Abstract
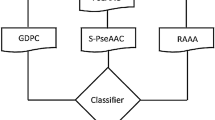
Cell-penetrating peptides (CPPs) are short length permeable proteins have emerged as drugs delivery tool of therapeutic agents including genetic materials and macromolecules into cells. Recently, CPP has become a hotspot avenue for life science research and paved a new way of disease treatment without harmful impact on cell viability due to nontoxic characteristic. Therefore, the correct identification of CPPs will provide hints for medical applications. Considering the shortcomings of traditional experimental CPPs identification, it is urgently needed to design intelligent predictor for accurate identification of CPPs for the large scale uncharacterized sequences. We develop a novel computational method, called TargetCPP, to discriminate CPPs from Non-CPPs with improved accuracy. In TargetCPP, first the peptide sequences are formulated with four distinct encoding methods i.e., composite protein sequence representation, composition transition and distribution, split amino acid composition, and information theory features. These dominant feature vectors were fused and applied intelligent minimum redundancy and maximum relevancy feature selection method to choose an optimal subset of features. Finally, the predictive model is learned through different classification algorithms on the optimized features. Among these classifiers, gradient boost decision tree algorithm achieved excellent performance throughout the experiments. Notably, the TargetCPP tool attained high prediction Accuracy of 93.54% and 88.28% using jackknife and independent test, respectively. Empirical outcomes prove the superiority and potency of proposed bioinformatics method over state-of-the-art methods. It is highly anticipated that the outcomes of this study will provide a strong background for large scale prediction of CPPs and instructive guidance in clinical therapy and medical applications.




Similar content being viewed by others
References
Chou K-C, Shen H-B (2007) Recent progress in protein subcellular location prediction. Anal Biochem 370(1):1–16
Krogh A et al (2001) Predicting transmembrane protein topology with a hidden Markov model: application to complete genomes. J Mol Biol 305(3):567–580
Lennernäs H et al (1996) Comparison between active and passive drug transport in human intestinal epithelial (Caco-2) cells in vitro and human jejunum in vivo. Int J Pharm 127(1):103–107
Ramsey JD, Flynn NH (2015) Cell-penetrating peptides transport therapeutics into cells. Pharmacol Ther 154:78–86
Khalili P et al (2006) A non–RGD-based integrin binding peptide (ATN-161) blocks breast cancer growth and metastasis in vivo. Mol Cancer Ther 5(9):2271–2280
Trabulo S et al (2013) Cell-penetrating peptides as nucleic acid delivery systems: from biophysics to biological applications. Curr Pharm Des 19(16):2895–2923
Gebbing M et al (2015) Gene therapeutic approaches to inhibit hepatitis B virus replication. World J Hepatol 7(2):150
Michiue H et al (2014) The acceleration of boron neutron capture therapy using multi-linked mercaptoundecahydrododecaborate (BSH) fused cell-penetrating peptide. Biomaterials 35(10):3396–3405
Frankel AD, Pabo CO (1988) Cellular uptake of the tat protein from human immunodeficiency virus. Cell 55(6):1189–1193
Green M, Loewenstein PM (1988) Autonomous functional domains of chemically synthesized human immunodeficiency virus tat trans-activator protein. Cell 55(6):1179–1188
Fonseca SB, Pereira MP, Kelley SO (2009) Recent advances in the use of cell-penetrating peptides for medical and biological applications. Adv Drug Deliv Rev 61(11):953–964
Nakase I et al (2011) Efficient intracellular delivery of nucleic acid pharmaceuticals using cell-penetrating peptides. Acc Chem Res 45(7):1132–1139
Lakshmanan M et al (2012) Rapid and efficient gene delivery into plant cells using designed peptide carriers. Biomacromol 14(1):10–16
Rüter C et al (2010) A newly identified bacterial cell-penetrating peptide that reduces the transcription of pro-inflammatory cytokines. J Cell Sci 123(13):2190–2198
Zhang D, Wang J, Xu D (2016) Cell-penetrating peptides as noninvasive transmembrane vectors for the development of novel multifunctional drug-delivery systems. J Control Release 229:130–139
Patel A, Cholkar K, Mitra AK (2014) Recent developments in protein and peptide parenteral delivery approaches. Therapeutic delivery 5(3):337–365
Soler M et al (2014) Identification of BP16 as a non-toxic cell-penetrating peptide with highly efficient drug delivery properties. Org Biomol Chem 12(10):1652–1663
Kato T et al (2016) Plasmid DNA delivery by arginine-rich cell-penetrating peptides containing unnatural amino acids. Bioorg Med Chem 24(12):2681–2687
Farkhani SM et al (2016) Enhanced cellular internalization of CdTe quantum dots mediated by arginine-and tryptophan-rich cell-penetrating peptides as efficient carriers. Artificial cells, nanomedicine, and biotechnology 44(6):1424–1428
Mäger I et al (2012) The role of endocytosis on the uptake kinetics of luciferin-conjugated cell-penetrating peptides. Biochim Biophys Acta (BBA) 1818(3):502–511
Zaro JL, Shen W-C (2015) Cationic and amphipathic cell-penetrating peptides (CPPs): Their structures and in vivo studies in drug delivery. Front Chem Sci Eng 9(4):407–427
Zhang R et al (2006) Molecular modeling studies of peptide drug candidates against SARS. Med Chem 2(3):309–314
Chou K-C et al (2006) Progress in computational approach to drug development against SARS. Curr Med Chem 13(27):3263–3270
Zhang Q, Gao H, He Q (2015) Taming cell penetrating peptides: never too old to teach old dogs new tricks. Mol Pharm 12(9):3105–3118
Yang Y et al (2015) Synergistic targeted delivery of payload into cancer cells using liposomes co-modified with photolabile-caged cell-penetrating peptides and targeting ligands. J Control Release 213:e128
Otvos L (2008) In Peptide-based drug design. Peptide-based drug design: here and now, Springer. pp 1–8.
Raucher D, Ryu JS (2015) Cell-penetrating peptides: strategies for anticancer treatment. Trends Mol Med 21(9):560–570
Simon MJ et al. (2010) Evaluation of the cell-penetrating peptide TAT as a trans-blood-brain barrier delivery vehicle. In: Bioengineering Conference, Proceedings of the 2010 IEEE 36th Annual Northeast. IEEE
Gao S et al (2011) An unusual cell penetrating peptide identified using a plasmid display-based functional selection platform. ACS Chem Biol 6(5):484–491
Lee JH et al (2012) Screening of cell-penetrating peptides using mRNA display. Biotechnol J 7(3):387–396
Hansen M, Kilk K, Langel Ü (2008) Predicting cell-penetrating peptides. Adv Drug Deliv Rev 60(4–5):572–579
Dobchev A et al (2010) Prediction of cell-penetrating peptides using artificial neural networks. Curr Comput-Aided Drug Des 6(2):79–89
Sanders WS et al (2011) Prediction of cell penetrating peptides by support vector machines. PLoS Comput Biol 7(7):e1002101
Gautam, A., et al., CPPsite: a curated database of cell penetrating peptides. Database, 2012. 2012.
Holton TA et al (2013) CPPpred: prediction of cell penetrating peptides. Bioinformatics 29(23):3094–3096
Chen L et al (2015) Prediction and analysis of cell-penetrating peptides using pseudo-amino acid composition and random forest models. Amino Acids 47(7):1485–1493
Tang H et al (2016) Prediction of cell-penetrating peptides with feature selection techniques. Biochem Biophys Res Commun 477(1):150–154
Diener C et al (2016) Effective design of multifunctional peptides by combining compatible functions. PLoS Comput Biol 12(4):e1004786
Wei L, Tang J, Zou Q (2017) SkipCPP-Pred: an improved and promising sequence-based predictor for predicting cell-penetrating peptides. BMC Genomics 18(7):1
Wei L et al (2017) CPPred-RF: a sequence-based predictor for identifying cell-penetrating peptides and their uptake efficiency. J Proteome Res 16(5):2044–2053
Zhang L et al (2015) An ensemble method to distinguish bacteriophage virion from non-virion proteins based on protein sequence characteristics. Int J Mol Sci 16(9):21734–21758
Zhang Y-N et al (2012) Predicting protein-ATP binding sites from primary sequence through fusing bi-profile sampling of multi-view features. BMC Bioinform 13(1):118
Han GS et al (2013) An ensemble method for predicting subnuclear localizations from primary protein structures. PLoS ONE 8(2):e57225
Kabir M et al (2018) Intelligent computational method for discrimination of anticancer peptides by incorporating sequential and evolutionary profiles information. Chemom Intell Lab Syst 182:158–165
Ali F et al (2018) DBPPred-PDSD: Machine learning approach for prediction of DNA-binding proteins using Discrete Wavelet Transform and optimized integrated features space. Chemom Intell Lab Syst 182:21–30
Li W, Godzik A (2006) Cd-hit: a fast program for clustering and comparing large sets of protein or nucleotide sequences. Bioinformatics 22(13):1658–1659
Wei Z-S et al (2015) A cascade random forests algorithm for predicting protein-protein interaction sites. IEEE Trans Nanobiosci 14(7):746–760
Hu J et al. (2019) TargetDBP: accurate DNA-binding protein prediction via sequence-based multi-view feature learning. IEEE/ACM transactions on computational biology and bioinformatics
Ahmad K, Waris M, Hayat M (2016) Prediction of protein submitochondrial locations by incorporating dipeptide composition into Chou’s general pseudo amino acid composition. J Membr Biol 249:1–12
Kurgan L et al (2009) CRYSTALP2: sequence-based protein crystallization propensity prediction. BMC Struct Biol 9(1):50
Golmohammadi SK et al. (2007) Classification of cell membrane proteins. in Frontiers in the Convergence of Bioscience and Information Technologies, 2007. FBIT 2007. IEEE
Xia X, Li W-H (1998) What amino acid properties affect protein evolution? J Mol Evol 47(5):557–564
Qiu W-R et al (2017) iKcr-PseEns: identify lysine crotonylation sites in histone proteins with pseudo components and ensemble classifier. Genomics 110:239–246
Hayat M, Khan A (2011) Predicting membrane protein types by fusing composite protein sequence features into pseudo amino acid composition. J Theor Biol 271(1):10–17
Chen W et al (2016) iACP: a sequence-based tool for identifying anticancer peptides. Oncotarget 7(13):16895–16909
Hayat M, Khan A, Yeasin M (2012) Prediction of membrane proteins using split amino acid and ensemble classification. Amino Acids 42(6):2447–2460
Singh O, Su EC-Y (2016) Prediction of HIV-1 protease cleavage site using a combination of sequence, structural, and physicochemical features. BMC Bioinform 17(17):478
Tang H, Chen W, Lin H (2016) Identification of immunoglobulins using Chou's pseudo amino acid composition with feature selection technique. Mol BioSyst 12(4):1269–1275
Eisenberg D et al (1984) Analysis of membrane and surface protein sequences with the hydrophobic moment plot. J Mol Biol 179(1):125–142
Karshikoff A, Nilsson L, Ladenstein R (2015) Rigidity versus flexibility: the dilemma of understanding protein thermal stability. FEBS J 282(20):3899–3917
Huber R (1987) Flexibility and rigidity, requirements for the function of proteins and protein pigment complexes. Portland Press Limited, London
Kedarisetti KD, Kurgan L, Dick S (2006) Classifier ensembles for protein structural class prediction with varying homology. Biochem Biophys Res Commun 348(3):981–988
Dubchak I et al (1995) Prediction of protein folding class using global description of amino acid sequence. Proc Natl Acad Sci 92(19):8700–8704
Bhadra P et al (2018) AmPEP: Sequence-based prediction of antimicrobial peptides using distribution patterns of amino acid properties and random forest. Sci Rep 8(1):1697
Nair R, Rost B (2005) Mimicking cellular sorting improves prediction of subcellular localization. J Mol Biol 348(1):85–100
Cai C et al (2003) SVM-Prot: web-based support vector machine software for functional classification of a protein from its primary sequence. Nucleic Acids Res 31(13):3692–3697
Jones A. et al. (2015) RF-Phos: random forest-based prediction of phosphorylation sites. in 2015 IEEE international conference on bioinformatics and biomedicine (BIBM). IEEE
Zhang X, Liu S (2016) RBPPred: predicting RNA-binding proteins from sequence using SVM. Bioinformatics 33(6):854–862
Govindan G, AS Nair (2011) Composition, transition and distribution (CTD)—a dynamic feature for predictions based on hierarchical structure of cellular sorting. in India Conference (INDICON), 2011 Annual IEEE. IEEE
Capra JA, Singh M (2007) Predicting functionally important residues from sequence conservation. Bioinformatics 23(15):1875–1882
Wei L. et al. (2017) Fast prediction of protein methylation sites using a sequence-based feature selection technique. IEEE/ACM Transactions On Computational Biology and Bioinformatics
Ismail HD, Newman RH (2016) RF-Hydroxysite: a random forest based predictor for hydroxylation sites. Mol BioSyst 12(8):2427–2435
Arif M, Hayat M, Jan Z (2018) iMem-2LSAAC: a two-level model for discrimination of membrane proteins and their types by extending the notion of SAAC into Chou's pseudo amino acid composition. J Theor Biol 442:11–21
Khan ZU, Hayat M, Khan MA (2015) Discrimination of acidic and alkaline enzyme using Chou’s pseudo amino acid composition in conjunction with probabilistic neural network model. J Theor Biol 365:197–203
Ahmad S, Kabir M, Hayat M (2015) Identification of Heat Shock Protein families and J-protein types by incorporating Dipeptide Composition into Chou's general PseAAC. Comput Methods Programs Biomed 122(2):165–174
Mirza MT et al (2013) MitProt-Pred: Predicting mitochondrial proteins of Plasmodium falciparum parasite using diverse physiochemical properties and ensemble classification. Comput Biol Med 43(10):1502–1511
Wang S et al (2016) Analysis and identification of aptamer-compound interactions with a maximum relevance minimum redundancy and nearest neighbor algorithm. BioMed Res Int 102:1567–1577
Cheng F et al (2012) Prediction of chemical-protein interactions network with weighted network-based inference method. PLoS ONE 7(7):e41064
Iqbal M, Hayat M (2016) “iSS-Hyb-mRMR”: identification of splicing sites using hybrid space of pseudo trinucleotide and pseudo tetranucleotide composition. Comput Methods Programs Biomed 128:1–11
Win TS et al (2018) PAAP: A web server for predicting antihypertensive activity of peptides. Future Med Chem 10(15):1749–1767
Peng H, Long F, Ding C (2005) Feature selection based on mutual information criteria of max-dependency, max-relevance, and min-redundancy. IEEE Trans Pattern Anal Mach Intell 27(8):1226–1238
Zhang Y, Ding C, Li T (2008) Gene selection algorithm by combining reliefF and mRMR. BMC genomics 9(2):S27
Xu Y et al (2014) Identification of thyroid carcinoma related genes with mRMR and shortest path approaches. PLoS ONE 9(4):e94022
Li Z et al (2014) The prediction of methylation states in human DNA sequences based on hexanucleotide composition and feature selection. Anal Methods 6(6):1897–1904
Cai Y et al (2012) Prediction of lysine ubiquitination with mRMR feature selection and analysis. Amino Acids 42(4):1387–1395
Ding H et al (2013) Prediction of Golgi-resident protein types by using feature selection technique. Chemom Intell Lab Syst 124:9–13
Peng H, Long F, Ding C (2005) Feature selection based on mutual information: criteria of max-dependency, max-relevance, and min-redundancy. IEEE Trans Pattern Anal Mach Intell 8:1226–1238
Chen T, C. Guestrin (2016) Xgboost: A scalable tree boosting system. In: Proceedings of the 22nd acm sigkdd international conference on knowledge discovery and data mining. ACM
Pal M, Mather PM (2003) An assessment of the effectiveness of decision tree methods for land cover classification. Remote Sens Environ 86(4):554–565
Feng PM et al (2013) Naive Bayes classifier with feature selection to identify phage virion proteins. Computat Math Methods Med. https://doi.org/10.1155/2013/530696
Friedman JH (2001) Greedy function approximation: a gradient boosting machine. Ann Stat 29:1189–1232
Chen F et al (2017) Prediction of luciferase inhibitors by the high-performance MIEC-GBDT approach based on interaction energetic patterns. Phys Chem Chem Phys 19(15):10163–10176
Liao Z et al (2016) In silico prediction of gamma-aminobutyric acid type-A receptors using novel machine-learning-based SVM and GBDT approaches. BioMed Res Int. https://doi.org/10.1155/2016/2375268
Zhou C et al (2017) Multi-scale encoding of amino acid sequences for predicting protein interactions using gradient boosting decision tree. PLoS ONE 12(8):e0181426
Rawi R et al (2017) PaRSnIP: sequence-based protein solubility prediction using gradient boosting machine. Bioinformatics 34(7):1092–1098
Arif M et al (2020) Pred-BVP-Unb: fast prediction of bacteriophage Virion proteins using un-biased multi-perspective properties with recursive feature elimination. Genomics 112(2):1565–1574
Wang Y et al (2017) Improved detection of DNA-binding proteins via compression technology on PSSM information. PLoS ONE 12(9):e185587
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Nos. 61772273 and 61373062) and Fundamental Research Funds for the Central Universities (No. 30918011104).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that they have no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Arif, M., Ahmad, S., Ali, F. et al. TargetCPP: accurate prediction of cell-penetrating peptides from optimized multi-scale features using gradient boost decision tree. J Comput Aided Mol Des 34, 841–856 (2020). https://doi.org/10.1007/s10822-020-00307-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10822-020-00307-z