris, C. R., Millman, K. J., van der Walt, S. J., Gommers, R., Virtanen, P., Cournapeau, D., Wieser, E., Taylor, J., Berg, S., Smith, N. J., et al. (2020). Array programming with numpy. Nature, 585(7825), 357–362. https://doi.org/10.1038/s41586-020-2649-2
Harris, K. D., Henze, D. A., Hirase, H., Leinekugel, X., Dragoi, G., Czurkó, A., & Buzsáki, G. (2002). Spike train dynamics predicts theta-related phase precession in hippocampal pyramidal cells. Nature, 417(6890), 738–741. https://doi.org/10.1038/nature00808
Hunter, J. D. (2007). Matplotlib: A 2d graphics environment. Computing in science and engineering, 9(3), 90–95. https://doi.org/10.1109/MCSE.2007.55
Jeffery, K. J. (2011). Place cells, grid cells, attractors, and remapping. Neural plasticity, 2011, 182602. https://doi.org/10.1155/2011/182602
Kamondi, A., Acsády, L., Wang, X. J., & Buzsáki, G. (1998). Theta oscillations in somata and dendrites of hippocampal pyramidal cells in vivo: activity-dependent phase-precession of action potentials. Hippocampus, 8(3), 244–261. https://doi.org/10.1002/(SICI)1098-1063(1998)8:3<244::AID-HIPO7>3.0.CO;2-J
Kempter, R., Leibold, C., Buzsáki, G., Diba, K., & Schmidt, R. (2012). Quantifying circular-linear associations: hippocampal phase precession. Journal of Neuroscience Methods, 207(1), 113–124. https://doi.org/10.1016/j.jneumeth.2012.03.007
Klausberger, T., Magill, P. J., Márton, L. F., Roberts, J. D. B., Cobden, P. M., Buzsáki, G., & Somogyi, P. (2003). Brain-state- and cell-type-specific firing of hippocampal interneurons in vivo. Nature, 421(6925), 844–848. https://doi.org/10.1038/nature01374
Lam, S. K., Pitrou, A., & Seibert, S. (2015). Numba: A LLVM-based Python JIT compiler, pp. 1–6. ACM Press. https://doi.org/10.1145/2833157.2833162
Lasztóczi, B., & Klausberger, T. (2016). Hippocampal place cells couple to three different gamma oscillations during place field traversal. Neuron, 91(1), 34–40. https://doi.org/10.1016/j.neuron.2016.05.036
Lee, A. (2010). Circular data. Wiley Interdisciplinary Reviews: Computational Statistics, 2(4), 477–486. https://doi.org/10.1002/wics.98
Losonczy, A., Zemelman, B. V., Vaziri, A., & Magee, J. C. (2010). Network mechanisms of theta related neuronal activity in hippocampal ca1 pyramidal neurons. Nature Neuroscience, 13(8), 967–972. https://doi.org/10.1038/nn.2597
Magee, J. C. (2001). Dendritic mechanisms of phase precession in hippocampal ca1 pyramidal neurons. Journal of Neurophysiology, 86(1), 528–532. https://doi.org/10.1152/jn.2001.86.1.528
Malvache, A., Reichinnek, S., Villette, V., Haimerl, C., & Cossart, R. (2016). Awake hippocampal reactivations project onto orthogonal neuronal assemblies. Science, 353(6305), 1280–1283. https://doi.org/10.1126/science.aaf3319
Mardia, J. (1999). Directional Statistics. John Wiley and Sons, Inc. https://doi.org/10.1002/9780470316979
Mardia, K. V. (1976). Linear-circular correlation coefficients and rhythmometry. Biometrika, 63(2), 403. https://doi.org/10.2307/2335637
Markiewicz, C. J., Gorgolewski, K. J., Feingold, F., Blair, R., Halchenko, Y. O., Miller, E., Hardcastle, N., Wexler, J., Esteban, O., Goncavles, M., et al. (2021). The openneuro resource for sharing of neuroscience data. eLife, 10. https://doi.org/10.7554/eLife.71774
Marshall, L., Henze, D. A., Hirase, H., Leinekugel, X., Dragoi, G., & Buzsáki, G. (2002). Hippocampal pyramidal cell-interneuron spike transmission is frequency dependent and responsible for place modulation of interneuron discharge. The Journal of Neuroscience, 22(2), 197.
Masurkar, A. V., Srinivas, K. V., Brann, D. H., Warren, R., Lowes, D. C., & Siegelbaum, S. A. (2017). Medial and lateral entorhinal cortex differentially excite deep versus superficial ca1 pyramidal neurons. Cell Reports, 18(1), 148–160. https://doi.org/10.1016/j.celrep.2016.12.012
Maurer, A. P., Cowen, S. L., Burke, S. N., Barnes, C. A., & McNaughton, B. L. (2006). Phase precession in hippocampal interneurons showing strong functional coupling to individual pyramidal cells. The Journal of Neuroscience, 26(52), 13485–13492. https://doi.org/10.1523/JNEUROSCI.2882-06.2006
Mehta, M. R., Lee, A. K., & Wilson, M. A. (2002). Role of experience and oscillations in transforming a rate code into a temporal code. Nature, 417(6890), 741–746. https://doi.org/10.1038/nature00807
Mizuseki, K., Diba, K., Pastalkova, E., Teeters, J., Sirota, A., & Buzsáki, G. (2014). Neurosharing: large-scale data sets (spike, lfp) recorded from the hippocampal-entorhinal system in behaving rats. [version 1; peer review: 4 approved]. F1000Research, 3, 98. https://doi.org/10.12688/f1000research.3895.1
Mizuseki, K., Royer, S., Diba, K., & Buzsáki, G. (2012). Activity dynamics and behavioral correlates of ca3 and ca1 hippocampal pyramidal neurons. Hippocampus, 22(8), 1659–1680. https://doi.org/10.1002/hipo.22002
Mizuseki, K., Sirota, A., Pastalkova, E., & Buzsáki, G. (2009). Theta oscillations provide temporal windows for local circuit computation in the entorhinal-hippocampal loop. Neuron, 64(2), 267–280. https://doi.org/10.1016/j.neuron.2009.08.037
Montgomery, S. M., Betancur, M. I., & Buzsáki, G. (2009). Behavior-dependent coordination of multiple theta dipoles in the hippocampus. The Journal of Neuroscience, 29(5), 1381–1394. https://doi.org/10.1523/JNEUROSCI.4339-08.2009
O’Keefe, J., & Burgess, N. (2005). Dual phase and rate coding in hippocampal place cells: theoretical significance and relationship to entorhinal grid cells. Hippocampus, 15(7), 853–866. https://doi.org/10.1002/hipo.20115
O’Keefe, J., & Recce, M. L. (1993). Phase relationship between hippocampal place units and the eeg theta rhythm. Hippocampus, 3(3), 317–330. https://doi.org/10.1002/hipo.450030307
Oliva, A., Fernández-Ruiz, A., Buzsáki, G., & Berényi, A. (2016). Spatial coding and physiological properties of hippocampal neurons in the cornu ammonis subregions. Hippocampus, 26(12), 1593–1607. https://doi.org/10.1002/hipo.22659
Petersen, P. C., Hernandez, M., & Buzsáki, G. (2020). The buzsaki lab databank - public electrophysiological datasets from awake animals. Zenodo. https://doi.org/10.5281/zenodo.4307883
Pinsky, P. F., & Rinzel, J. (1994). Intrinsic and network rhythmogenesis in a reduced traub model for ca3 neurons. Journal of computational neuroscience, 1(1–2), 39–60.
Quiroga, R. Q., Reddy, L., Kreiman, G., Koch, C., & Fried, I. (2005). Invariant visual representation by single neurons in the human brain. Nature, 435(7045), 1102–1107. https://doi.org/10.1038/nature03687
Royer, S., Zemelman, B. V., Losonczy, A., Kim, J., Chance, F., Magee, J. C., & Buzsaki, G. (2012). Control of timing, rate and bursts of hippocampal place cells by dendritic and somatic inhibition. Nature Neuroscience, 15(5), 769–775. https://doi.org/10.1038/nn.3077
Sanchez-Aguilera, A., Wheeler, D. W., Jurado-Parras, T., Valero, M., Nokia, M. S., Cid, E., Fernandez-Lamo, I., Sutton, N., García-Rincón, D., dela Prida, L. M., et al. (2021). An update to hippocampome.org by integrating single-cell phenotypes with circuit function in vivo. PLoS Biology, 19(5), 3001213. https://doi.org/10.1371/journal.pbio.3001213
Somogyi, P., & Klausberger, T. (2005). Defined types of cortical interneurone structure space and spike timing in the hippocampus. The Journal of Physiology, 562(Pt 1), 9–26. https://doi.org/10.1113/jphysiol.2004.078915
Somogyi, P., Katona, L., Klausberger, T., Lasztóczi, B., & Viney, T. J. (2014). Temporal redistribution of inhibition over neuronal subcellular domains underlies state-dependent rhythmic change of excitability in the hippocampus. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, 369(1635), 20120518. https://doi.org/10.1098/rstb.2012.0518
Storn, R., & Price, K. (1997). Differential evolution - asimple and efficient heuristic for global optimization over continuous spaces. Springer Science and Business Media LLC. https://doi.org/10.1023/a:1008202821328
Sugar, J., & Moser, M.-B. (2019). Episodic memory: Neuronal codes for what, where, and when. Hippocampus, 29(12), 1190–1205. https://doi.org/10.1002/hipo.23132
Thurley, K., Leibold, C., Gundlfinger, A., Schmitz, D., & Kempter, R. (2008). Phase precession through synaptic facilitation. Neural Computation, 20(5), 1285–1324. https://doi.org/10.1162/neco.2008.07-06-292
Traub, R. D., Wong, R. K., Miles, R., & Michelson, H. (1991). A model of a ca3 hippocampal pyramidal neuron incorporating voltage-clamp data on intrinsic conductances. Journal of Neurophysiology, 66(2), 635–650.
Valero, M., Navas-Olive, A., de la Prida, L. M., & Buzsáki, G. (2022). Inhibitory conductance controls place field dynamics in the hippocampus. Cell reports, 40(8), 111232. https://doi.org/10.1016/j.celrep.2022.111232
Valero, M., Zutshi, I., Yoon, E., & Buzsáki, G. (2022). Probing subthreshold dynamics of hippocampal neurons by pulsed optogenetics. Science (New York, N.Y.), 375(6580), 570–574. https://doi.org/10.1126/science.abm1891
Varga, C., Golshani, P., & Soltesz, I. (2012). Frequency-invariant temporal ordering of interneuronal discharges during hippocampal oscillations in awake mice. Proceedings of the National Academy of Sciences of the United States of America, 109(40), 2726–34. https://doi.org/10.1073/pnas.1210929109
Vinogradova, O. S. (2001). Hippocampus as comparator: role of the two input and two output systems of the hippocampus in selection and registration of information. Hippocampus, 11(5), 578–598. https://doi.org/10.1002/hipo.1073
Virtanen, P., Gommers, R., Oliphant, T. E., Haberland, M., Reddy, T., Cournapeau, D., Burovski, E., Peterson, P., Weckesser, W., Bright, J., et al. (2020). Author correction: Scipy 1.0: fundamental algorithms for scientific computing in python. Nature Methods, 17(3), 352. https://doi.org/10.1038/s41592-020-0772-5
Wallenstein, G. V., & Hasselmo, M. E. (1997). Gabaergic modulation of hippocampal population activity: sequence learning, place field development, and the phase precession effect. Journal of Neurophysiology, 78(1), 393–408. https://doi.org/10.1152/jn.1997.78.1.393
Wheeler, D. W., White, C. M., Rees, C. L., Komendantov, A. O., Hamilton, D. J., & Ascoli, G. A. (2015). Hippocampome.org: a knowledge base of neuron types in the rodent hippocampus. eLife, 4. https://doi.org/10.7554/eLife.09960
Wilson, M. A., & McNaughton, B. L. (1993). Dynamics of the hippocampal ensemble code for space. Science, 261(5124), 1055–1058. https://doi.org/10.1126/science.8351520
Wormington, M., Panaccione, C., Matney, K. M., & Bowen, D. K. (1999). Characterization of structures from x-ray scattering data using genetic algorithms. Philosophical Transactions of the Royal Society of London. Series A: Mathematical, Physical and Engineering Sciences, 357(1761), 2827–2848. https://doi.org/10.1098/rsta.1999.0469
Zutshi, I., Valero, M., Fernández-Ruiz, A., & Buzsáki, G. (2022). Extrinsic control and intrinsic computation in the hippocampal ca1 circuit. Neuron, 110(4), 658–6735. https://doi.org/10.1016/j.neuron.2021.11.015
Funding
This work was supported by the Russian Science Foundation (grant number 20-71-10109)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
We declare no conflict of interests.
Additional information
Action editor: Jonathan D. Victor
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
Equations for currents:
where j \(\in\) S, D, S and D denote the soma and dendrite of pyramidal neurons, respectively (Table 2); E is the reversal potential of the currents (Table 3); g is the maximal conductance (Table 2). The equations and parameters for the synaptic currents (Eq. (3)) are given in the next section. All conductances are measured in the units \(mS/cm^2\). The potential is measured in the unit mV. \([Ca^{2+}]\) - mM.
The equations for intracellular calcium concentration [Ca2+] are:
Equations for calcium concentration
where \(\phi _{Ca} = 0.13 \ mM \cdot cm^2 \cdot nA^{-1}\) is a scaling constant that converts the inward calcium current into internal calcium concentration, \(\beta = 0.075 \ ms^{-1}\).
The gating variables \(h_j\) , \(n_j\), \(s_j\) , \(c_j\) , \(q_j\) , \(j\in \{S,D\}\) are each governed by an equation of a form:
The associated steady state value and time constant are defined in the usual manner
Equations for gate variables
The parameter p is the proportion of the cell area taken by the somatic compartment.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vandyshev, G., Mysin, I. Homogeneous inhibition is optimal for the phase precession of place cells in the CA1 field. J Comput Neurosci 51, 389–403 (2023). https://doi.org/10.1007/s10827-023-00855-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10827-023-00855-x