Abstract
Purpose
To evaluate the effectiveness and safety of the dexamethasone intravitreal implant (DEX-I) in Non-Infectious Uveitis (NIU) in Chinese patients.
Methods
Ninety-one eyes of 77 patients (56 men, 21 women) receiving 130 implant injections for NIU were included. Treatment indication, uveitis diagnosis, best-corrected visual acuity (BCVA), central retinal thickness (CRT), vitreous haze score, intraocular pressure, phakic status, number of injections, time to reinjection, and systemic treatments were collected at baseline, 1 week, 1 month, 3 and 6 months after treatment.
Results
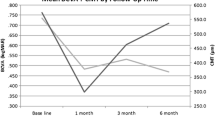
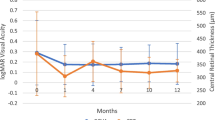
All patients were followed for at least 12 weeks and had a mean follow-up period of 5.1 months (range, 3–14 months) after the first implant. The main treatment indications were macular edema (ME), retinal vasculitis, retinal vasculitis with ME. Sixty-one eyes (67.03%) received only one injection, while 31 eyes (32.97%) received two or more. In eyes that received 2 injections, the mean time to the second injection was 3.83 months and in those that received 3 injections, the mean time to the third injection was 7.5 months. BCVA and CRT significantly improved at 1 week, 1 month, 3 months, and 6 months after treatment. When compared to baseline, the mean prednisone (or equivalent) dosage significantly decreased at 3- and 6-month follow-up evaluations after DEX implantation.14.29% of eyes developed a transient increase in intraocular pressure, and a cataract was removed from 1 phakic eye.
Conclusions
DEX implants, either alone or in combination with common adjunctive NIU treatments, is safe and effective in the treatment of NIU in Chinese patients.


Similar content being viewed by others
References
Gritz DC, Wong IG (2004) Incidence and prevalence of uveitis in Northern California; the Northern California epidemiology of uveitis study. Ophthalmology 111(3):491–500. https://doi.org/10.1016/j.ophtha.2003.06.014 (discussion 500)
Suhler EB, Lloyd MJ, Choi D, Rosenbaum JT, Austin DF (2008) Incidence and prevalence of uveitis in veterans affairs medical centers of the Pacific Northwest. Am J Ophthalmol 146(6):890-896 e898. https://doi.org/10.1016/j.ajo.2008.09.014
Acharya NR, Tham VM, Esterberg E, Borkar DS, Parker JV, Vinoya AC, Uchida A (2013) Incidence and prevalence of uveitis: results from the pacific ocular inflammation study. JAMA Ophthalmol 131(11):1405–1412. https://doi.org/10.1001/jamaophthalmol.2013.4237
Miserocchi E, Fogliato G, Modorati G, Bandello F (2013) Review on the worldwide epidemiology of uveitis. Eur J Ophthalmol 23(5):705–717. https://doi.org/10.5301/ejo.5000278
Durrani OM, Tehrani NN, Marr JE, Moradi P, Stavrou P, Murray PI (2004) Degree, duration, and causes of visual loss in uveitis. Br J Ophthalmol 88(9):1159–1162. https://doi.org/10.1136/bjo.2003.037226
Thorne JE, Skup M, Tundia N, Macaulay D, Revol C, Chao J, Joshi A, Dick AD (2016) Direct and indirect resource use, healthcare costs and work force absence in patients with non-infectious intermediate, posterior or panuveitis. Acta Ophthalmol 94(5):e331-339. https://doi.org/10.1111/aos.12987
Wakefield D, Chang JH (2005) Epidemiology of uveitis. Int Ophthalmol Clin 45(2):1–13. https://doi.org/10.1097/01.iio.0000155938.83083.94
Deschenes J, Murray PI, Rao NA, Nussenblatt RB, International Uveitis Study G (2008) International uveitis study group (IUSG): clinical classification of uveitis. Ocul Immunol Inflamm 16(1):1–2. https://doi.org/10.1080/09273940801899822
Tomkins-Netzer O, Taylor SR, Lightman S (2012) Corticosteroid-sparing agents: new treatment options. Dev Ophthalmol 51:47–56. https://doi.org/10.1159/000336186
Sella R, Oray M, Friling R, Umar L, Tugal-Tutkun I, Kramer M (2015) Dexamethasone intravitreal implant (Ozurdex(R)) for pediatric uveitis. Graefes Arch Clin Exp Ophthalmol 253(10):1777–1782. https://doi.org/10.1007/s00417-015-3124-x
Lightman S, Belfort R Jr, Naik RK, Lowder C, Foster CS, Rentz AM, Cui H, Whitcup SM, Kowalski JW, Revicki DA (2013) Vision-related functioning outcomes of dexamethasone intravitreal implant in noninfectious intermediate or posterior uveitis. Invest Ophthalmol Vis Sci 54(7):4864–4870. https://doi.org/10.1167/iovs.12-10981
Lowder C, Belfort R Jr, Lightman S, Foster CS, Robinson MR, Schiffman RM, Li XY, Cui H, Whitcup SM, Ozurdex HSG (2011) Dexamethasone intravitreal implant for noninfectious intermediate or posterior uveitis. Arch Ophthalmol 129(5):545–553. https://doi.org/10.1001/archophthalmol.2010.339
Moisseiev E, Goldstein M, Waisbourd M, Barak A, Loewenstein A (2013) Long-term evaluation of patients treated with dexamethasone intravitreal implant for macular edema due to retinal vein occlusion. Eye (Lond) 27(1):65–71. https://doi.org/10.1038/eye.2012.226
Fabiani C, Vitale A, Lopalco G, Iannone F, Frediani B, Cantarini L (2016) Different roles of TNF inhibitors in acute anterior uveitis associated with ankylosing spondylitis: state of the art. Clin Rheumatol 35(11):2631–2638. https://doi.org/10.1007/s10067-016-3426-3
Fardeau C, Champion E, Massamba N, LeHoang P (2016) Uveitic macular edema. Eye (Lond) 30(10):1277–1292. https://doi.org/10.1038/eye.2016.115
Fabiani C, Alio JL (2015) Local (topical and intraocular) therapy for ocular Adamantiades-Behcet’s disease. Curr Opin Ophthalmol 26(6):546–552. https://doi.org/10.1097/ICU.0000000000000210
Jabs DA, Nussenblatt RB, Rosenbaum JT, Standardization of Uveitis Nomenclature Working G (2005) Standardization of uveitis nomenclature for reporting clinical data Results of the first international workshop. Am J Ophthalmol 140(3):509–516. https://doi.org/10.1016/j.ajo.2005.03.057
Nobre-Cardoso J, Champion E, Darugar A, Fel A, Lehoang P, Bodaghi B (2017) Treatment of non-infectious uveitic macular edema with the intravitreal dexamethasone implant. Ocul Immunol Inflamm 25(4):447–454. https://doi.org/10.3109/09273948.2015.1132738
Habot-Wilner Z, Sorkin N, Goldenberg D, Goldstein M (2015) Bilateral effect of unilateral dexamethasone intravitreal implant in a case of noninfectious uveitic macular edema and vitritis. Retin Cases Brief Rep 9(2):151–153. https://doi.org/10.1097/ICB.0000000000000122
Sharma A, Sheth J, Madhusudan RJ, Sundaramoorthy SK (2013) Effect of intravitreal dexamethasone implant on the contralateral eye: a case report. Retin Cases Brief Rep 7(3):217–219. https://doi.org/10.1097/ICB.0b013e31828993a1
Funding
This study was funded by the National Natural Science Foundation of China (81300752), the Jilin Province Science and Technology Development Plan Project (20200201333JC) and the Jilin Province Health Special Project (2020SCZT058).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest, or non-financial interest in the subject matter or materials discussed in this manuscript.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Ethics Committee of the Second Hospital of Jilin University and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zeng, S., Yang, L., Bai, F. et al. Intravitreal dexamethasone implant for noninfectious uveitis in Chinese patients. Int Ophthalmol 42, 2063–2069 (2022). https://doi.org/10.1007/s10792-021-02204-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-021-02204-2