Abstract
Introduction
Topical beta-blockers are a proven and safe medication used in the treatment of glaucoma and ocular hypertension. Local and systemic side effects are, however, well documented. Systemic side effects can include severe cardio-respiratory impairment, endocrine dysfunction, as well as headache and hyper-somnolence. Disorders involving fibrosis such as Peyronie’s disease, Dupuytren’s contracture, and retroperitoneal fibrosis are rarely described in the literature.
Case report
We describe a case of a 55-year-old male patient undergoing treatment for ocular hypertension with topical timolol 0.25% to both eyes, who subsequently developed both Peyronie’s disease and Dupuytren’s contracture. To our knowledge, this is the first case of Dupuytren’s contracture secondary to the use of topical timolol.
Conclusions
Whilst uncommon, beta-blockers are known to be associated with such fibrotic conditions and have been reported with the use of topical timolol. This case serves to increase the current understanding of this association.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Topical beta-blockers were introduced for the management of glaucoma in the 1970s and remain a vital component in the management of glaucoma worldwide. Soon after the introduction of topical beta-blockers, however, systemic side effects soon became well documented. Other than cardio-pulmonary complications, central nervous system (CNS) disturbances were reported, as well as reduced exercise tolerance [1].
Less well described and rarely reported, beta-blockers have been associated with fibrosing disease such as retroperitoneal fibrosis, Peyronie’s disease, and Dupuytren’s contracture [2,3,4,5]. Dupuytren’s contracture is a deforming, fibrotic condition of the palmar fascia, whereas Peyronie’s disease is a fibroblastic proliferative disorder of the penile tunica albuginea.
Only one previous case report describing Peyronie’s disease in association with topical beta-blockers exists [6]. No previous reports have been published associating Dupuytren’s contracture with topical timolol. To our knowledge, we describe the first case of an adult male developing both Peyronie’s disease and Dupuytren’s contracture shortly after commencing timolol 0.25% for the treatment of ocular hypertension (OHT).
Case report
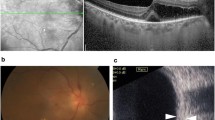
A 55-year-old male diagnosed with OHT was commenced on topical timolol 0.25% twice a day to both eyes. At routine review, it was divulged that he had developed Peyronie’s disease, as well as fibrotic bands representing early Dupuytren’s contracture, as shown in Fig. 1. The development of both of these conditions had occurred within 3 months of starting topical timolol, with no prior history described.
Of note the patient was not a smoker, was not known to be diabetic, and took no other regular medications that may have contributed towards the development of these conditions. Specifically he was not on anticonvulsants and had no history of immunosuppression. Additionally, there was no history of excess alcohol intake.
Both the Dupuytren’s contracture and Peyronie’s disease were interfering significantly with his quality of life. The topical beta-blocker preparation was therefore stopped, and the patient was referred to both a urological surgeon and a hand surgeon, though no active intervention was subsequently sought.
In place of timolol 0.25%, the patient was commenced on a prostaglandin analogue as well as a topical carbonic anhydrase inhibitor. The patient experienced no worsening of either Dupuytren’s contracture or Peyronie’s disease, though symptoms did not resolve following cessation of timolol.
Discussion
The use of beta-blockers, both topical and oral, has been strongly associated with fibrosing disease. There are numerous case reports linking beta-blockers to retroperitoneal fibrosis [2,3,4]. Additionally, topical beta-blockers have also been reported to contribute to the development of Peyronie’s disease [6,7,8].
The previous report of Peyronie’s disease secondary to the use of timolol 0.25% eye drops had similarities to our case, particularly in relation to the timeline of development of the condition. Unlike this case, however, the previous report did not describe the development of Dupuytren’s contracture.
The mechanism of developing fibrosing disease secondary to beta-blockers is incompletely understood, though putative mechanisms do exist. It has been shown that endogenous beta adrenergic agonists inhibit fibrosis through reduced fibroblast proliferation. This process is inhibited by exogenous beta-blocking preparations, which leads to fibrosis of tissue and replacement of elastic tissue with inelastic collagen fibres [6].
This case report adds to the growing body of evidence that beta-blockers, including topical preparations, are implicated in the development of fibrotic disease processes such as Peyronie’s disease, Dupuytren’s contracture, and retroperitoneal fibrosis. It is prudent that patients are counselled thoroughly regarding possible systemic side effects of topical beta-blockers prior to commencement of treatment.
References
Steward WC, Castelli WP (1996) Systemic side effects of topical beta-adrenergic blockers. Clin Cardiol 19:691–697
Delorme E, Vial T, Rabilloud M et al (1990) Retroperitoneal fibrosis with eye-drops containing timolol. J Urol (Paris) 96(8):449–451
Rimmer E, Richens A, Forster ME et al (1983) Retroperitoneal fibrosis associated with timolol. Lancet 1(8319):300
Thompson J, Julian DG (1982) Retroperitoneal fibrosis associated with metoprolol. Br Med J (Clin Res Ed) 284(6309):83–84
Kristensen BO (1979) Labetalol-induced Peyronie’s disease? A case report. Acta Med Scand 206(6):511–512
Ross JJ, Rahman I, Walters RF (2006) Peyronie’s disease following long-term use of topical timolol. Eye (Lond) 20(8):974–976
Hambleton G, Macleod AF, Binless JT (1983) Is Peyronie’s disease iatrogenic? J R Coll Gen Pract 33(246):60
Osborne DR (1977) Propranolol and Peyronie’s disease. Lancet 1(8021):1111
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No conflicts of interest to declare.
Ethical approval
This article does not contain any studies with human participants performed by any of the authors.
Informed consent
Informed consent obtained from patient described in study.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Oliphant, H., Gouws, P. Peyronie’s disease and Dupuytren’s contracture secondary to topical timolol. Int Ophthalmol 39, 683–685 (2019). https://doi.org/10.1007/s10792-018-0837-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-018-0837-y