Abstract
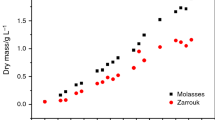
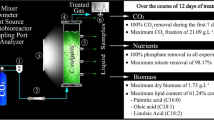
As a new interest for biodegradable non-hazardous biolubricant from renewable resources, microalgae lipid is suggested as a new feedstock by introducing the microalgae-based lubricants. Chlorella vulgaris was successfully grown in a cheap substrate-based mixotrophic medium. The kinetic modeling of microalgae growth, lipid production, and substrate consumption was carried out in optimum conditions of biomass productivity and lipid production to enhance microalgae lipid for biolubricant production. Designed models have good compatibility with more than 95 % confidence level when compared to the cultivation system. Validation of the models with additional experiments confirmed the accuracy of the models to predict new conditions. The highest biomass concentration of C. vulgaris was 2.9 g L−1 with a lipid content of 30 % of dry weight. The model proposed for lipid production indicated that the lipid was produced simultaneous with growth. Microalgae lipid had sufficient lubricating property showing that this microalgal lipid could be used as potential feedstock for biolubricant production.

Kinetic modeling of mixotrophic growth of Chlorella vulgaris was carried out in optimum conditions of growth parameters. Microalgae lipid was suggested as a new feedstock for biolubricants.





Similar content being viewed by others
References
Abreu AP, Fernandes B, Vicente AA, Teixeira J, Dragone G (2012) Mixotrophic cultivation of Chlorella vulgaris using industrial dairy waste as organic carbon source. Bioresour Technol 118:61–66
Adesanya VO, Davey MP, Scott SA, Smith AG (2014) Kinetic modelling of growth and storage molecule production in microalgae under mixotrophic and autotrophic conditions. Bioresour Technol 157:293–304
Barbosa MJ, Hoogakker J, Wijffels RH (2003) Optimisation of cultivation parameters in photobioreactors for microalgae cultivation using the A-stat technique. Biomol Eng 20:115–123
Bitaubé Pérez E, Caro Pina I, Pérez Rodríguez L (2008) Kinetic model for growth of Phaeodactylum tricornutum in intensive culture photobioreactor. Biochem Eng J 40:520–525
Cavalcante IM, Rocha NRC, Maier ME, de Lima APD, Andrade Neto DM, de Brito DHA, Petzhold CL, Schanz MTGF, Ricardo NMPS (2014) Synthesis and characterization of new esters of oleic acid and glycerol analogues as potential lubricants. Ind Crop Prod 62:453–459
Chojnacka K, Noworyta A (2004) Evaluation of Spirulina sp. growth in photoautotrophic, heterotrophic and mixotrophic cultures. Enzyme Microb Technol 34:461–465
Dörmő N, Bélafi-Bakó K, Bartha L, Ehrenstein U, Gubicza L (2004) Manufacture of an environmental-safe biolubricant from fusel oil by enzymatic esterification in solvent-free system. Biochem Eng J 21(3):229–234
DuBois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Economou CN, Aggelis G, Pavlou S, Vayenas DV (2011) Modeling of single-cell oil production under nitrogen-limited and substrate inhibition conditions. Biotechnol Bioeng 108:1049–1055
Gharibzahedi SMT, Razavi SH, Mousavi M (2013) Kinetic analysis and mathematical modeling of cell growth and canthaxanthin biosynthesis by Dietzia natronolimnaea HS-1 on waste molasses hydrolysate. RSC Adv 3:23495–23502
Halim R, Gladman B, Danquah MK, Webley PA (2011) Oil extraction from microalgae for biodiesel production. Bioresour Technol 102:178–185
Heredia-Arroyo T, Wei W, Ruan R, Hu B (2011) Mixotrophic cultivation of Chlorella vulgaris and its potential application for the oil accumulation from non-sugar materials. Biomass Bioenergy 35:2245–2253
Kamalakar K, Rajak AK, Prasad RBN, Karuna MSL (2013) Rubber seed oil-based biolubricant base stocks: a potential source for hydraulic oils. Ind Crop Prod 51:249–257
Kleinaitė E, Jaška V, Tvaska B, Matijošytė I (2014) A cleaner approach for biolubricant production using biodiesel as a starting material. J Clean Prod 75:40–44
Knothe G (2009) Improving biodiesel fuel properties by modifying fatty ester composition. Energy Environ Sci 2:759–766
Kumar K, Dasgupta CN, Das D (2014) Cell growth kinetics of Chlorella sorokiniana and nutritional values of its biomass. Bioresour Technol 167:358–366
Leesing R, Sihawong S, Duangkeaw N (2013) Producing of microalgal lipid by isolated microalgae under photoautotrophic and heterotrophic cultivations. APCBEE Procedia 7:48–53
Liang Y (2013) Producing liquid transportation fuels from heterotrophic microalgae. Appl Energy 104:860–868
Liang Y, Sarkany N, Cui Y (2009) Biomass and lipid productivities of Chlorella vulgaris under autotrophic, heterotrophic and mixotrophic growth conditions. Biotechnol Lett 31:1043–1049
Lichtenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. In: Lester Packer RD (ed) Methods Enzymol., Volume 148. Academic Press, pp 350–382
Lowrey J, Brooks M, McGinn P (2015) Heterotrophic and mixotrophic cultivation of microalgae for biodiesel production in agricultural wastewaters and associated challenges—a critical review. J Appl Phycol 27:1485–1498
Metcalfe LD, Schmitz AA, Pelka JR (1966) Rapid preparation of fatty acid esters from lipids for gas chromatographic analysis. Anal Chem 38:514–515
Mohammad Mirzaie MA, Kalbasi M, Mousavi SM, Ghobadian B (2015) Statistical evaluation and modeling of cheap substrate-based cultivation medium of Chlorella vulgaris to enhance microalgae lipid as new potential feedstock for biolubricant. Prep Biochem Biotechnol. doi:10.1080/10826068.2015.1031398
Mohammad Mirzaie MA, Kalbasi M, Mousavi SM, Ghobadian B (2016) Investigation of mixotrophic, heterotrophic, and autotrophic growth of Chlorella vulgaris under agricultural waste medium. Prep Biochem Biotechnol 46:150–156
Moya MJ, Sánchez-Guardamino ML, Vilavella A, Barberà E (1997) Growth of Haematococcus lacustris: a contribution to kinetic modelling. J Chem Technol Biotechnol 68:303–309
Naidir F, Yunus R, Rashid U, Masood H, Ghazi TIM, Ramli I (2012) The kinetics of epoxidation of trimethylolpropane ester. Eur J Lipid Sci Technol 114:816–822
Najafi G, Ghobadian B, Yusaf TF (2011) Algae as a sustainable energy source for biofuel production in Iran: a case study. Renew Sust Energ Rev 15:3870–3876
Nor Halaliza Alias RY, Idris A, Omar R (2009) Effects of additives on oxidation characteristics of palm oilbased trimethylolpropane ester in hydraulics applications. Eur J Lipid Sci Technol 111:368–375
Ogbonna J, Tanaka H (2000) Light requirement and photosynthetic cell cultivation—development of processes for efficient light utilization in photobioreactors. J Appl Phycol 12:207–218
Orus IM, Martinez F (1991) Chlorophyll a/b ratio and thylakoid stacking modification in response to glucose in Chlorella vulgaris UAM 101. Biochem Physiol Pflanz 187:197–202
Paz B, Vázquez JA, Riobó P, Stolte W, Franco JM (2009) Mathematical description of yessotoxin production by Protoceratium reticulatum in culture. Harmful Algae 8:730–735
Pruvost J, Van Vooren G, Le Gouic B, Couzinet-Mossion A, Legrand J (2011) Systematic investigation of biomass and lipid productivity by microalgae in photobioreactors for biodiesel application. Bioresour Technol 102:150–158
Sripada PK, Sharma RV, Dalai AK (2013) Comparative study of tribological properties of trimethylolpropane-based biolubricants derived from methyl oleate and canola biodiesel. Ind Crop Prod 50:95–103
Wang E, Ma X, Tang S, Yan R, Wang Y, Riley WW, Reaney MJT (2014) Synthesis and oxidative stability of trimethylolpropane fatty acid triester as a biolubricant base oil from waste cooking oil. Biomass Bioenergy 66:371–378
Wong Y-S, Teng TT, Ong S-A, Morad N, Rafatullah M (2014) Suspended growth kinetic analysis on biogas generation from newly isolated anaerobic bacterial communities for palm oil mill effluent at mesophilic temperature. RSC Adv 4:64659–64667
Xie J, Zhang Y, Li Y, Wang Y (2001) Mixotrophic cultivation of Platymonas subcordiformis. J Appl Phycol 13:343–347
Yang J, Rasa E, Tantayotai P, Scow KM, Yuan H, Hristova KR (2011) Mathematical model of Chlorella minutissima UTEX2341 growth and lipid production under photoheterotrophic fermentation conditions. Bioresour Technol 102:3077–3082
Yu G, Shi D, Cai Z, Cong W, Ouyang F (2011) Growth and physiological features of cyanobacterium Anabaena sp. strain PCC 7120 in a glucose-mixotrophic culture. Chin J Chem Eng 19:108–115
Zhang XW, Zhang YM, Chen F (1998) Kinetic models for phycocyanin production by high cell density mixotrophic culture of the microalga Spirulina platensis. J Ind Microbiol Biotechnol 21:283–288
Zhang XW, Zhang YM, Chen F (1999) Application of mathematical models to the determination optimal glucose concentration and light intensity for mixotrophic culture of Spirulina platensis. Process Biochem 34:477–481
Zhang D, Yu Y, Li C, Chai C, Liu L, Liu J, Feng Y (2015) Factors affecting microalgae harvesting efficiencies using electrocoagulation-flotation for lipid extraction. RSC Adv 5:5795–5800
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mohammad Mirzaie, M.A., Kalbasi, M., Ghobadian, B. et al. Kinetic modeling of mixotrophic growth of Chlorella vulgaris as a new feedstock for biolubricant. J Appl Phycol 28, 2707–2717 (2016). https://doi.org/10.1007/s10811-016-0841-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-016-0841-4