Abstract
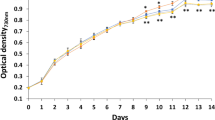
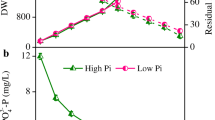
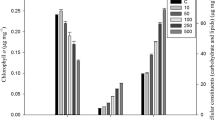
Synechocystis sp. PCC 7338 (hereafter referred to as Synechocystis 7338) is a marine cyanobacterium that has the potential to produce photosynthetic pigments. In this study, we investigated the effects of various NaCl concentrations (0, 0.4, 0.8, and 1.2 M) on cell growth, photosynthetic pigments, and metabolites and intact lipid species profiles in Synechocystis 7338. The overall growth pattern of Synechocystis 7338 was similar under 0, 0.4, and 0.8 M NaCl conditions. Cell growth was retarded after reaching the exponential phase under 1.2 M NaCl; however, a similar growth pattern was observed after the exponential phase under 0.4 M NaCl (control group). The highest production of chlorophyll a (4.18 mg L−1), allophycocyanin (4.08 mg L−1), and phycoerythrin (1.70 mg L−1) were achieved under 1.2 M NaCl conditions. Altered metabolic and lipidomic profiles were observed at different NaCl conditions; significantly increased relative yields of glucosylglycerol, one diacylglyceryltrimethylhomoserine, one monogalactosyldiacylglycerol, and four phosphatidylglycerol species were observed under 1.2 M NaCl conditions using gas chromatography–mass spectrometry and direct infusion–mass spectrometry analyses. In addition, it was revealed that the photosynthetic activity recovered under 1.2 M NaCl conditions in long-term culture. Hydrogen peroxide content significantly increased under 1.2 M NaCl conditions. It is believed that glutathione content also significantly increased under high salinity conditions to retain the normal functioning of Synechocystis 7338. These results indicate that high salinity conditions for Synechocystis 7338 culture could be used for the large-scale production of chlorophyll a, allophycocyanin, phycoerythrin, and other bioactive metabolites.





Similar content being viewed by others
References
Allakhverdiev SI, Murata N (2008) Salt stress inhibits photosystems II and I in cyanobacteria. Photosynth Res 98:529–539
Allakhverdiev SI, Kinoshita M, Inaba M, Suzuki I, Murata N (2001) Unsaturated fatty acids in membrane lipids protect the photosynthetic machinery against salt-induced damage in Synechococcus. Plant Physiol 125:1842–1853
Bräucker R, Cogoli A, Hemmersbach R (2002) Graviperception and graviresponse at the cellular level. In: Horneck G, Baumstak-Khan C (eds) Astrobiology. Springer, Berlin, pp 287–296
Cid A, Herrero C, Torres E, Abalde J (1995) Copper toxicity on the marine microalga Phaeodactylum tricornutum: effects on photosynthesis and related parameters. Aquat Toxicol 31:165–174
De Marsac NT, Houmard J (1988) Complementary chromatic adaptation: physiological conditions and action spectra. In: Packer L, Glazer AN (eds) Methods in enzymology. Academic Press, Cambridge, pp 318–328
Dittmann E, Fewer DP, Neilan BA (2013) Cyanobacterial toxins: biosynthetic routes and evolutionary roots. FEMS Microbiol Rev 37:23–43
Eriksson L, Johansson E, Kettaneh-Wold N et al (2006) PLS. In: Eriksson L (ed) Multi-and megavariate data analysis. Umetrics AB, Umeå, pp 63–101
Ferjani A, Mustardy L, Sulpice R, Marin K, Suzuki I, Hagemann M, Murata N (2003) Glucosylglycerol, a compatible solute, sustains cell division under salt stress. Plant Physiol 131:1628–1637
Geada P, Gkelis S, Teixeira J, Vasconcelos V, Vicente AA, Fernandes B (2017) Cyanobacterial toxins as a high value-added product. In: Gonzalez-Fernandez C, Munoz R (eds) Microalgae-based biofuels and bioproducts: from feedstock cultivation to end-products. Woodhead Publishing, Sawston, pp 401–428
Guskov A, Kern J, Gabdulkhakov A, Broser M, Zouni A, Saenger W (2009) Cyanobacterial photosystem II at 2.9-Å resolution and the role of quinones, lipids, channels and chloride. Nat Struct Mol Biol 16:334–342
Hagemann M (2011) Molecular biology of cyanobacterial salt acclimation. FEMS Microbiol Rev 35:87–123
Haque F, Banayan S, Yee J, Chiang YW (2017) Extraction and applications of cyanotoxins and other cyanobacterial secondary metabolites. Chemosphere 183:164–175
Herrmann AJ, Gehringer MM (2019) An investigation into the effects of increasing salinity on photosynthesis in freshwater unicellular cyanobacteria during the late Archaean. Geobiology 17:343–359
Hong SJ, Lee CG (2008) Statistical optimization of culture media for production of phycobiliprotein by Synechocystis sp. PCC 6701. Biotechnol Bioprocess Eng 13:491–498
Hufiejt ME, Tremolieres A, Pineau B, Lang JK, Hatheway J, Packer L (1990) Changes in membrane lipid composition during saline growth of the fresh water cyanobacterium Synechococcus 6311. Plant Physiol 94:1512–1521
Ilieva V, Kondeva-Burdina M, Georgieva T, Pavlova V (2019) Toxicity of cyanobacteria. Organotropy of cyanotoxins and toxicodynamics of cyanotoxins by species. Pharmacia 66:91–97
Joset F, Jeanjean R, Hagemann M (1996) Dynamics of the response of cyanobacteria to salt stress: deciphering the molecular events. Physiol Plant 96:738–744
Kaneko T, Sato S, Kotani H, Tanaka A, Asamizu E, Nakamura Y, Miyajima N, Hirosawa M, Sugiura M, Sasamoto S, Kimura T, Hosouchi T, Matsuno A, Muraki A, Nakazaki N, Naruo K, Okumura S, Shimpo S, Takeuchi C, Wada T, Watanabe A, Yamada M, Yasuda M, Tabata S (1996) Sequence analysis of the genome of the unicellular cyanobacterium Synechocystis sp. strain PCC6803. II. Sequence determination of the entire genome and assignment of potential protein-coding regions. DNA Res 3:109–136
Kanesaki Y, Suzuki I, Allakhverdiev SI, Mikami K, Murata N (2002) Salt stress and hyperosmotic stress regulate the expression of different sets of genes in Synechocystis sp. PCC 6803. Biochem Biophys Res Commun 290:339–348
Kim ZH, Park H, Ryu YJ, Shin DW, Hong SJ, Tran HL, Lim SM, Lee GC (2015) Algal biomass and biodiesel production by utilizing the nutrients dissolved in seawater using semi-permeable membrane photobioreactors. J Appl Phycol 27:1763–1773
Kim SH, Lim SR, Hong SJ, Cho BK, Lee H, Lee CG, Choi HK (2016) Effect of ethephon as an ethylene-releasing compound on the metabolic profile of Chlorella vulgaris. J Agric Food Chem 64:4807–4816
Kim HY, Lee H, Kim S, Jin H, Bae J, Choi H-K (2017a) Discovery of potential biomarkers in human melanoma cells with different metastatic potential by metabolic and lipidomic profiling. Sci Rep 7:1–14
Kim JY, Kim HY, Jeon JY, Kim DM, Zhou Y, Lee JS, Lee H, Choi H-K (2017b) Effects of coronatine elicitation on growth and metabolic profiles of Lemna paucicostata culture. PLoS One 12:e0187622
Kumar J, Singh D, Tyagi MB, Kumar A (2019) Cyanobacteria: applications in biotechnology. In: Mishra AK, Tiwari DN, Rai AN (eds) Cyanobacteria from basic science to applications. Academic Press, Amsterdam, pp 327–346
Lagarde D, Beuf L, Vermaas W (2000) Increased production of zeaxanthin and other pigments by application of genetic engineering techniques to Synechocystis sp. strain PCC 6803. Appl Environ Microbiol 66:64–72
Latifi A, Ruiz M, Zhang CC (2009) Oxidative stress in cyanobacteria. FEMS Microbiol Rev 33:258–278
Lee HS, Kim ZH, Park H, Lee CG (2016) Specific light uptake rates can enhance astaxanthin productivity in Haematococcus lacustris. Bioprocess Biosyst Eng 39:815–823
Ludwig M, Bryant DA (2012) Synechococcus sp. strain PCC 7002 transcriptome: acclimation to temperature, salinity, oxidative stress, and mixotrophic growth conditions. Front Microbiol 3:1–14
Manchanda H, Sharma V (2018) Effect of salinity and lipid content of cyanobacterium Calothrix marchica. J Biol Chem Res 35:1009–1014
Martins R, Fernandez N, Beiras R, Vasconcelos V (2007) Toxicity assessment of crude and partially purified extracts of marine Synechocystis and Synechococcus cyanobacterial strains in marine invertebrates. Toxicon 50:791–799
Matyash V, Liebisch G, Kurzchalia TV, Shevchenko A, Schwidke D (2008) Lipid extraction by methyl-tert-butyl ether for high-throughput lipidomics. J Lipid Res 49:1137–1146
Mizusawa N, Wada H (2012) The role of lipids in photosystem II. Biochim Biophys Acta Bioenerg 1817:194–208
Mostafa EM, Tammam AA (2012) The oxidative stress caused by NaCl in Azolla caroliniana is mitigated by nitrate. J Plant Interact 7:356–366
Noctor G, Mhamdi A, Chaouch S, Han Y, Neukermans J, Marquez-Garcia B, Queval G, Foyer CH (2012) Glutathione in plants: an integrated overview. Plant Cell Environ 35:454–484
Osanai T, Kuwahara A, Iijima H, Toyooka K, Sato M, Tanaka K, Ikeuchi M, Saito K, Hirai MY (2013) Pleiotropic effect of sigE over-expression on cell morphology, photosynthesis and hydrogen production in Synechocystis sp. PCC 6803. Plant J 76:456–465
Pade N, Hagemann M (2015) Salt acclimation of cyanobacteria and their application in biotechnology. Life 5:25–49
Pagels F, Guedes AC, Amaro HM, Kijjoa A, Vasconcelos V (2019) Phycobiliproteins from cyanobacteria: chemistry and biotechnological applications. Biotechnol Adv 37:422–443
Paliwal C, Mitra M, Bhayani K, Bhadadwaj SVV, Ghosh T, Dubey S, Mishra S (2017) Abiotic stresses as tools for metabolites in microalgae. Bioresour Technol 244:1216–1226
Park BI, Hong SJ, Kwan CB et al (2018) Characterization of culture conditions to increase phycobiliprotein production of Synechocystis sp. PCC 7338. Abstract of the 2018 KSBB International Academica-Industry Joint Meeting 460
Pasteur Institution (2002) CRBIP-catalogue. http://catalogue-crbip.pasteur.fr/. Accessed 20 Sept 2019
Patterson BD, MacRae EA, Ferguson IB (1984) Estimation of hydrogen peroxide in plant extracts using titanium(IV). Anal Biochem 139:487–492
Rai AK, Abraham G (1993) Salinity tolerance and growth analysis of the cyanobacterium Anabaena doliolum. Bull Environ Contam Toxicol 51:724–731
Řezanka T, Víden I, Go JV, Dor I, Dembitsky VM (2003) Polar lipids and fatty acids of three wild cyanobacterial strains of the genus Chroococcidiopsis. Folia Microbiol (Praha) 48:781–786
Rippka R, Deruelles J, Waterbury JB (1979) Generic assignments, strain histories and properties of pure cultures of cyanobacteria. J Gen Microbiol 111:1–61
Ritter D, Yopp JH (1993) Plasma membrane lipid composition of the halophilic cyanobacterium Aphanothece halophytica. Arch Microbiol 159:435–439
Ruangsomboon S (2014) Effect of media and salinity on lipid content of cyanobacterium Hapalosiphon sp. Chiang Mai J Sci 41:307–315
Sato N, Hagio M, Wada H, Tsuzuki M (2000) Requirement of phosphatidylglycerol for photosynthetic function in thylakoid membranes. Proc Natl Acad Sci U S A 97:10655–10660
Schubert H, Fulda S, Hagemann M (1993) Effects of adaptation to different salt concentrations on photosynthesis and pigmentation of the cyanobacterium Synechocystis sp. PCC 6803. J Plant Physiol 142:291–295
Schwarz D, Orf I, Kopka J, Hagemann M (2013) Recent applications of metabolomics toward cyanobacteria. Metabolites 3:72–100
Sharma G, Kumar M, Ali MI, Jasuja ND (2014) Effect of carbon content, salinity and pH on Spirulina platensis for phycocyanin, allophycocyanin and phycoerythrin accumulation. J Microb Biochem Technol 6:202–206
Singh SC, Sinha RP, Häder DP (2002) Role of lipids and fatty acids in stress tolerance in cyanobacteria. Acta Protozool 41:297–308
Stadnichuk IN, Tropin IV (2017) Phycobiliproteins: structure, functions and biotechnological applications. Appl Biochem Microbiol 53:1–10
Tian X, Chen L, Wang J, Qiao J, Zhang W (2013) Quantitative proteomics reveals dynamic responses of Synechocystis sp. PCC 6803 to next-generation biofuel butanol. J Proteome 78:326–345
Tran DT, Yeh KL, Chen CL, Chang JS (2012) Enzymatic transesterification of microalgal oil from Chlorella vulgaris ESP-31 for biodiesel synthesis using immobilized Burkholderia lipase. Bioresour Technol 108:119–127
Vijayakumar S, Menakha M (2015) Pharmaceutical applications of cyanobacteria-a review. J Acute Med 5:15–23
Wada H, Murata N (1998) Membrane lipids in cyanobacteria. In: Siegenthaler PA, Murata N (eds) Lipids in photosynthesis: structure, function and genetics. Springer, Dordrecht, pp 65–81
Waditee R, Hibino T, Nakamura T, Icharoensadki A, Takabe T (2002) Overexpression of a Na+/H+ antiporter confers salt tolerance on a freshwater cyanobacterium, making it capable of growth in sea water. Proc Natl Acad Sci U S A 99:4109–4114
Xue Y, He Q (2015) Cyanobacteria as cell factories to produce plant secondary metabolites. Front Bioeng Biotechnol 3:1–6
Yoshihara S, Geng X, Ikeuchi M (2002) pilG gene cluster and split pilL genes involved in pilus biogenesis, motility and genetic transformation in the cyanobacterium Synechocystis sp. PCC 6803. Plant Cell Physiol 43:513–521
Zahra Z, Kim S-Y, Kim H-Y, Lee H, Lee H, Jeon J-Y, Kim D-M, Kim D-M, Hong S-J, Cho B-K, Lee H, Lee C-G, Arshad M, Choi H-K (2018) Phycobiliproteins production enhancement and lipidomic alteration by titanium dioxide nanoparticles in Synechocystis sp. PCC 6803 culture. J Agric Food Chem 66:8522–8529
Zakhozhii IG, Matalin DA, Popova LG, Balnokin YV (2012) Responses of photosynthetic apparatus of the halotolerant microalga Dunalliella maritima to hyperosmotic salt shock. Russ J Plant Physiol 59:42–49
Funding
This work was supported by grants from the National Research Foundation of Korea (NRF) (NRF-2015R1A5A1008958 and NRF-2016M1A5A1027464), funded by the Korean government (MSIP).
Author information
Authors and Affiliations
Contributions
HL contributed to performing experiments, analysis, and interpretation of the data, and writing and revising the manuscript. YN and SH contributed to performing experiments and revising the manuscript. HL, DK, BC, and CL contributed to the conception of the experiments and supervising the research. HC contributed to the conception of the experiments, supervising the research, and writing and revising the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 277 kb)
Rights and permissions
About this article
Cite this article
Lee, H., Noh, Y., Hong, SJ. et al. Photosynthetic pigment production and metabolic and lipidomic alterations in the marine cyanobacteria Synechocystis sp. PCC 7338 under various salinity conditions. J Appl Phycol 33, 197–209 (2021). https://doi.org/10.1007/s10811-020-02273-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-020-02273-3