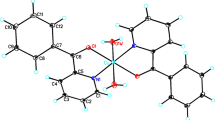
The X-ray absorption fine structure (XAFS) spectra at the K-edge of the copper complexes Cu(PDC)(Mim)3 H2O (1) and Cu(PDC)2(EA)2H2O (2) (where PDC – Pyridine-2,3-dicarboxylic acid, Mim – 2-methylimidazole, and EA – ethyl acetate) have been investigated. The experimental extended X-ray absorption fine structure data of complex 1 have been analyzed by fitting the theoretical model generated from its own crystallographic data. The crystallographic data for complex 2 are not available. It has been found by comparing the intensity of the pre-edge peaks and X-ray absorption near edge structure features of complexes 1 and 2 that both complexes possess square pyramidal geometry around the copper centers and thus complex 2 is analogous to complex 1. Hence, the theoretical model generated for complex 1 has been fitted to the experimental EXAFS data of complex 2 to determine the structural parameters of complex 2. The coordination geometry of both complexes has been depicted. Further, the chemical shifts have been used to determine the oxidation state as well as to estimate the effective nuclear charge on the copper atom.
Similar content being viewed by others
References
F. Semerci, O. Z. Yesilel, and E. Sahin, J. Inorg. Organomet. Polym., 20, 334 (2010).
A. Gaur, B. D. Shrivastava, K. Shrivastava, and J. Prasad, J. Appl. Phys., 113, 073701 (2013).
A. Gaur, B. D. Shrivastava, and H. L. Nigam, Proc. Indian Natl. Sci. Acad., 79, 921 (2013).
Abhijeet Gaur, D Ah. Dar, B. D. Shrivastava, S. N. Jha, and D. Bhattacharyya, Indian J. Phys., 89, No. 5 (2015); doi 10.1007/s12648-014-0610-7.
B. Ravel and M. Newville, J. Synchrotron Rad., 12, 537 (2005).
S. D. Kelly, D. Hesterberg, and B. Ravel, In "Methods of Soil Analysis", Eds. A. L. Ulery and L. R. Drees, Soil Science Society of America, Madison, USA, 387 (2008).
S. Calvin, XAFS for Everyone, CRC Press, New York, 13 (2013)
A. M. Amado and P. J. A.Ribeiro-Claro, Inorg. Biochem., 98, 561 (2004).
M. Giorgetti, M. Pellei, G. G. Lobbia, and C. Santini, J. Phys. Conf. Ser., 190, 012146 (2009).
A. Manceau and A. Matynia, Geochim. Cosmochim. Acta, 74, 2556 (2010).
L. S. Kau, D. J. Spira-Solomon, J. E. Penner-Hahn, K. O. Hodgson, and E. I. Solomon, J. Am. Chem. Soc., 109, 6433 (1987).
J. L. Fulton, M. M. Hoffman, J. G. Darab, B. J. Palmer, and E. A. Stern, J. Phys. Chem. A, 104, 11651 (2000).
A. K. Nigam and M. K. Gupta, J. Phys. F: Met. Phys., 4, 1084 (1974).
R. G. Shulman, Y. Yafet, P. Eisenberger, and W. E. Blumberg, Proc. Natl. Acad. Sci. USA, 73, 1384 (1976).
J. E. Penner-Hahn, R. A. Scott, K. O. Hodgson, S. Doniach, S. R. Desjardins, and E. I. Solomon, Chem. Phys. Lett., 88, 595 (1982).
A. L. Roe, D. J. Schneider, R. J. Mayer, J. W. Pyrz, J. Widom, and L. J. Que, J. Am. Chem. Soc., 106, 1676 (1984).
I. J. Pickering and G. N. George, Inorg. Chem., 34, 3142 (1995).
S. Della Longa, A. Bianconi, L. Palladino, B. Simonelli, A. Congiu Castellano, E. Borghi, M. Barteri, M. Beltramini, G. P. Rocco, and B. Salvato, Biophys. J., 65, 2680 (1993).
C. Santini, M. Pellei, G. G. Lobbia, A. Cingolani, R. Spagna, and M. Camalli, Inorg. Chem. Commun., 5, 430 (2002).
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Zhurnal Prikladnoi Spektroskopii, Vol. 82, No. 2, pp. 278–284, March–April, 2015.
Rights and permissions
About this article
Cite this article
Dar, D.A., Gaur, A., Soni, B. et al. X-Ray Absorption Fine Structure Investigation of Copper(II) Mixed Ligand Complexes with Pyridinedicarboxylic Acid as Primary Ligand. J Appl Spectrosc 82, 272–277 (2015). https://doi.org/10.1007/s10812-015-0097-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10812-015-0097-9