Abstract
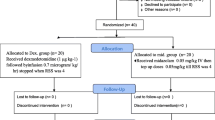
Children with autism and autism spectrum disorders have a high incidence of neurologic comorbidities. Consequently, evaluation with magnetic resonance imaging (MRI) is deemed necessary. Sedating these patients poses several challenges. This retrospective study compared the efficacy and safety of dexmedetomidine to propofol in sedating autistic patients undergoing MRI. There were 56 patients in the dexmedetomidine group and 49 in the propofol group. All of the patients successfully completed the procedure. Recovery and discharge times were significantly lower in the propofol group, while the dexmedetomidine group maintained more stable hemodynamics. Both propofol and dexmedetomidine proved to be adequate and safe medications in the sedation of autistic children undergoing MRI.


Similar content being viewed by others
References
Ahmed, S. S., Unland, T., Slaven, J. E., Nitu, M. E., & Rigby, M. R. (2014). Successful use of intravenous dexmedetomidine for magnetic resonance imaging sedation in autistic children. Southern Medical Journal, 107(9), 559–564. https://doi.org/10.14423/SMJ.0000000000000160.
Berkenbosch, J. W., Wankum, P. C., & Tobias, J. D. (2005). Prospective evaluation of dexmedetomidine for noninvasive procedural sedation in children. Pediatric Critical Care Medicine, 6(4), 435–439. https://doi.org/10.1097/01.PCC.0000163680.50087.93. quiz 440.
Bhana, N., Goa, K. L., & McClellan, K. J. (2000). Dexmedetomidine. Drugs, 59(2), 263–268; discussion 269–270.
Canitano, R., Luchetti, A., & Zappella, M. (2005). Epilepsy, electroencephalographic abnormalities, and regression in children with autism. Journal of Child Neurology, 20(1), 27–31. https://doi.org/10.1177/08830738050200010401.
Chakrabarti, S., & Fombonne, E. (2005). Pervasive developmental disorders in preschool children: Confirmation of high prevalence. American Journal of Psychiatry, 162(6), 1133–1141. https://doi.org/10.1176/appi.ajp.162.6.1133.
Chiaretti, A., Benini, F., Pierri, F., Vecchiato, K., Ronfani, L., Agosto, C., … Barbi, E. (2014). Safety and efficacy of propofol administered by paediatricians during procedural sedation in children. Acta Paediatrica, 103(2), 182–187. https://doi.org/10.1111/apa.12472.
Coté, C. J., Wilson, S., PEDIATRICS, A. A. O., & DENTISTRY, A. A. O. P. (2016). Guidelines for monitoring and management of pediatric patients before, during, and after sedation for diagnostic and therapeutic procedures: Update 2016. Pediatrics. https://doi.org/10.1542/peds.2016-1212.
Coté, C. J., Wilson, S., Pediatrics, A. A. o., Dentistry, A. A. o., P., & Sedation, W. G. o. (2006). Guidelines for monitoring and management of pediatric patients during and after sedation for diagnostic and therapeutic procedures: An update. Pediatrics, 118(6), 2587–2602. https://doi.org/10.1542/peds.2006-2780.
Cravero, J. P., Beach, M. L., Blike, G. T., Gallagher, S. M., Hertzog, J. H., & Consortium, P. S. R. (2009). The incidence and nature of adverse events during pediatric sedation/anesthesia with propofol for procedures outside the operating room: A report from the Pediatric Sedation Research Consortium. Anesthesia & Analgesia, 108(3), 795–804. https://doi.org/10.1213/ane.0b013e31818fc334.
Deegan, R. J. (1992). Propofol: A review of the pharmacology and applications of an intravenous anesthetic agent. American Journal of the Medical Sciences, 304(1), 45–49.
Diagnostic and Statistical Manual of Mental Disorders. (2000). Diagnostic and statistical manual of mental disorders (4th ed.). Washington, DC: American Psychiatric Association.
Disabilities, C. o. C. W., Pediatrics, S. o. D. B., Committee, B. F. S., & Committee, M. H. I. f. C. W. S. N. P. A. (2006). Identifying infants and young children with developmental disorders in the medical home: An algorithm for developmental surveillance and screening. Pediatrics, 118(1), 405–420. https://doi.org/10.1542/peds.2006-1231.
Dyck, J. B., Maze, M., Haack, C., Vuorilehto, L., & Shafer, S. L. (1993). The pharmacokinetics and hemodynamic effects of intravenous and intramuscular dexmedetomidine hydrochloride in adult human volunteers. Anesthesiology, 78(5), 813–820.
Gutstein, H. B., Johnson, K. L., Heard, M. B., & Gregory, G. A. (1992). Oral ketamine preanesthetic medication in children. Anesthesiology, 76(1), 28–33.
Heard, C., Burrows, F., Johnson, K., Joshi, P., Houck, J., & Lerman, J. (2008). A comparison of dexmedetomidine-midazolam with propofol for maintenance of anesthesia in children undergoing magnetic resonance imaging. Anesthesia & Analgesia, 107(6), 1832–1839. https://doi.org/10.1213/ane.0b013e31818874ee.
Hertzog, J. H., Dalton, H. J., Anderson, B. D., Shad, A. T., Gootenberg, J. E., & Hauser, G. J. (2000). Prospective evaluation of propofol anesthesia in the pediatric intensive care unit for elective oncology procedures in ambulatory and hospitalized children. Pediatrics, 106(4), 742–747.
Kallio, A., Scheinin, M., Koulu, M., Ponkilainen, R., Ruskoaho, H., Viinamäki, O., & Scheinin, H. (1989). Effects of dexmedetomidine, a selective alpha 2-adrenoceptor agonist, on hemodynamic control mechanisms. Clinical Pharmacology & Therapeutics, 46(1), 33–42.
Kamat, P. P., McCracken, C. E., Gillespie, S. E., Fortenberry, J. D., Stockwell, J. A., Cravero, J. P., & Hebbar, K. B. (2015). Pediatric critical care physician-administered procedural sedation using propofol: A report from the Pediatric Sedation Research Consortium Database. Pediatric Critical Care Medicine, 16(1), 11–20. https://doi.org/10.1097/PCC.0000000000000273.
Kanto, J., & Gepts, E. (1989). Pharmacokinetic implications for the clinical use of propofol. Clinical Pharmacokinetics, 17(5), 308–326. https://doi.org/10.2165/00003088-198917050-00002.
Koroglu, A., Demirbilek, S., Teksan, H., Sagir, O., But, A. K., & Ersoy, M. O. (2005). Sedative, haemodynamic and respiratory effects of dexmedetomidine in children undergoing magnetic resonance imaging examination: preliminary results. Br J Anaesth, 94(6), 821–824. https://doi.org/10.1093/bja/aei119.
Koroglu, A., Teksan, H., Sagir, O., Yucel, A., Toprak, H. I., & Ersoy, O. M. (2006). A comparison of the sedative, hemodynamic, and respiratory effects of dexmedetomidine and propofol in children undergoing magnetic resonance imaging. Anesthesia & Analgesia, 103(1), 63–67. https://doi.org/10.1213/01.ANE.0000219592.82598.AA.
Kotani, Y., Shimazawa, M., Yoshimura, S., Iwama, T., & Hara, H. (2008). The experimental and clinical pharmacology of propofol, an anesthetic agent with neuroprotective properties. CNS Neuroscience & Therapeutics, 14(2), 95–106. https://doi.org/10.1111/j.1527-3458.2008.00043.x.
Krauss, B., & Green, S. M. (2006). Procedural sedation and analgesia in children. Lancet, 367(9512), 766–780. https://doi.org/10.1016/S0140-6736(06)68230-5.
Lubisch, N., Roskos, R., & Berkenbosch, J. W. (2009). Dexmedetomidine for procedural sedation in children with autism and other behavior disorders. Pediatric Neurology, 41(2), 88–94. https://doi.org/10.1016/j.pediatrneurol.2009.02.006.
Mason, K. P., Sanborn, P., Zurakowski, D., Karian, V. E., Connor, L., Fontaine, P. J., & Burrows, P. E. (2004). Superiority of pentobarbital versus chloral hydrate for sedation in infants during imaging. Radiology, 230(2), 537–542. https://doi.org/10.1148/radiol.2302030107.
Mason, K. P., Zgleszewski, S. E., Dearden, J. L., Dumont, R. S., Pirich, M. A., Stark, C. D., … Zurakowski, D. (2006). Dexmedetomidine for pediatric sedation for computed tomography imaging studies. Anesthesia & Analgesia, 103(1), 57–62. https://doi.org/10.1213/01.ane.0000216293.16613.15.
Mehta, U. C., Patel, I., & Castello, F. V. (2004). EEG sedation for children with autism. Journal of Developmental & Behavioral Pediatrics, 25(2), 102–104.
Myers, S. M., Johnson, C. P., & Disabilities, A. A. o. P. C. o. C. W. (2007). Management of children with autism spectrum disorders. Pediatrics, 120(5), 1162–1182. https://doi.org/10.1542/peds.2007-2362.
Rainey, L., & van der Walt, J. H. (1998). The anaesthetic management of autistic children. Anaesth Intensive Care, 26(6), 682–686.
Seid, M., Sherman, M., & Seid, A. B. (1997). Perioperative psychosocial interventions for autistic children undergoing ENT surgery. International Journal of Pediatric Otorhinolaryngology, 40(2–3), 107–113.
Shelly, M. P. (2001). Dexmedetomidine: a real innovation or more of the same? British Journal of Anaesthesia, 87(5), 677–678.
Sulton, C., McCracken, C., Simon, H. K., Hebbar, K., Reynolds, J., Cravero, J., … Kamat, P. (2016). Pediatric procedural sedation using dexmedetomidine: A Report From the Pediatric Sedation Research Consortium. Hospital Pediatrics, 6(9), 536–544. https://doi.org/10.1542/hpeds.2015-0280.
Tramèr, M. R., Moore, R. A., & McQuay, H. J. (1997). Propofol and bradycardia: causation, frequency and severity. British Journal of Anaesthesia, 78(6), 642–651.
Vardi, A., Salem, Y., Padeh, S., Paret, G., & Barzilay, Z. (2002). Is propofol safe for procedural sedation in children? A prospective evaluation of propofol versus ketamine in pediatric critical care. Critical Care Medicine, 30(6), 1231–1236.
Weerink, M. A. S., Struys, M. M. R. F., Hannivoort, L. N., Barends, C. R. M., Absalom, A. R., & Colin, P. (2017). Clinical pharmacokinetics and pharmacodynamics of dexmedetomidine. Clinical Pharmacokinectics, 56(8), 893–913. https://doi.org/10.1007/s40262-017-0507-7.
Acknowledgments
The authors have reported that no funding was received for this study.
Author information
Authors and Affiliations
Contributions
KA helped gathering data, conceptualizing and drafting the initial manuscript, and approved the final manuscript as submitted. RL and RL carried out the initial analyses, reviewed and revised the manuscript, and approved the final manuscript as submitted. SSA designed and conceptualized the study, and coordinated and supervised data collection, critically reviewed and approved the final manuscript as submitted.
Corresponding author
Rights and permissions
About this article
Cite this article
Abulebda, K., Louer, R., Lutfi, R. et al. A Comparison of Safety and Efficacy of Dexmedetomidine and Propofol in Children with Autism and Autism Spectrum Disorders Undergoing Magnetic Resonance Imaging. J Autism Dev Disord 48, 3127–3132 (2018). https://doi.org/10.1007/s10803-018-3582-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10803-018-3582-1