Abstract
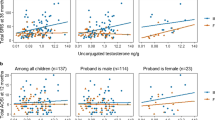
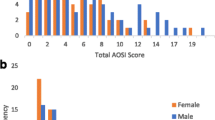
Fetal androgen exposure may be associated with autism spectrum disorder (ASD). We studied 1777 mother–child pairs in the prospective Odense Child Cohort. Prenatal androgen exposure was assessed by maternal 3rd trimester testosterone concentrations, maternal polycystic ovary syndrome (PCOS), and 3 months offspring anogenital distance. ASD traits were assessed at age 3 years with the ASD-symptom scale of the Child Behavior Checklist for ages 1½–5 years. Maternal testosterone was positively associated with traits of ASD in boys (p < 0.05). Maternal PCOS was associated with increased offspring ASD traits (p = 0.046), but became non-significant after excluding parental psychiatric diagnosis. Offspring anogenital distance was not linked to ASD traits. Higher prevalence of ASD in boys could be linked to higher susceptibility to fetal androgen exposure.


Similar content being viewed by others
References
A. P. A. (2013). Diagnostic and statistical manual of mental disorders: DSM-5 (5th ed). American Psychiatric Association.
Atkinson, G., Campbell, D. J., Cawood, M. L., & Oakey, R. E. (1996). Steroids in human intrauterine fluids of early pregnancy. Clinical Endocrinology - Oxford, 44(4), 435–440. https://doi.org/10.1046/j.1365-2265.1996.710532.x
Auyeung, B., Baron-Cohen, S., Ashwin, E., Knickmeyer, R., Taylor, K., & Hackett, G. (2009). Fetal testosterone and autistic traits. British Journal of Psychology, 100(Pt 1), 1–22. https://doi.org/10.1348/000712608x311731
Auyeung, B., Taylor, K., Hackett, G., & Baron-Cohen, S. (2010). Foetal testosterone and autistic traits in 18 to 24-month-old children. Mol Autism, 1(1), 11. https://doi.org/10.1186/2040-2392-1-11
Bammann, B. L., Coulam, C. B., & Jiang, N. S. (1980). Total and free testosterone during pregnancy. American Journal of Obstetrics and Gynecology, 137(3), 293–298.
Baron-Cohen, S. (2002). The extreme male brain theory of autism. Trends in Cognitive Sciences, 6(6), 248–254. https://doi.org/10.1016/s1364-6613(02)01904-6
Baron-Cohen, S., Auyeung, B., Nørgaard-Pedersen, B., Hougaard, D. M., Abdallah, M. W., Melgaard, L., Cohen, A. S., Chakrabarti, B., Ruta, L., & Lombardo, M. V. (2015). Elevated fetal steroidogenic activity in autism. Molecular Psychiatry, 20(3), 369–376. https://doi.org/10.1038/mp.2014.48
Baron-Cohen, S., Knickmeyer, R. C., & Belmonte, M. K. (2005). Sex differences in the brain: Implications for explaining autism. Science, 310(5749), 819–823. https://doi.org/10.1126/science.1115455
Baron-Cohen, S., Lombardo, M. V., Auyeung, B., Ashwin, E., Chakrabarti, B., & Knickmeyer, R. (2011). Why are autism spectrum conditions more prevalent in males? PLoS Biology, 9(6), e1001081. https://doi.org/10.1371/journal.pbio.1001081
Cesta, C. E., Öberg, A. S., Ibrahimson, A., Yusuf, I., Larsson, H., Almqvist, C., D’Onofrio, Brian M., Bulik, Cynthia M., Fernández, Lorena, de la Cruz, David, & Mataix-Cols, MA Rosenqvist. (2020). Maternal polycystic ovary syndrome and risk of neuropsychiatric disorders in offspring: prenatal androgen exposure or genetic confounding? Psychological Medicine, 50(4), 616–624. https://doi.org/10.1017/s0033291719000424
Cherskov, A., Pohl, A., Allison, C., Zhang, H., Payne, R. A., & Baron-Cohen, S. (2018). Polycystic ovary syndrome and autism: A test of the prenatal sex steroid theory. Translational Psychiatry, 8(1), 136. https://doi.org/10.1038/s41398-018-0186-7
Cheslack-Postava, K., Jokiranta, E., Suominen, A., Lehti, V., Sourander, A., & Brown, A. S. (2014). Variation by diagnostic subtype in risk for autism spectrum disorders associated with maternal parity among Finnish births. Paediatric and Perinatal Epidemiology, 28(1), 58–66. https://doi.org/10.1111/ppe.12094
Conway, G., Dewailly, D., Diamanti-Kandarakis, E., Escobar-Morreale, H. F., Franks, S., Gambineri, A., Kelestimur, Fahrettin, Macut, Djuro, Micic, Dragan, Pasquali, Renato, Pfeifer, Marija, Pignatelli, Duarte, Michel Pugeat, B. O., & Yildiz. (2014). The polycystic ovary syndrome: A position statement from the European Society of Endocrinology. European Journal of Endocrinology, 171(4), P1-29. https://doi.org/10.1530/eje-14-0253
Dean, A., & Sharpe, R. M. (2013). Clinical review: Anogenital distance or digit length ratio as measures of fetal androgen exposure: Relationship to male reproductive development and its disorders. Journal of Clinical Endocrinology and Metabolism, 98(6), 2230–2238. https://doi.org/10.1210/jc.2012-4057
Duan, C., Pei, T., Li, Y., Cao, Q., Chen, H., & Fu, J. (2020). Androgen levels in the fetal cord blood of children born to women with polycystic ovary syndrome: A meta-analysis. Reproductive Biology and Endocrinology, 18(1), 81. https://doi.org/10.1186/s12958-020-00634-8
Filová, B., Ostatníková, D., Celec, P., & Hodosy, J. (2013). The effect of testosterone on the formation of brain structures. Cells, Tissues, Organs, 197(3), 169–177. https://doi.org/10.1159/000345567
Finnbogadottir, S. K., Glintborg, D., Jensen, T. K., Kyhl, H. B., Nohr, E. A., & Andersen, M. (2017). Insulin resistance in pregnant women with and without polycystic ovary syndrome, and measures of body composition in offspring at birth and three years of age. Acta Obstetricia Et Gynecologica Scandinavica, 96(11), 1307–1314. https://doi.org/10.1111/aogs.13200
Fombonne, E., MacFarlane, H., & Salem, A. C. (2021). Epidemiological surveys of ASD: Advances and remaining challenges. Journal of Autism and Developmental Disorders. https://doi.org/10.1007/s10803-021-05005-9
Geschwind, D. H. (2008). Autism: Many genes, common pathways? Cell, 135(3), 391–395. https://doi.org/10.1016/j.cell.2008.10.016
Glintborg, D., Hass Rubin, K., Nybo, M., Abrahamsen, B., & Andersen, M. (2015). Morbidity and medicine prescriptions in a nationwide Danish population of patients diagnosed with polycystic ovary syndrome. European Journal of Endocrinology, 172(5), 627–638. https://doi.org/10.1530/eje-14-1108
Glintborg, D., Jensen, R. C., Bentsen, K., Schmedes, A. V., Brandslund, I., Kyhl, H. B., Bilenberg, N., & Andersen, M. S. (2018). Testosterone levels in 3rd trimester in polycystic ovary syndrome. Odense Child Cohort. Journal of Molecular Medicine. https://doi.org/10.1210/jc.2018-00889
Glintborg, D., Jensen, R. C., Schmedes, A. V., Brandslund, I., Kyhl, H. B., Jensen, T. K., & Andersen, M. S. (2019). Anogenital distance in children born of mothers with polycystic ovary syndrome: The Odense Child Cohort. Human Reproduction, 34(10), 2061–2070. https://doi.org/10.1093/humrep/dez122
Idring, S., Magnusson, C., Lundberg, M., Ek, M., Rai, D., Svensson, A. C., Dalman, C., Karlsson, H., & Lee, B. K. (2014). Parental age and the risk of autism spectrum disorders: Findings from a Swedish population-based cohort. International Journal of Epidemiology, 43(1), 107–115. https://doi.org/10.1093/ije/dyt262
James, W. H., & Grech, V. (2020). Is exposure to high levels of maternal intrauterine testosterone a causal factor common to male sex, autism, gender dysphoria, and non-right-handedness? Early Human Development, 141, 104872. https://doi.org/10.1016/j.earlhumdev.2019.104872
Jensen, R. C., Jensen, D. M., Gibbons, K. S., Glintborg, D., Jensen, T. K., McIntyre, H. D., & Andersen, M. (2021). Adapting fasting plasma glucose threshold for GDM diagnosis according to the population distribution - An approach to the Danish paradox. Diabetes Research and Clinical Practice, 175, 108832. https://doi.org/10.1016/j.diabres.2021.108832
Karlson, K., Breen, R., & Holm, A. (2013). Total, direct, and indirect effects in logit and probit models. Sociological Methods & Amp Research. https://doi.org/10.1177/0049124113494572
Katsigianni, M., Karageorgiou, V., Lambrinoudaki, I., & Siristatidis, C. (2019). Maternal polycystic ovarian syndrome in autism spectrum disorder: A systematic review and meta-analysis. Molecular Psychiatry, 24(12), 1787–1797. https://doi.org/10.1038/s41380-019-0398-0
Kelley, A. S., Puttabyatappa, M., Ciarelli, J. N., Zeng, L., Smith, Y. R., Lieberman, R., Pennathur, S., & Padmanabhan, V. (2019). Prenatal testosterone excess disrupts placental function in a sheep model of polycystic ovary syndrome. Endocrinology, 160(11), 2663–2672. https://doi.org/10.1210/en.2019-00386
Kosidou, K., Dalman, C., Widman, L., Arver, S., Lee, B. K., Magnusson, C., & Gardner, R. M. (2016). Maternal polycystic ovary syndrome and the risk of autism spectrum disorders in the offspring: A population-based nationwide study in Sweden. Molecular Psychiatry, 21(10), 1441–1448. https://doi.org/10.1038/mp.2015.183
Kristensen, S., Henriksen, T. B., & Bilenberg, N. (2010). The Child Behavior Checklist for Ages 1.5–5 (CBCL/1(1/2)-5): assessment and analysis of parent- and caregiver-reported problems in a population-based sample of Danish preschool children. Nordic Journal of Psychiatry, 64(3), 203–209. https://doi.org/10.3109/08039480903456595
Kung, K. T. F., Thankamony, A., Ong, K. K. L., Acerini, C. L., Dunger, D. B., Hughes, I. A., & Hines, M. (2021). No relationship between prenatal or early postnatal androgen exposure and autistic traits: Evidence using anogenital distance and penile length measurements at birth and 3 months of age. Journal of Child Psychology and Psychiatry, 62(7), 876–883. https://doi.org/10.1111/jcpp.13335
Kyhl, H. B., Jensen, T. K., Barington, T., Buhl, S., Norberg, L. A., Jorgensen, J. S., Jensen, D. F., Christesen, H. T., Lamont, R. F., & Husby, S. (2015). The odense child cohort: Aims, design, and cohort profile. Paediatric and Perinatal Epidemiology, 29(3), 250–258. https://doi.org/10.1111/ppe.12183
Larsson, M., Weiss, B., Janson, S., Sundell, J., & Bornehag, C. G. (2009). Associations between indoor environmental factors and parental-reported autistic spectrum disorders in children 6–8 years of age. Neurotoxicology, 30(5), 822–831. https://doi.org/10.1016/j.neuro.2009.01.011
Levy, S. E., Rescorla, L. A., Chittams, J. L., Kral, T. J., Moody, E. J., Pandey, J., Pinto-Martin, Jennifer A., Pomykacz, Alison T., Ramirez, AnnJosette, Reyes, Nuri, Rosenberg, Cordelia Robinson, Schieve, Laura, Thompson, Aleda, Young, Lisa, Zhang, Jing, & Wiggins, L. (2019). ASD screening with the child behavior checklist/1.5–5 in the study to explore early development. Journal of Autism and Developmental Disorders, 49(6), 2348–2357. https://doi.org/10.1007/s10803-019-03895-4
Lord, C., Brugha, T. S., Charman, T., Cusack, J., Dumas, G., Frazier, T., Jones, Emily J. H., Jones, Rebecca M., Pickles, Andrew, State, Matthew W., Taylor, Julie Lounds, & Veenstra-VanderWeele, J. (2020). Autism spectrum disorder. Nature Reviews Disease Primers, 6(1), 5. https://doi.org/10.1038/s41572-019-0138-4
Lung, F. W., Chiang, T. L., Lin, S. J., Lee, M. C., & Shu, B. C. (2018). Advanced maternal age and maternal education disparity in children with autism spectrum disorder. Maternal and Child Health Journal, 22(7), 941–949. https://doi.org/10.1007/s10995-018-2470-9
Maher, G. M., O’Keeffe, G. W., Dalman, C., Kearney, P. M., McCarthy, F. P., Kenny, L. C., & Khashan, A. S. (2020). Association between preeclampsia and autism spectrum disorder: A population-based study. Journal of Child Psychology and Psychiatry, 61(2), 131–139. https://doi.org/10.1111/jcpp.13127
Makieva, S., Saunders, P. T., & Norman, J. E. (2014). Androgens in pregnancy: Roles in parturition. Human Reproduction Update, 20(4), 542–559. https://doi.org/10.1093/humupd/dmu008
Maliqueo, M., Lara, H. E., Sánchez, F., Echiburú, B., Crisosto, N., & Sir-Petermann, T. (2013). Placental steroidogenesis in pregnant women with polycystic ovary syndrome. European Journal of Obstetrics, Gynecology, and Reproductive Biology, 166(2), 151–155. https://doi.org/10.1016/j.ejogrb.2012.10.015
Matias, S. L., Pearl, M., Lyall, K., Croen, L. A., Kral, T. V. E., Fallin, D., Lee, Li-Ching., Bradley, Chyrise B., Schieve, Laura A., & Windham, G. C. (2021). Maternal prepregnancy weight and gestational weight gain in association with autism and developmental disorders in offspring. Obesity (Silver Spring), 29(9), 1554–1564. https://doi.org/10.1002/oby.23228
Muratori, F., Narzisi, A., Tancredi, R., Cosenza, A., Calugi, S., Saviozzi, I., Santocchi, E., & Calderoni, S. (2011). The CBCL 1.5–5 and the identification of preschoolers with autism in Italy. Epidemiology and Psychiatric Sciences, 20(4), 329–338. https://doi.org/10.1017/s204579601100045x
Narzisi, A., Calderoni, S., Maestro, S., Calugi, S., Mottes, E., & Muratori, F. (2013). Child Behavior Check List 1½-5 as a tool to identify toddlers with autism spectrum disorders: A case-control study. Research in Developmental Disabilities, 34(4), 1179–1189. https://doi.org/10.1016/j.ridd.2012.12.020
Nguyen, T. V. (2018). Developmental effects of androgens in the human brain. Journal of Neuroendocrinology. https://doi.org/10.1111/jne.12486
O’Leary, P., Boyne, P., Flett, P., Beilby, J., & James, I. (1991). Longitudinal assessment of changes in reproductive hormones during normal pregnancy. Clinical Chemistry, 37(5), 667–672.
Organization, W. H. (2019). International Statistical Classification of Diseases (ICD-11).
Ostatníková, D., Lakatošová, S., Babková, J., Hodosy, J., & Celec, P. (2020). Testosterone and the brain: From cognition to autism. Physiological Research, 69(Suppl 3), S403–S419. https://doi.org/10.33549/physiolres.934592
Palm, C. V. B., Glintborg, D., Kyhl, H. B., McIntyre, H. D., Jensen, R. C., Jensen, T. K., Møller, D., & Andersen, M. (2018). Polycystic ovary syndrome and hyperglycaemia in pregnancy. A narrative review and results from a prospective Danish cohort study. Diabetes Research and Clinical Practice. https://doi.org/10.1016/j.diabres.2018.04.030
Park, B. Y., Lee, B. K., Burstyn, I., Tabb, L. P., Keelan, J. A., Croen, L. A., Whitehouse, J. O., Fallin, M. D., Hertz-Picciotto, I., Montgomery, O., & Newschaffer, C. J. (2017). Umbilical cord blood androgen levels and ASD-related phenotypes at 12 and 36 months in an enriched risk cohort study. Molecular Autism, 8, 3. https://doi.org/10.1186/s13229-017-0118-z
Priskorn, L., Petersen, J. H., Jørgensen, N., Kyhl, H. B., Andersen, M. S., Main, K. M., Andersson, A.-M., Skakkebaek, N. E., & Jensen, T. K. (2018). Anogenital distance as a phenotypic signature through infancy. Pediatric Research, 83(3), 573–579. https://doi.org/10.1038/pr.2017.287
Raperport, C., & Homburg, R. (2019). The source of polycystic ovarian syndrome. Clin Med Insights Reprod Health, 13, 1179558119871467. https://doi.org/10.1177/1179558119871467
Recabarren, S. E., Padmanabhan, V., Codner, E., Lobos, A., Durán, C., Vidal, M., Foster, D. L., & Sir-Petermann, T. (2005). Postnatal developmental consequences of altered insulin sensitivity in female sheep treated prenatally with testosterone. American Journal of Physiology Endocrinology and Metabolism, 289(5), E801-806. https://doi.org/10.1152/ajpendo.00107.2005
Rescorla, L. (1988). Cluster analytic identification of autistic preschoolers. Journal of Autism and Developmental Disorders, 18(4), 475–492. https://doi.org/10.1007/bf02211868
Rescorla, L. A., Adams, A., & Ivanova, M. Y. (2020). The CBCL/1½-5’s DSM-ASD scale: Confirmatory factor analyses across 24 societies. Journal of Autism and Developmental Disorders, 50(9), 3326–3340. https://doi.org/10.1007/s10803-019-04189-5
Rescorla, L. A., Ghassabian, A., Ivanova, M. Y., Jaddoe, V. W., Verhulst, F. C., & Tiemeier, H. (2019). Structure, longitudinal invariance, and stability of the Child Behavior Checklist 1½–5’s Diagnostic and Statistical Manual of Mental Disorders-Autism Spectrum Disorder scale: Findings from Generation R (Rotterdam). Autism, 23(1), 223–235. https://doi.org/10.1177/1362361317736201
Rescorla, L. A., Given, C., Glynn, S., Ivanova, M. Y., & Achenbach, T. M. (2019). International comparisons of autism spectrum disorder behaviors in preschoolers rated by parents and caregivers/teachers. Autism, 23(8), 2043–2054. https://doi.org/10.1177/1362361319839151
Rescorla, L., Kim, Y. A., & Oh, K. J. (2015). Screening for ASD with the Korean CBCL/1½-5. Journal of Autism and Developmental Disorders, 45(12), 4039–4050. https://doi.org/10.1007/s10803-014-2255-y
. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Fertility and Sterility, 81(1), 19–25.
Rivarola, M. A., Forest, M. G., & Migeon, C. J. (1968). Testosterone, androstenedione and dehydroepiandrosterone in plasma during pregnancy and at delivery: Concentration and protein binding. Journal of Clinical Endocrinology and Metabolism, 28(1), 34–40. https://doi.org/10.1210/jcem-28-1-34
Sathishkumar, K., Elkins, R., Chinnathambi, V., Gao, H., Hankins, G. D., & Yallampalli, C. (2011). Prenatal testosterone-induced fetal growth restriction is associated with down-regulation of rat placental amino acid transport. Reproductive Biology and Endocrinology, 9, 110. https://doi.org/10.1186/1477-7827-9-110
Sathyanarayana, S., Grady, R., Redmon, J. B., Ivicek, K., Barrett, E., Janssen, S., Nguyen, R., & Swan, S. H. (2015). Anogenital distance and penile width measurements in The Infant Development and the Environment Study (TIDES): Methods and predictors. Journal of Pediatric Urology, 11(2), 76.e71-76. https://doi.org/10.1016/j.jpurol.2014.11.018
Sikora, D. M., Hall, T. A., Hartley, S. L., Gerrard-Morris, A. E., & Cagle, S. (2008). Does parent report of behavior differ across ADOS-G classifications: Analysis of scores from the CBCL and GARS. Journal of Autism and Developmental Disorders, 38(3), 440–448. https://doi.org/10.1007/s10803-007-0407-z
So, P., Greaves-Lord, K., van der Ende, J., Verhulst, F. C., Rescorla, L., & de Nijs, P. F. (2013). Using the Child Behavior Checklist and the Teacher’s Report Form for identification of children with autism spectrum disorders. Autism, 17(5), 595–607. https://doi.org/10.1177/1362361312448855
Sun, M., Sun, B., Qiao, S., Feng, X., Li, Y., Zhang, S., Lin, Y., & Hou, L. (2020). Elevated maternal androgen is associated with dysfunctional placenta and lipid disorder in newborns of mothers with polycystic ovary syndrome. Fertility and Sterility, 113(6), 1275-1285.e1272. https://doi.org/10.1016/j.fertnstert.2020.02.005
Taylor, M. J., Martin, J., Lu, Y., Brikell, I., Lundström, S., Larsson, H., & Lichtenstein, P. (2019). Association of Genetic Risk Factors for Psychiatric Disorders and Traits of These Disorders in a Swedish Population Twin Sample. JAMA Psychiatry, 76(3), 280–289. https://doi.org/10.1001/jamapsychiatry.2018.3652
Thankamony, A., Pasterski, V., Ong, K. K., Acerini, C. L., & Hughes, I. A. (2016). Anogenital distance as a marker of androgen exposure in humans. Andrology, 4(4), 616–625. https://doi.org/10.1111/andr.12156
Traver, S., Geoffray, M. M., Mazières, L., Geneviève, D., Michelon, C., Picot, M. C., & Baghdadli, A. (2021). Association between prenatal and perinatal factors and the severity of clinical presentation of children with ASD: Report from the ELENA COHORT. Journal of Psychiatric Research, 137, 634–642. https://doi.org/10.1016/j.jpsychires.2020.11.005
Vermeulen, A., Verdonck, L., & Kaufman, J. M. (1999). A critical evaluation of simple methods for the estimation of free testosterone in serum. Journal of Clinical Endocrinology and Metabolism, 84(10), 3666–3672. https://doi.org/10.1210/jcem.84.10.6079
Whitehouse, A. J., Mattes, E., Maybery, M. T., Dissanayake, C., Sawyer, M., Jones, R. M., Pennell, C. E., Keelan, J. A., & Hickey, M. (2012). Perinatal testosterone exposure and autistic-like traits in the general population: A longitudinal pregnancy-cohort study. Journal of Neurodevelopmental Disorders, 4(1), 25. https://doi.org/10.1186/1866-1955-4-25
Funding
The study was funded by the Danish Foundation for Scientific Innovation and Technology (09-067180); the Ronald McDonald Children Foundation; Odense University Hospital; the Region of Southern Denmark; Odense Municipality; the Mental Health Services in the Region of Southern Denmark; the Danish Council for Strategic Research; the Danish Programme Commission on Health, Food and Welfare (2101-08-0058); Odense Patient data Exploratory Network (OPEN); Novo Nordisk Foundation (Grant No. NNF15OC00017734); the Danish Council for Independent Research; and the Foundation for research collaboration between Rigshospitalet and Odense University Hospital; the Health Foundation (Helsefonden); the National Board of Social Services; and Lundbeckfonden (F-61171-19-27). There is no conflict of interest of any author that could be perceived as prejudicing the impartiality of the research reported.
Author information
Authors and Affiliations
Contributions
Laura G. Find began the initial data analysis and manuscript construction. Cilia M. Dalgaard and Anja F. Dreyer helped with the initial data analysis. Camilla V. B. Palm took over the data analysis with statistical expert knowledge from Pia V. Larsen. Dorte Glintborg and Camilla V. B. Palm completed the data interpretation and writing of the manuscript, with input and proofreading from Niels Bilenberg, Marianne S. Andersen, Henriette Boye and Tina K. Jensen.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Palm, C.V.B., Glintborg, D., Find, L.G. et al. Prenatal Androgen Exposure and Traits of Autism Spectrum Disorder in the Offspring: Odense Child Cohort. J Autism Dev Disord 53, 1053–1065 (2023). https://doi.org/10.1007/s10803-022-05446-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10803-022-05446-w