Abstract
Purpose
The aim of this study was to evaluate the effect of intravitreal injection of aflibercept (IVA) on blood coagulation tests in neovascular age-related macular degeneration (AMD) patients.
Methods
Thirty-four patients with neovascular AMD (study group) and 32 healthy individuals (control group) were enrolled. Prothrombin time (PT) and activated partial thromboplastin time (aPTT) were measured at different times in patients with neovascular AMD.
Results
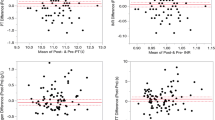
The levels of PT and aPTT after IVA were decreased at 1 month after the first injection and 1 month after the second injection compared to the baseline measurement in the study group.
Conclusions
IVA may cause a decrease in the levels of PT and aPTT at 1 month after the first injection and 1 month after the second injection although these results are not statistically significant in our study.
Similar content being viewed by others
References
Klein R, Wang Q, Klein BE et al (1995) The relationship of age-related maculopathy, cataract, and glaucoma to visual acuity. Invest Ophthalmol Vis Sci 36:182–191
Munoz B, West SK, Rubin GS et al (2000) Causes of blindness and visual impairment in a population of older Americans: the salisbury eye evaluation study. Arch Ophthalmol 118:819–825
Attebo K, Mitchell P, Smith W (1996) Visual acuity and the causes of visual loss in Australia: the blue mountains eye study. Ophthalmology 103:357–364
Wroblewski JJ, Wells JA, Adamis AP et al (2009) Central retinal vein occlusion study group. Pegaptanib sodium for macular edema secondary to central retinal vein occlusion. Arch Ophthalmol 127:374–380
Ehlers JP, Decroos FC, Fekrat S (2001) Intravitreal bevacizumab for macular edema secondary to branch retinal vein occlusion. Retina 31:1856–1862
Campochiaro PA, Heier JS, Feiner L et al (2010) BRAVO investigators. Ranibizumab for macular edema following branch retinal vein occlusion: six-month primary end point results of a phase III study. Ophthalmology 117:1102–1112
Campochiaro PA, Clark WL, Boyer DS et al (2015) Intravitreal aflibercept for macular edema following branch retinal vein occlusion: the 24-week results of the VIBRANT study. Ophthalmology 122:538–544
Evoy KE, Abel SR (2013) Aflibercept: newly approved for the treatment of macular edema following central retinal vein occlusion. Ann Pharmacother 47:819–827
Sang DN, Damore PA (2008) Is blockade of vascular endothelial growth factor beneficial for all types of diabetic retinopathy? Diabetologia 51:1570–1573
Von Hanno T, Kinge B, Fossen K (2010) Retinal artery occlusion following intravitreal anti-VEGF therapy. Acta Ophthalmol 88:263–266
Stewart MW (2012) The expanding role of vascular endothelial growth factor inhibitors in ophthalmology. Mayo Clin Proc 87:77–88
Costagliola C, Agnifili L, Arcidiacono B et al (2012) Systemic thromboembolic adverse events in patients treated with intravitreal anti-VEGF drugs for neovascular age-related macular degeneration. Expert Opin Biol Ther 12:1299–1313
Scappaticci FA, Skillings JR, Holden SN et al (2007) Arterial thromboembolic events in patients with metastatic carcinoma treated with chemotherapy and bevacizumab. J Natl Cancer Inst 99:1232–1239
Yoshida I, Shiba T, Taniguchi H et al (2014) Evaluation of plasma vascular endothelial growth factor levels after intravitreal injection of ranibizumab and aflibercept for exudative age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol 252:1483–1489
Tripodi A, Mannucci PM (2006) Activated partial thromboplastin time (APTT). New indications for an old test? J Thromb Haemost 4(4):750–751
Favaloro EJ, Lippi G, Adcock DM (2008) Preanalytical and postanalytical variables in haemostasis: the leading causes of diagnostic error? Semin Thromb Hemost 34(7):612–634
Lippi G, Favaloro EJ (2008) Activated partial thromboplastin time: new tricks for an old dogma. Semin Thromb Hemost 34(7):604–611
Tripodi A, Chantarangkul V, Martinelli I et al (2004) A shortened activated partial thromboplastin time is associated with the risk of venous thromboembolism. Blood 104(12):3631–3634
Senthil M, Chaudhary P, Smith DD et al (2014) A shortened activated partial thromboplastin time predicts the risk of catheter-associated venous thrombosis in cancer patients. Thromb Res 134(1):165–168
Korte W, Clarke S, Lefkowiz JB (2000) Short activated partial thromboplastin times are related to increased thrombin generation and an increased risk for thromboembolism. Am J Clin Pathol 113(1):123–127
Legnani C, Mattarozzi S, Cini M et al (2006) Abnormally short activated partial thromboplastin time values are associated with increased risk of recurrence of venous thromboembolism after oral anticoagulation withdrawal. Br J Haematol 134(2):227–232
Qian J, Jiang YR (2011) Decreased prothrombin time after intravitreal bevacizumab in the early period in patients with proliferative diabetic retinopathy. Acta Ophthalmol 89:332–335
Yi Z, Chen C, Su Y et al (2015) Changes in clotting time, plasma fibrinogen levels, and blood viscosity after administration of ranibizumab for treatment of choroidal neovascularization. Curr Eye Res 40(11):1166–1171
Molenaar PJ, Leyte A (2011) Pre-acquisition system assessment of the Sysmex Coagulation System CS-2100i and comparison with end-user verification; a model fort he regional introduction of new analysers and methods. Clin Chem Lab Med 49(9):1479–1489
Heier JS, Brown DM, Chong V et al (2012) Intravitreal aflibercept (VEGF trap-eye) in wet age-related macular degeneration. Ophthalmology 119:2537–2548
Schmidt-Erfurth U, Kaiser PK, Korobelnik JF et al (2014) Intravitreal aflibercept injection for neovascular age-related macular degeneration: ninety-six-week results of the VIEW studies. Ophthalmology 121:193–201
Ferrara N (2004) Vascular endothelial growth factor: basic science and clinical progress. Endocr Rev 25:581–611
Zakarija A, Soff G (2005) Update on angiogenesis inhibitors. Curr Opin Oncol 17:578–583
Ng EWM, Adamis AP (2005) Targeting angiogenesis, the underlying disorder in neovascular age-related macular degeneration. Can J Ophthalmol 40:353–368
Ferrara N (2001) Role of vascular endothelial growth factor in regulation of physiological angiogenesis. Am J Physiol Cell Physiol 280:1358–1366
Schmid MK, Bachmann LM, Fas L et al (2015) Efficacy and adverse events of aflibercept, ranibizumab and bevacizumab in age-related macular degeneration: a trade-off analysis. Br J Ophthalmol 99:141–146
Ueta T, Noda Y, Toyama T et al (2014) Systemic vascular safety of ranibizumab for age-related macular degeneration: systematic review and meta-analysis of randomized trials. Ophthalmology 121:2193–2203
Thompson SG, Kienast J, Pyke SD et al (1995) Hemostatic factors and the risk of myocardial infarction or sudden death in patients with angina pectoris. European concerted action on thrombosis and disabilities angina pectoris study group. N Engl J Med 332:635–641
Tan JS, Wang JJ, Liew G et al (2008) Age related macular degeneration and mortality from cardiovascular disease or stroke. Br J Ophthalmol 92:509–512
Sun C, Klein R, Wong TY (2009) Age-related macular degeneration and risk of coronary heart disease and stroke: the cardiovascular health study. Ophthalmology 116:1913–1919
Hu CC, Ho JD, Lin HC (2010) Neovascular age-related macular degeneration and the risk of stroke: a 5-year population-based follow-up study. Stroke 41:613–617
Ozkan B, Karabas LV, Altıntas O et al (1995) Plasma antiphospholipid antibody levels in age-related macular degeneration. Can J Ophthalmol 47:264–268
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no financial or proprietary interests.
Ethical approval
The study protocol followed the principles in the declaration of Helsinki.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Altinkaynak, H., Kars, M.E., Kurkcuoglu, P.Z. et al. Blood coagulation parameters after intravitreal injection of aflibercept in patients with neovascular age-related macular degeneration. Int Ophthalmol 38, 2397–2402 (2018). https://doi.org/10.1007/s10792-017-0741-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-017-0741-x