Abstract
Purpose
To present the developed preimplantation genetic testing (PGT) for spinocerebellar ataxia type 1 (SCA1) and the outcomes of IVF with PGT.
Methods
PGT was performed for two unrelated couples from the Republic of Sakha (Yakutia) with the risk of SCA1 in one spouse. We have developed a system for PGT of a monogenic disease (PGT-M) for SCA1, which includes the analysis of a panel of 11 polymorphic STR markers linked to the ATXN1 gene and a pathogenic variant of the ATXN1 gene using nested PCR and fragment analysis. IVF/ICSI programs were performed according to standard protocols. Multiple displacement amplification (MDA) was used for whole genome amplification (WGA) and array comparative genomic hybridization (aCGH) for aneuploidy testing (PGT-A).
Results
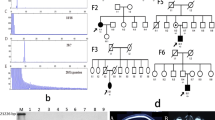
Eight STRs were informative for the first couple and ten for the second. Similarity of the haplotypes carrying pathogenic variants of the ATXN1 gene was noted. In the first case, during IVF/ICSI-PGT, three embryos reached the blastocyst stage and were biopsied. One embryo was diagnosed as normal by maternal STR haplotype and the ATXN1 allele. PGT-A revealed euploidy. The embryo transfer resulted in a singleton pregnancy, and a healthy boy was born. Postnatal diagnosis confirmed normal ATXN1. In the second case, two blastocysts were biopsied. Both were diagnosed as normal by PGT-M, but PGT-A revealed aneuploidy.
Conclusion
Birth of a healthy child after PGT for SCA1 was the first case of successful preimplantation prevention of SCA1 for the Yakut couple and the first case of successful PGT for SCA1 in Russia.


Similar content being viewed by others
References
Opal P, Ashizawa T. Spinocerebellar Ataxia Type 1. 1998 Oct 1 [Updated 2023 Feb 2]. In: Adam MP, Mirzaa GM, Pagon RA, et al., editors. GeneReviews® [Internet]. Seattle: University of Washington; 1993–2023. Bookshelf URL: https://www.ncbi.nlm.nih.gov/books/.
Zoghbi HY, Orr HT. Pathogenic mechanisms of a polyglutamine-mediated neurodegenerative disease, spinocerebellar ataxia type 1. J Biol Chem. 2009;284(12):7425–9. https://doi.org/10.1074/jbc.R800041200.
Illarioshkin SN, Slominsky PA, Ovchinnikov IV, et al. Spinocerebellar ataxia type 1 in Russia. J Neurol. 1996;243(7):506–10. https://doi.org/10.1007/BF00886871.
Orr HT. Cell biology of spinocerebellar ataxia. J Cell Biol. 2012;197(2):167–77. https://doi.org/10.1083/jcb.201105092.
Srinivasan SR, Shakkottai VG. Moving towards therapy in SCA1: insights from molecular mechanisms, identification of novel targets, and planning for human trials. Neurotherapeutics. 2019;16(4):999–1008. https://doi.org/10.1007/s13311-019-00763-y.
Tejwani L, Lim J. Pathogenic mechanisms underlying spinocerebellar ataxia type 1. Cell Mol Life Sci. 2020;77(20):4015–29. https://doi.org/10.1007/s00018-020-03520-z.
Goldfarb LG, Chumakov MP, Petrov PA, et al. Olivopontocerebellar atrophy in a large Iakut kinship in eastern Siberia. Neurology. 1989;39(11):1527–30. https://doi.org/10.1212/wnl.39.11.1527.
Fedorov AI, Sukhomyasova AL, Golikova PI, et al. The prevalence of type 1 spinocerebellar ataxia in Yakutia: current status. Medical genetics. 2020;19(7):29–30. https://doi.org/10.25557/2073-7998.2020.07.29-30. (in Russian).
Platonov FA, Tyryshkin K, Tikhonov DG, et al. Genetic fitness and selection intensity in a population affected with high-incidence spinocerebellar ataxia type 1. Neurogenetics. 2016;17(3):179–85. https://doi.org/10.1007/s10048-016-0481-5.
Koneva LA, Konev AV, Kucher AN. Simulation of the distribution of spinocerebellar ataxia type 1 in Yakut populations: model parameters and results of simulation. Genetika. 2010;46(7):990–9 (Russian. PMID: 20795504).
Carvalho F, Moutou C, ESHRE PGT-M Working Group, et al. ESHRE PGT Consortium good practice recommendations for the detection of monogenic disorders. Hum Reprod Open. 2020;2020(3):hoaa018. https://doi.org/10.1093/hropen/hoaa018.
Liao CH, Chang MY, Ma GC, et al. Preimplantation genetic diagnosis of neurodegenerative diseases: review of methodologies and report of our experience as a regional reference laboratory. Diagnostics (Basel). 2019;9(2):44. https://doi.org/10.3390/diagnostics9020044.
Verlinsky Y, Kuliev A. An atlas of preimplantation genetic diagnosis: an illustrated textbook & reference for clinicians, 2nd edn. CRC Press; 2004. https://doi.org/10.1201/b14655.
Spinella F, Bronet F, Carvalho F, et al. ESHRE PGT Consortium data collection XXI: PGT analyses in 2018. Hum Reprod Open. 2023;2023(2):hoad010. https://doi.org/10.1093/hropen/hoad010.
Drüsedau M, Dreesen JC, De Die-Smulders C, et al. Preimplantation genetic diagnosis of spinocerebellar ataxia 3 by (CAG)(n) repeat detection. Mol Hum Reprod. 2004;10(1):71–5. https://doi.org/10.1093/molehr/gah008.
Moutou C, Nicod JC, Gardes N, Viville S. Birth after pre-implantation genetic diagnosis (PGD) of spinocerebellar ataxia 2 (Sca2). Prenat Diagn. 2008;28(2):126–30. https://doi.org/10.1002/pd.1909.
Tur-Kaspa I, Jeelani R, Doraiswamy PM. Preimplantation genetic diagnosis for inherited neurological disorders. Nat Rev Neurol. 2014;10(7):417–24. https://doi.org/10.1038/nrneurol.2014.84. (Epub 2014 May 27 PMID: 24866878).
Shi D, Xu J, Niu W, et al. Live births following preimplantation genetic testing for dynamic mutation diseases by karyomapping: a report of three cases. J Assist Reprod Genet. 2020;37(3):539–48. https://doi.org/10.1007/s10815-020-01718-5.
Cahn S, Rosen A, Wilmot G. Spinocerebellar ataxia patient perceptions regarding reproductive options. Mov Disord Clin Pract. 2019;7(1):37–44. https://doi.org/10.1002/mdc3.12859.
Lunkes A, Goldfarb LG, Platonov FA, et al. Autosomal dominant spinocerebellar ataxia (SCA) in a Siberian founder population: assignment to the SCA1 locus. Exp Neurol. 1994;126(2):310–2. https://doi.org/10.1006/exnr.1994.1070.
Assisted reproductive technology and artificial insemination. Clinical guidelines (treatment protocol). Ministry of Health of the Russian Federation March 05, 2019 No. 15–4/and/2–1908. 169p. [in Russian]. http://www.rahr.ru/pech_mat_norm.php.
WHO laboratory manual for the examination and processing of human semen, sixth edition. Geneva: World Health Organization; 2021. Licence: CC BY-NC-SA 3.0 IGO. https://www.who.int/publications/i/item/9789240030787.
Gardner DK, Schoolcraft WB. In vitro culture of human blastocysts. In: Jansen R, Mortimer D, editors. Toward Reproductive Certainty: Fertility and Genetics Beyond 1999. UK: Parthenon Publishing London; 1999. p. 378–88.
Coonen E, Rubio C, ESHRE PGT-SR/PGT-A Working Group, et al. ESHRE PGT Consortium good practice recommendations for the detection of structural and numerical chromosomal aberrations. Hum Reprod Open. 2020;3:hoaa017. https://doi.org/10.1093/hropen/hoaa017.
Lai YC, Wang WC, Yang JJ, Li SY. Expansion of CAG repeats in the spinocerebellar ataxia type 1 (SCA1) gene in idiopathic oligozoospermia patients. J Assist Reprod Genet. 2009;26(5):257–61. https://doi.org/10.1007/s10815-009-9325-7.
Sequeiros J, Seneca S, Martindale J. Consensus and controversies in best practices for molecular genetic testing of spinocerebellar ataxias. Eur J Hum Genet. 2010;18(11):1188–95. https://doi.org/10.1038/ejhg.2010.10.
Ma S, Liao J, Zhang S, et al. Exploring the efficacy and beneficial population of preimplantation genetic testing for aneuploidy start from the oocyte retrieval cycle: a real-world study. J Transl Med. 2023;21(1):779. https://doi.org/10.1186/s12967-023-04641-2.
Morales C. Current applications and controversies in preimplantation genetic testing for aneuploidies (PGT-A) in in vitro fertilization. Reprod Sci. 2024;31:66–80. https://doi.org/10.1007/s43032-023-01301-0.
Kononova SK, Sidorova OG, Fedorova SA, et al. Bioethical issues of preventing hereditary diseases with late onset in the Sakha Republic (Yakutia). Int J Circumpolar Health. 2014;24(73):25062. https://doi.org/10.3402/ijch.v73.25062.
Ethics Committee of the American Society for Reproductive Medicine. Use of preimplantation genetic testing for monogenic defects (PGT-M) for adult-onset conditions: an Ethics Committee opinion. Fertil Steril. 2018 Jun;109(6):989–992. https://doi.org/10.1016/j.fertnstert.2018.04.003.
Gonzalez-Pena V, Natarajan S, Xia Y, et al. Accurate genomic variant detection in single cells with primary template-directed amplification. Proc Natl Acad Sci U S A. 2021;118(24):e2024176118. https://doi.org/10.1073/pnas.2024176118.
Wakisaka A, Sasaki H, Takada A, et al. Spinocerebellar ataxia 1 (SCA1) in the Japanese in Hokkaido may derive from a single common ancestry. J Med Genet. 1995;32(8):590–2. https://doi.org/10.1136/jmg.32.8.590.
Krysa W, Sulek A, Rakowicz M, et al. High relative frequency of SCA1 in Poland reflecting a potential founder effect. Neurol Sci. 2016;37(8):1319–25. https://doi.org/10.1007/s10072-016-2594-x. (Epub 2016 May 19).
Funding
This work was supported by the Ministry of Science and Higher Education of the Russian Federation (the Federal scientific-technical program for genetic technologies development for 2019–2027, agreement. 075–15-2021–1061, RF 193021X0029).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Consent for publication
The patients in this case report provided their consent for publication.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 18.5 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Soloveva, E.V., Skleimova, M.M., Minaycheva, L.I. et al. PGT-M for spinocerebellar ataxia type 1: development of a STR panel and a report of two clinical cases. J Assist Reprod Genet (2024). https://doi.org/10.1007/s10815-024-03105-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10815-024-03105-w