Abstract
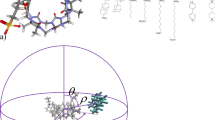
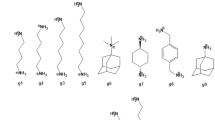
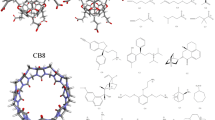
The prediction of host–guest binding affinities with computational modelling is still a challenging task. In the 7th statistical assessment of the modeling of proteins and ligands (SAMPL) challenge, a new host named TrimerTrip was synthesized and the thermodynamic parameters of 16 structurally diverse guests binding to the host were characterized. In the TrimerTrip-guest challenge, only structures of the host and the guests are provided, which indicates that the predictions of both the binding poses and the binding affinities are under assessment. In this work, starting from the binding poses obtained from our previous enhanced sampling simulations in the configurational space, we perform extensive alchemical and end-point free energy calculations to calculate the host–guest binding affinities retrospectively. The alchemical predictions with two widely accepted charge schemes (i.e. AM1-BCC and RESP) are in good agreement with the experimental reference, while the end-point estimates perform poorly in reproducing the experimental binding affinities. Aside from the absolute value of the binding affinity, the rank of binding free energies is also crucial in drug design. Surprisingly, the end-point MM/PBSA method seems very powerful in reproducing the experimental rank of binding affinities. Although the length of our simulations is long and the intermediate spacing is dense, the convergence behavior is not very good, which may arise from the flexibility of the host molecule. Enhanced sampling techniques in the configurational space may be required to obtain fully converged sampling. Further, as the length of sampling in alchemical free energy calculations already achieves several hundred ns, performing direct simulations of the binding/unbinding event in the physical space could be more useful and insightful. More details about the binding pathway and mechanism could be obtained in this way. The nonequilibrium method could also be a nice choice if one insists to use the alchemical method, as the intermediate sampling is avoided to some extent.



Similar content being viewed by others
References
Ndendjio SZ, Liu W, Yvanez N, Meng Z, Zavalij PY, Isaacs L (2020) Triptycene walled glycoluril trimer: synthesis and recognition properties. New J Chem 44:338–345
Caldararu O, Olsson MA, Ignjatović MM, Wang M, Ryde U (2018) Binding free energies in the SAMPL6 octa-acid host–guest challenge calculated with MM and QM methods. J Comput Aided Mol Des 32:1027–1046
Rizzi A, Murkli S, McNeill JN, Yao W, Sullivan M, Gilson MK, Chiu MW, Isaacs L, Gibb BC, Mobley DL (2018) Overview of the SAMPL6 host–guest binding affinity prediction challenge. J Comput Aided Mol Des 32:937–963
Eken Y, Patel P, Díaz T, Jones MR, Wilson AK (2018) SAMPL6 host–guest challenge: binding free energies via a multistep approach. J Comput Aided Mol Des 32:1097–1115
Mobley DL, Wymer KL, Lim NM, Guthrie JP (2014) Blind prediction of solvation free energies from the SAMPL4 challenge. J Comput Aided Mol Des 28:135–150
Monroe JI, Shirts MR (2014) Converging free energies of binding in cucurbit[7]uril and octa-acid host–guest systems from SAMPL4 using expanded ensemble simulations. J Comput Aided Mol Des 28:401–415
Song LF, Bansal N, Zheng Z, Merz KM (2018) Detailed potential of mean force studies on host–guest systems from the SAMPL6 challenge. J Comput Aided Mol Des 32:1013–1026
Procacci P, Guarrasi M, Guarnieri G (2018) SAMPL6 host–guest blind predictions using a non equilibrium alchemical approach. J Comput Aided Mol Des 32:965–982
Nishikawa N, Han K, Wu X, Tofoleanu F, Brooks BR (2018) Comparison of the umbrella sampling and the double decoupling method in binding free energy predictions for SAMPL6 octa-acid host–guest challenges. J Comput Aided Mol Des 32:1075–1086
Sun Z, Zhang JZH (2020) Thermodynamic insights of base flipping in TNA duplex: force fields, salt concentrations, and free-energy simulation methods. CCS Chem 2:1026–1039
Wang X, Xingzhao T, Boming D, John ZH, Sun Z (2019) (2019) BAR-based optimum adaptive steered MD for configurational sampling. J Comput Chem 40:1270–1289
Wang X, Tu X, Zhang JZH, Sun Z (2018) BAR-based optimum adaptive sampling regime for variance minimization in alchemical transformation: the nonequilibrium stratification. Phys Chem Chem Phys 20:2009–2021
Sun ZX, Wang XH, Zhang JZH (2017) BAR-based optimum adaptive sampling regime for variance minimization in alchemical transformation. Phys Chem Chem Phys 19:15005–15020
Sun Z, Wang X, Zhang JZH, He Q (2019) Sulfur-substitution-induced base flipping in the DNA duplex. Phys Chem Chem Phys 21:14923–14940
Swope WC (1982) A computer simulation method for the calculation of equilibrium constants for the formation of physical clusters of molecules: application to small water clusters. J Chem Phys 76:637
Pham TT, Shirts MR (2011) Identifying low variance pathways for free energy calculations of molecular transformations in solution phase. J Chem Phys 135:034114
Procacci P, Chelli R (2017) Statistical mechanics of ligand-receptor noncovalent association, revisited: binding site and standard state volumes in modern alchemical theories. J Chem Theory Comput 13:1924–1933
Sun Z, Wang X, Zhang JZ (2020) Theoretical understanding of the thermodynamics and interactions in transcriptional regulator TtgR-ligand binding. Phys Chem Chem Phys 22:1511–1524
Sun Z, Wang X, Zhao Q, Zhu T (2019) Understanding aldose reductase-inhibitors interactions with free energy simulation. J Mol Graph Model 91:10–21
Wang X, Sun Z (2019) Understanding PIM-1 kinase inhibitor interactions with free energy simulation. Phys Chem Chem Phys 21:7544–7558
Zwanzig RW (1954) High temperature equation of state by a perturbation method. J Chem Phys 22:1420–1426
Hummer G (2001) Fast-growth thermodynamic integration: error and efficiency analysis. J Chem Phys 114:7330–7337
Eastwood MP, Hardin C, Luthey-Schulten Z, Wolynes PG (2002) Statistical mechanical refinement of protein structure prediction schemes: cumulant expansion approach. J Chem Phys 117:4602–4615
Shirts MR, Bair E, Hooker G, Pande VS (2003) Equilibrium free energies from nonequilibrium measurements using maximum-likelihood methods. Phys Rev Lett 91:140601
Bennett CH (1976) Efficient estimation of free energy differences from Monte Carlo data. J Comput Phys 22:245–268
Fenwick MK, Escobedo FA (2004) On the use of Bennett's acceptance ratio method in multi-canonical-type simulations. J Chem Phys 120:3066–3074
Tan Z (2004) On a likelihood approach for Monte Carlo integration. J Am Stat Assoc 99:1027–1036
Shirts MR, Chodera JD (2008) Statistically optimal analysis of samples from multiple equilibrium states. J Chem Phys 129:124105
Wang X, Sun Z A (2018) Theoretical interpretation of variance-based convergence citeria in perturbation-based theories. arXiv:1803.03123
Sun Z, Wang X, Song J (2017) Extensive assessment of various computational methods for Aspartate’s pKa Shift. J Chem Inf Model 57:1621–1639
Jarzynski C (1997) Equilibrium free-energy differences from nonequilibrium measurements: a master-equation approach. Phys Rev E 56:5018–5035
Wang X, Sun Z (2019) Determination of base flipping free energy landscapes from nonequilibrium stratification. J Chem Inf Model 59:2980–2994
Wang X, He Q, Sun Z (2019) BAR-based multi-dimensional nonequilibrium pulling for indirect construction of a QM/MM free energy landscape. Phys Chem Chem Phys 21:6672–6688
Sun Z (2019) BAR-based multi-dimensional nonequilibrium pulling for indirect construction of QM/MM free energy landscapes: from semi-empirical to ab initio. Phys Chem Chem Phys 21:21942–21959
Bruckner S, Boresch S (2011) Efficiency of alchemical free energy simulations. II. Improvements for thermodynamic integration. J Comput Chem 32:1320–1333
Resat H, Mezei M (1993) Studies on free energy calculations. I. Thermodynamic integration using a polynomial path. J Chem Phys 99:6052–6061
Resat H, Mezei M (1994) Studies on free energy calculations. II. A theoretical approach to molecular solvation. J Chem Phys 101:6126–6140
Kirkwood JG (1935) Statistical mechanics of fluid mixtures. J Chem Phys 3:300
Pearlman DA, Kollman PA (1989) The lag between the Hamiltonian and the system configuration in free energy perturbation calculations. J Chem Phys 91:7831–7839
Straatsma TP, Mccammon JA (1989) Treatment of rotational isomers in free energy calculations. II. Molecular dynamics simulation study of 18-crown-6 in aqueous solution as an example of systems with large numbers of rotational isomeric states. J Chem Phys 91:3631–3637
Jarzynski C (1997) A nonequilibrium equality for free energy differences. Phys Rev Lett 78:2690–2693
Crooks GE (1998) Nonequilibrium measurements of free energy differences for microscopically reversible Markovian systems. J Stat Phys 90:1481–1487
Aqvist J, Medina C, Samuelsson JE (1994) A new method for predicting binding affinity in computer-aided drug design. Protein Eng 7:385–391
Carlson HA, Jorgensen WL (1995) An extended linear response method for determining free energies of hydration. J Phys Chem 99:10667–10673
Wang W, Wang J, Kollman PA (1999) What determines the van der Waals coefficient β in the LIE (linear interaction energy) method to estimate binding free energies using molecular dynamics simulations? Proteins-Struct Funct Bioinform 34:395–402
Leach AR (2001) Molecular modeling principles & applications. Pearson education, Harlow
Lee FS, Chu ZT, Bolger MB, Warshel A (1992) Calculations of antibody-antigen interactions: microscopic and semi-microscopic evaluation of the free energies of binding of phosphorylcholine analogs to McPC603. Protein Eng 5:215–228
Ferrari AM, Degliesposti G, Sgobba M, Rastelli G (2007) Validation of an automated procedure for the prediction of relative free energies of binding on a set of aldose reductase inhibitors. Bioorg Med Chem 15:7865–7877
Rapp C, Kalyanaraman C, Schiffmiller A, Schoenbrun EL, Jacobson MP (2011) A molecular mechanics approach to modeling protein-ligand interactions: relative binding affinities in congeneric series. J Chem Inf Model 51:2082–2089
Miller BR, Mcgee TD, Swails JM, Homeyer N, Gohlke H, Roitberg AE (2012) MMPBSA.py: an efficient program for end-state free energy calculations. J Chem Theory Comput 8:3314–3321
Honig B, Nicholls A (1995) Classical electrostatics in biology and chemistry. Science 268:1144–1149
Su PC, Tsai CC, Mehboob S, Hevener KE, Johnson ME (2015) Comparison of radii sets, entropy, QM methods, and sampling on MM-PBSA, MM-GBSA, and QM/MM-GBSA ligand binding energies of F. tularensis enoyl-ACP reductase (F abI). J Comput Chem 36:1859–1873
Sangpheak W, Khuntawee W, Wolschann P, Pongsawasdi P, Rungrotmongkol T (2014) Enhanced stability of a naringenin/2, 6-dimethyl β-cyclodextrin inclusion complex: molecular dynamics and free energy calculations based on MM-and QM-PBSA/GBSA. J Mol Graph Model 50:10–15
Tsitsanou KE, Hayes JM, Keramioti M, Mamais M, Oikonomakos NG, Kato A, Leonidas DD, Zographos SE (2013) Sourcing the affinity of flavonoids for the glycogen phosphorylase inhibitor site via crystallography, kinetics and QM/MM-PBSA binding studies: comparison of chrysin and flavopiridol. Food Chem Toxicol 61:14–27
Moraca F, Amato J, Ortuso F, Artese A, Pagano B, Novellino E, Alcaro S, Parrinello M, Limongelli V (2017) Ligand binding to telomeric G-quadruplex DNA investigated by funnel-metadynamics simulations. Proc Natl Acad Sci USA 114:E2136–E2145
Valsson O, Tiwary P, Parrinello M (2016) Enhancing important fluctuations: rare events and metadynamics from a conceptual viewpoint. Annu Rev Phys Chem 67:159
Tiwary P, Limongelli V, Salvalaglio M, Parrinello M (2015) Kinetics of protein-ligand unbinding: Predicting pathways, rates, and rate-limiting steps. Proc Natl Acad Sci USA 112:386–391
Huang N, Kalyanaraman C, Bernacki K, Jacobson MP (2006) Molecular mechanics methods for predicting protein–ligand binding. Phys Chem Chem Phys 8:5166–5177
Mendels D, Piccini G, Parrinello M (2018) Collective variables from local fluctuations. J Phys Chem Lett 9:2776–2781
Tiwary P, van de Walle A (2013) Accelerated molecular dynamics through stochastic iterations and collective variable based basin identification. Phys Rev B 87:094304
Hashemian B, Arroyo M (2013) Modeling and enhanced sampling of molecular systems with smooth and nonlinear data-driven collective variables. J Chem Phys 139:214101
https://github.com/samplchallenges/SAMPL7/tree/master/host_guest/Isaacs_clip
Sun Z (2020) SAMPL7 TrimerTrip host-guest binding poses and binding affinities from spherical-coordinates-biased simulations. J Comput Aided Mol Des. https://doi.org/10.1007/s10822-020-00335-9
Sun Z, He Q, Li X, Zhu Z (2020) SAMPL6 host–guest binding affinities and binding poses from spherical-coordinates-biased simulations. J Comput Aided Mol Des 34:589–600
Capelli R, Carloni P, Parrinello M (2019) Exhaustive search of ligand binding pathways via volume-based metadynamics. J Phys Chem Lett 10:3495–3499
Jakalian A, Jack DB, Bayly CI (2002) Fast, efficient generation of high-quality atomic charges. AM1-BCC model: II. Parameterization and validation. J Comput Chem 23:1623–1641
Wang J, Wolf RM, Caldwell JW, Kollman PA, Case DA (2004) Development and testing of a general amber force field. J Comput Chem 25:1157–1173
Jorgensen WL, Chandrasekhar J, Madura JD, Impey RW, Klein ML (1983) Comparison of simple potential functions for simulating liquid water. J Chem Phys 79:926–935
Price DJ, Brooks CL III (2004) A Modified TIP3P water potential for simulation with Ewald summation. J Chem Phys 121:10096–10103
Joung IS, Cheatham TE III (2008) Determination of alkali and halide monovalent ion parameters for use in explicitly solvated biomolecular simulations. J Phys Chem B 112:9020–9041
Joung IS, Cheatham TE (2009) Molecular dynamics simulations of the dynamic and energetic properties of alkali and halide ions using water-model-specific ion parameters. J Phys Chem B 113:13279–13290
Steinbrecher T, Mobley DL, Case DA (2007) Nonlinear scaling schemes for Lennard-Jones interactions in free energy calculations. J Chem Phys 127:214108
Zacharias M, Straatsma TP, Mccammon JA (1994) Separation-shifted scaling, a new scaling method for Lennard-Jones interactions in thermodynamic integration. J Chem Phys 100:9025–9031
Beutler TC, Mark AE, Schaik RCV, Gerber PR, Gunsteren WFV (1994) Avoiding singularities and numerical instabilities in free energy calculations based on molecular simulations. Chem Phys Lett 222:529–539
Levitt M (1983) Protein folding by restrained energy minimization and molecular dynamics. J Mol Biol 170:723–764
Klimovich PV, Shirts MR, Mobley DL (2015) Guidelines for the analysis of free energy calculations. J Comput Aided Mol Des 29:397–411
Chodera JD, Swope WC, Pitera JW, Seok C, Dill KA (2007) Use of the weighted histogram analysis method for the analysis of simulated and parallel tempering simulations. J Chem Theory Comput 3:26–41
Chen W, Deng Y, Russell E, Wu Y, Abel R, Wang L (2018) Accurate calculation of relative binding free energies between ligands with different net charges. J Chem Theory Comput 14:6346–6358
Morgan BR, Massi F (2010) Accurate estimates of free energy changes in charge mutations. J Chem Theory Comput 6:1884–1893
Rocklin GJ, Mobley DL, Dill KA, Hünenberger PH (2013) Calculating the binding free energies of charged species based on explicit-solvent simulations employing lattice-sum methods: An accurate correction scheme for electrostatic finite-size effects. J Chem Phys 139:184103
Lin Y-L, Aleksandrov A, Simonson T, Roux B (2014) An overview of electrostatic free energy computations for solutions and proteins. J Chem Theory Comput 10:2690–2709
Baker NA, Sept D, Joseph S, Holst MJ, McCammon JA (2001) Electrostatics of nanosystems: application to microtubules and the ribosome. Proc Natl Acad Sci USA 98:10037–10041
Onufriev A, Bashford D, Case DA (2004) Exploring protein native states and large-scale conformational changes with a modified generalized born model. Proteins: Struct Funct Bioinf 55:383–394
Feig M, Onufriev A, Lee MS, Im W, Case DA (2004) Performance comparison of generalized born and Poisson methods in the calculation of electrostatic solvation energies for protein structures. J Comput Chem 25:265–284
Case DA (2010) Normal mode analysis of protein dynamics. Curr Opin Struct Biol 4:285–290
Chang CE, Chen W, Gilson MK (2005) Evaluating the accuracy of the quasiharmonic approximation. J Chem Theory Comput 1:1017–1028
Karplus M, Kushick JN (1981) Method for estimating the configurational entropy of macromolecules. Macromolecules 14:325–332
Qiu L, Yan Y, Sun Z, Song J, Zhang JZ (2018) Interaction entropy for computational alanine scanning in protein–protein binding. Wiley Interdisip Rev Comput Mol Sci 8:e1342
Sun Z, Yan YN, Yang M, Zhang JZ (2017) Interaction entropy for protein-protein binding. J Chem Phys 146:124124
Duan L, Liu X, Zhang JZ (2016) Interaction entropy: a new paradigm for highly efficient and reliable computation of protein–ligand binding free energy. J Am Chem Soc 138:5722–5728
Ryckaert JP, Ciccotti G, Berendsen HJC (1977) Numerical integration of the Cartesian equations of motion of a system with constraints: molecular dynamics of n -alkanes. J Comput Phys 23:327–341
Miyamoto S, Kollman PA (1992) Settle: an analytical version of the SHAKE and RATTLE algorithm for rigid water models. J Comput Chem 13:952–962
Pastor RW, Brooks BR, Szabo A (1988) An analysis of the accuracy of Langevin and molecular dynamics algorithms. Mol Phys 65:1409–1419
York DM, Darden TA, Pedersen LG (1993) The effect of long-range electrostatic interactions in simulations of macromolecular crystals: a comparison of the Ewald and truncated list methods. J Chem Phys 99:8345–8348
Tuckerman ME, Berne BJ, Martyna GJ (1991) Molecular dynamics algorithm for multiple time scales: systems with long range forces. J Chem Phys 94:6811–6815
Case DA, Cheatham TE, Tom D, Holger G, Luo R, Merz KM, Alexey O, Carlos S, Bing W, Woods RJ (2005) The amber biomolecular simulation programs. J Comput Chem 26:1668–1688
Martin A, David M (2020) SAMPL7 challenge overview: assessing the reliability of polarizable and non-polarizable methods for host-guest binding free energy calculations. ChemRxiv. https://doi.org/10.26434/chemrxiv.12768353.v1
Kendall MG (1938) A new measure of rank correlation. Biometrika 30:81–93
Pearlman DA, Charifson PS (2001) Are free energy calculations useful in practice? A comparison with rapid scoring functions for the p38 MAP kinase protein system. J Med Chem 44:3417–3423
Acknowledgements
This work is supported by China Postdoctoral Science Foundation. Dr. Zhaoxi Sun is supported by the PKU-Boya Postdoctoral Fellowship. We are grateful for many valuable and insightful comments on the performance of different charge schemes from the anonymous reviewers.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Huai, Z., Yang, H., Li, X. et al. SAMPL7 TrimerTrip host–guest binding affinities from extensive alchemical and end-point free energy calculations. J Comput Aided Mol Des 35, 117–129 (2021). https://doi.org/10.1007/s10822-020-00351-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10822-020-00351-9