Abstract
Purpose
While stimulation of women prior to assisted reproduction is associated with increased success rates, the total biological pregnancy potential per stimulation cycle is rarely assessed.
Methods
Retrospective sequential cohort study of the cumulative live birth rate in 1148 first IVF/ICSI-cycles and 5-year follow up of frozen embryo replacement (FER) cycles were used. Oocyte number, number of embryos transferred, and cryopreserved/thawed and transferred embryos in a FER cycle were registered for all patients. Children per oocyte and per transferred embryo and percentage of cycles with births were calculated.
Results
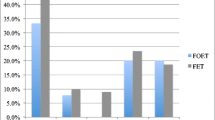
We obtained 9529 oocytes. Embryos (2507) were transferred in either fresh or FER cycles, resulting in 422 births and 474 live born children. Median age of the women was 32.5 years (range 20–41.5 years). In total, 34.3 % of all cycles ended with a live birth while in 65.7 % of the cycles, no oocytes were capable of developing into a child. The average number of oocytes needed per live born child after transfer of fresh and thawed embryos was 20 as only 5.0 % of oocytes aspirated in the first IVF/ICSI cycle had the competence to develop into a child.
Conclusions
In our setting, overall 5.0 % of the oocytes in a first cycle were biologically competent and in around 2/3 of all cycles, none of the oocytes had the potential to result in the birth of a child.
Similar content being viewed by others
References
Inge GB, Brinsden PR, Elder KT. Oocyte number per live birth in IVF: were Steptoe and Edwards less wasteful? Hum Reprod. 2005;2:588–92.
Patrizio P, Sakkas D. From oocyte to baby: a clinical evaluation of the biology efficiency of in vitro fertilization. Fertil Steril. 2009;91:1061–6.
Martin JR, Bromer JG, Sakkas D, Patrizio P. Live babies born per oocyte retrieved in a subpopulation of oocyte donors with repetitive reproductive success. Fertil Steril. 2010;94:2064–8.
Gnoth C, Maxrath B, Skonieczny T, Friol K, Godehardt E, Tigges J. Final ART success rates: a 10 years survey. Hum Reprod. 2011;26:2239–46.
Stoop D, Ermini B, Polyzos NP, Haentjens P, De Vos M, Verheyen G, et al. Reproductive potential of a metaphase II oocyte retrieved after ovarian stimulation: an analysis of 23,354 ICSI cycles. Hum Reprod. 2012;27:2030–5.
Oktay K, Cil AP, Bang H. Efficiency of oocyte cryopreservation: a meta-analysis. Fertil Steril. 2006;86:70–80.
ASRM. The practice committee of the society for assisted reproductive technology and the practice committee of the American society for reproductive medicine. Essential elements of informed consent for elective oocyte cryopreservation: a practice committee opinion. Fertil Steril. 2008;90:S134–5.
Goldman KN, Noyes NL, Knopman JM, McCaffrey C, Grifo JA. Oocyte efficiency: does live birth rate differ when analyzing cryopreserved and fresh oocytes on a per-oocyte basis? Fertil Steril. 2013;100:712–7.
Patrizio P, Bianchi V, Lalioti MD, Gerasimova T, Sakkas D. High rate of biological loss in assisted reproduction: it is in the seed, not in the soil. Reprod Biomed Online. 2007;14:92–5.
Ziebe SK, Petersen K, Lindenberg S, Andersen AG, Gabrielsen A, Andersen A. Embryo morphology or cleavage rate: how to select the best embryos for transfer after in vitro fertilization. Hum Reprod. 1997;12:1545–9.
Ziebe S, Bech B, Petersen K, Mikkelsen AL, Gabrielsen A, Nyboe AA. Resumption of mitosis during post-thaw culture: a key parameter in selecting the right embryos for transfer. Hum Reprod. 1998;13:178–81.
Hnida C, Engenheiro E, Ziebe S. Computer controlled multi-level morphometric analysis of blastomere size as biomarker of fragmentation and multinuclearity in human embryos. Hum Reprod. 2004;19:288–93.
Hnida C, Ziebe S. Total cytoplasmic volume as biomarker of fragmentation in human embryos. J Ass Reprod Gen. 2004;21:335–40.
Hnida C, Agerholm I, Ziebe S. Traditional detection versus computer-controlled multilevel analysis of nuclear structures from donated human embryos. Hum Reprod. 2005;20:665–7.
Lemmen JG, Agerholm I, Ziebe S. Timelapse recordings of IVF/ICSI fertilised oocytes. RBM Online. 2008;17:385–91.
Montag M, Toth B, Strowitzki T. New approaches to embryo selection. Reprod Biomed Online. 2013;27:539–46.
Ajduk A, Zernicka-Goetz M. Quality control of embryo development. Mol Asp Med. 2013;34:903–18.
Agerholm IE, Kølvraa S, Cruger DG, Berg C, Bruun-Petersen G, Ziebe S. Resumption of mitosis in frozen-thawed embryos is not related to the chromosomal constitution. Fertil Steril. 2008;90:1649–55.
Rienzi L, Cobo A, Paffoni A, Scarduelli C, Capalbo A, Vajta G, et al. Consistent and predictable delivery rates after oocyte vitrification: an observational longitudinal cohort multicentric study. Hum Reprod. 2012;27:1606–12.
Chang C-C, Elliott TA, Wright G, Shapiro DB, Toledo Nagy ZP. Prospective controlled study to evaluate laboratory and clinical outcomes of oocyte vitrification obtained in in vitro fertilization patients aged 30 to 39 years. Fertil Steril. 2013;99:1891–7.
Kim TJ, Laufer LR, Hong SW. Vitrification of oocytes produces high pregnancy rates when carried out in fertile women. Fertil Steril. 2010;93(2):467–74.
Cobo 2014, Oral presentation no O259. At ESHRE 2014 Munich
Edwards RG. Test-tube babies. Nature. 1981;293:253–6.
Pelinck MJ, Hoek A, Simons AH, Heineman MJ. Efficacy of natural cycle IVF: a review of the literature. Hum Reprod Update. 2002;8:129–39.
Meniru GI, Craft IL. Utilization of retrieved oocytes as an index of the efficiency of superovulation strategies for in-vitro fertilization treatment. Hum Reprod. 1997;10:2129–32.
Zinaman MJ, Clegg ED, Brown CC, O’Connor J, Selevan SG. Estimates of human fertility and pregnancy loss. Fertil Steril. 1996;65:503–9.
Franasiak JM, Forman EJ, Hong KH, Werner MD, Upham KM, Treff NR, et al. The nature of aneuploidy with increasing age of the female partner: a review of 15,169 consecutive trophectoderm biopsies evaluated with comprehensive chromosomal screening. Fertil Steril. 2014;101(3):656–63.
Baart EB, Martini E, Eijkemans MJ, Van Opstal D, Beckers NG, Verhoeff A, et al. Milder ovarian stimulation for in-vitro fertilization reduces aneuploidy in the human preimplantation embryo: a randomized controlled trial. Hum Reprod. 2007;22:980–8.
McAvey B, Zapantis A, Jindal SK, Lieman HJ, Polotsky AJ. How many eggs are needed to produce an assisted reproductive technology baby: is more always better? Fertil Steril. 2011;96:332–25.
Author information
Authors and Affiliations
Corresponding author
Additional information
Capsule In our setting, overall 5.0 % of the oocytes in a first cycle were biologically competent and in around 2/3 of all cycles, none of the oocytes had the potential to result in the birth of a child.
Rights and permissions
About this article
Cite this article
Lemmen, J.G., Rodríguez, N.M., Andreasen, L.D. et al. The total pregnancy potential per oocyte aspiration after assisted reproduction—in how many cycles are biologically competent oocytes available?. J Assist Reprod Genet 33, 849–854 (2016). https://doi.org/10.1007/s10815-016-0707-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-016-0707-3