Abstract
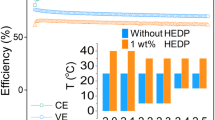
3-Aminopropylphosphonic acid (3APPA) and sodium 3-phosphonopropionate (3PPA-Na) are investigated as organophosphorus compound additives in a positive electrolyte of a vanadium redox flow battery (VRFB) to improve its thermal stability. The precipitation-inhibiting effects of 2 M vanadium electrolytes, including the above additives, are studied by UV–Vis spectroscopy, Raman spectrophotometry, cyclic voltammetry (CV), and charge–discharge cycling test in the redox flow battery. The UV–Vis spectroscopy and Raman spectrophotometry show that no new substances formed in both the V(IV) and V(V) electrolytes with 3APPA and 3PPA-Na additives. Cyclic voltammetry (CV) shows the electrochemical activity and reversibility of the electrolytes with and without the additives. In the flow cell, the discharge capacity retention values in the positive electrolyte after 100 cycles at 45 °C are 56.7, 64.7, and 48.4% in 3PPA-Na, 3APPA, and without the additives, respectively. These results indicate that the 3APPA additive increases the discharge capacity retention to more than 16% compared to the electrolyte without the additive due to the retardation of precipitation at the high temperature.
Graphical abstract









Similar content being viewed by others
References
Halls JE, Hawthornthwaite A, Hepworth RJ et al (2013) Energy Environ Sci 6:1026
Leung P, Li X, Ponce de Leon C et al (2012) RSC Adv 2:10125
Ding C, Zhang H, Li X et al (2013) J Phys Chem Lett 4:1281
Park S-K, Shim J, Yang JH et al (2014) Electrochim Acta 121:321
Skyllas-Kazacos M (1989) All-vanadium redox battery and additives. Patent: WO 89/05526: 15 June 1989, WO 1989005526A1
Rahman F, Skyllas-Kazacos M (1998) J Power Sources 72:105
Wang G, Chen J, Wang X et al (2014) J Energy Chem 23:73
Vijayakumar M, Li L, Graff G et al (2011) J Power Sources 196:3669
Rahman F, Skyllas-Kazacos M (2009) J Power Sources 189:1212
Kazacos M, Cheng M, Skyllas-Kazacos M (1990) J Appl Electrochem 20:463
Madic C, Begun G, Hahn R et al (1984) Inorg Chem 23:469
Kazacos MS, Kazacos M (2000) Stabilised electrolyte solutions, methods of preparation thereof and redox cells and batteries containing stabilised electrolyte solutions. Patent US6143443A
Kazacos MS, Kazacos M (2000) Stabilised electrolyte solutions, methods of preparation thereof and redox cells and batteries containing stabilised electrolyte solutions. Patent EP0729648A1
Kazacos MS, Kazacos M (2003) Stabilized vanadium electrolyte solutions for all-vanadium redox cells and batteries. Patent US6562514B1
Kazacos M, Kazacos MS (2002) High energy density vanadium electrolyte solutions, methods of preparation thereof and all-vanadium redox cells and batteries containing high energy vanadium electrolyte solutions. Patent US6468688B2
Li S, Huang K, Liu S et al (2011) Electrochim Acta 56:5483
Liang X, Peng S, Lei Y et al (2013) Electrochim Acta 95:80
Chang F, Hu C, Liu X et al (2012) Electrochim Acta 60:334
Zhang J, Li L, Nie Z et al (2011) J Appl Electrochem 41:1215
Gresser MJ, Tracey AS, Parkinson KM (1986) J Am Chem Soc 108:6229
Skyllas-Kazacos M, Peng C, Cheng M (1999) Electrochem Solid-State Lett 2:121
Li L, Kim S, Wang W et al (2011) Adv Energy Mater 1:394
Nadeem K, Asem M, Maria SK (2016) ChemElectroChem 3:276
Yeon S-H, So JY, Yun JH et al (2017) J Electrochem Energy Convers Storage 14:041007
Roe S, Menictas C, Skyllas-Kazacos M (2016) J Electrochem Soc 163:A5023
Choi NH, Kwon S-k, Kim H (2013) J Electrochem Soc 160:A973
Kausar N, Howe R, Skyllas-Kazacos M (2001) J Appl Electrochem 31:1327
Acknowledgements
This research was supported by the Energy Efficiency & Resources of the Korea Institute of Energy Technology Evaluation and Planning (KETEP) Grant funded by the Korean government Ministry of Trade, Industry & Energy (MOTIE, No. 20142010102930).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jin, CS., So, JY., Shin, KH. et al. Effect of organophosphorus compound additives for thermal stability on the positive electrolyte of a vanadium redox flow battery. J Appl Electrochem 48, 1019–1030 (2018). https://doi.org/10.1007/s10800-018-1227-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10800-018-1227-x