Abstract
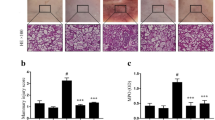
Rutin, found widely in traditional Chinese medicine materials, is used to treat eye swelling and pain, hypertension, and hyperlipidemia. In the present study, a mouse mastitis model induced by lipopolysaccharide (LPS) was established to explore rutin’s inhibitory mechanism on mastitis via nuclear factor kappa B (NF-κB) inflammatory signaling and the relationship between NF-κB signaling and endoplasmic reticulum (ER) stress. Mice were divided into six groups: Control group, LPS model group, LPS + rutin (25, 50, and 100 mg/kg) and LPS + dexamethasone (DEX) group. DEX, rutin, and PBS (control and LPS groups) were administered 1 h before and 12 h after perfusion of LPS. After LPS stimulation for 24 h, to evaluate rutin’s therapeutic effect on mastitis, the mammary tissues of each group were collected to detect histopathological injury, tumor necrosis factor alpha (TNF-α), interleukin (IL)-1β, and IL-6 mRNA and protein levels; and glucose-regulated protein, 78 kDa (GRP78) protein levels. The protein and mRNA levels of TNF-α, IL-1β, and IL-6 in the LPS + rutin group were significantly lower than those in the LPS model group. Similarly, p50/p105, phosphorylated (p)-p65/p65 and p-inhibitor of nuclear factor kappa b kinase subunit beta (p-IKKβ)/IKKβ ratios in the LPS + rutin group (50 mg/kg) and LPS + rutin group (100 mg/kg) decreased significantly. GRP78 protein expression was significantly higher in LPS + rutin group (100 mg/kg). The structure of mammary tissue became gradually more intact and vacuolization of acini decreased as the rutin concentration increased. The nuclear quantity of p65 in the LPS + rutin group decreased significantly in a rutin dose-dependent manner. Rutin had an anti-inflammatory effect in the LPS-induced mouse mastitis model, manifested by inhibition of NF-κB pathway activation and attenuation of ER stress.









Similar content being viewed by others
References
Albert S, Baldwin J (1996) THE NF-κB AND IκB PROTEINS: new discoveries and insights. Annu Rev Immunol 14:649–683
Alluwaimi AM (2004) The cytokines of bovine mammary gland: prospects for diagnosis and therapy. Res Vet Sci 77:211–222. https://doi.org/10.1016/j.rvsc.2004.04.006
Bannerman DD, Chockalingam A, Paape MJ, Hope JC (2005) The bovine innate immune response during experimentally-induced Pseudomonas aeruginosa mastitis. Vet Immunol Immunopathol 107:201–215. https://doi.org/10.1016/j.vetimm.2005.04.012
Chen H, Mo X, Yu J, Huang Z (2013) Alpinetin attenuates inflammatory responses by interfering toll-like receptor 4/nuclear factor kappa B signaling pathway in lipopolysaccharide-induced mastitis in mice. Int Immunopharmacol 17:26–32. https://doi.org/10.1016/j.intimp.2013.04.030
Clarkson RW, Watson CJ (1999) NF-kappaB and apoptosis in mammary epithelial cells. J Mammary Gland Biol Neoplasia 4:165–175
Crispie F (2008) Intramammary infusion of a live culture of Lactococcus lactis for treatment of bovine mastitis: comparison with antibiotic treatment in field trials. J Dairy Res 75:365–373
Engel MA, Kellermann CA, Burnat G, Hahn EG, Rau T, Konturek PC (2010) Mice lacking cannabinoid CB1-, CB2-receptors or both receptors show increased susceptibility to trinitrobenzene sulfonic acid (TNBS)-induced colitis. J Physiol Pharmacol 61:89–97
Gao M, Ma Y, Liu D (2013) Rutin suppresses palmitic acids-triggered inflammation in macrophages and blocks high fat diet-induced obesity and fatty liver in mice. Pharm Res 30:2940–2950. https://doi.org/10.1007/s11095-013-1125-1
Garg AD, Kaczmarek A, Krysko O, Vandenabeele P, Krysko DV, Agostinis P (2012) ER stress-induced inflammation: does it aid or impede disease progression? Trends Mol Med 18:589–598. https://doi.org/10.1016/j.molmed.2012.06.010
Gautam R, Singh M, Gautam S, Rawat JK, Saraf SA, Kaithwas G (2016) Rutin attenuates intestinal toxicity induced by Methotrexate linked with anti-oxidative and anti-inflammatory effects. BMC Complement Altern Med 16:99. https://doi.org/10.1186/s12906-016-1069-1
Ghiasi M, Azadnia A, Arabieh M, Zahedi M (2012) Protective effect of rutin (vitamin p) against heme oxidation: a quantum mechanical approach. Comput Theor Chem 996:28–36
Ghosh S, Hayden MS (2008) New regulators of NF-kappaB in inflammation. Nat Rev Immunol 8:837–848. https://doi.org/10.1038/nri2423
Ghosh S, Karin M (2002) Missing pieces in the NF-kappaB puzzle. Cell 109(Suppl):S81–S96
Grivennikov SI, Greten FR, Karin M (2010) Immunity, inflammation, and cancer. Cell 140:883–899. https://doi.org/10.1016/j.cell.2010.01.025
Guo YF, Xu NN, Sun W, Zhao Y, Li CY, Guo MY (2017) Luteolin reduces inflammation in Staphylococcus aureus-induced mastitis by inhibiting NF-κB activation and MMPs expression. Oncotarget 8:28481–28493
Hayden MS, Ghosh S (2004) Signaling to NF-kappaB. Genes Dev 18:2195–2224. https://doi.org/10.1101/gad.1228704
Hogeveen H, Huijps K, Lam TJ (2011) Economic aspects of mastitis: new developments. N Z Vet J 59:16–23. https://doi.org/10.1080/00480169.2011.547165
Hotamisligil GS (2010) Endoplasmic reticulum stress and the inflammatory basis of metabolic disease. Cell 140:900–917. https://doi.org/10.1016/j.cell.2010.02.034
Kerdudo A, Dingas A, Fernandez X, Faure C (2014) Encapsulation of rutin and naringenin in multilamellar vesicles for optimum antioxidant activity. Food Chem 159:12–19. https://doi.org/10.1016/j.foodchem.2014.03.005
Lai JL, Liu YH, Peng YC, Ge P, He CF, Liu C, Chen YY, Guo AZ, Hu CM (2017) Indirubin treatment of lipopolysaccharide-induced mastitis in a mouse model and activity in mouse mammary epithelial cells. Mediat Inflamm 2017:3082805. https://doi.org/10.1155/2017/3082805
Lee AS (2005) The ER chaperone and signaling regulator GRP78/BiP as a monitor of endoplasmic reticulum stress. Methods 35:373
Lee AS (2007) GRP78 induction in cancer: therapeutic and prognostic implications. Cancer Res 67:3496–3499. https://doi.org/10.1158/0008-5472.CAN-07-0325
Li F, Wang W, Cao Y, Liang D, Zhang W, Zhang Z, Jiang H, Guo M, Zhang N (2014) Inhibitory effects of astragalin on lipopolysaccharide-induced inflammatory response in mouse mammary epithelial cells. J Surg Res 192:573–581. https://doi.org/10.1016/j.jss.2014.05.059
Miao JF, Zhu YM, Gu BB, Wang XB, Zou SX, Deng YE (2007) Evaluation of the changes of immune cells during lipopolysaccharide-induced mastitis in rats. Cytokine 40:135–143. https://doi.org/10.1016/j.cyto.2007.08.012
Moutinho MSS, Aragao S, Carmo D, Casaca F, Silva S, Ribeiro J, Sousa H, Pires I, Queiroga F, Colaco B, Medeiros R, Oliveira PA, Lopes C, Bastos M, DA Costa RMG (2018) Curcumin and rutin down-regulate cyclooxygenase-2 and Reduce tumor-associated Inflammation in HPV16-transgenic mice. Anticancer Res 38:1461–1466. https://doi.org/10.21873/anticanres.12371
Nakajima S, Kato H, Takahashi S, Johno H, Kitamura M (2011) Inhibition of NF-κB by MG132 through ER stress-mediated induction of LAP and LIP. FEBS Lett 585:2249
Reshi AA, Husain I, Bhat SA, Rehman MU, Razak R, Bilal S, Mir MR (2015) Bovine mastitis as an evolving disease and its impact on the dairy industry. Solid State Phenom 7:194–2262375
So H, Kim H, Kim Y, Kim E, Pae HO, Chung HT, Kim HJ, Kwon KB, Lee KM, Lee HY (2008) Evidence that cisplatin-induced auditory damage is attenuated by downregulation of pro-inflammatory cytokines via Nrf2/HO-1. J Assoc Res Otolaryngol 9:290–306
Song X, Wang T, Zhang Z, Jiang H, Wang W, Cao Y, Zhang N (2015) Leonurine exerts anti-inflammatory effect by regulating inflammatory signaling pathways and cytokines in LPS-induced mouse mastitis. Inflammation 38:79–88. https://doi.org/10.1007/s10753-014-0009-9
Taniguchi F, Uegaki T, Nakamura K, Mon KY, Harada T, Ohbayashi T, Harada T (2018) Inhibition of IAP (inhibitor of apoptosis) proteins represses inflammatory status via nuclear factor-kappa B pathway in murine endometriosis lesions. Am J Reprod Immunol 79:e12780. https://doi.org/10.1111/aji.12780
Wang Y, Zarlenga DS, Paape MJ, Dahl GE (2002) Recombinant bovine soluble CD14 sensitizes the mammary gland to lipopolysaccharide. Vet Immunol Immunopathol 86:115–124
Wang J, Zhao L, Wei Z, Zhang X, Wang Y, Li F, Fu Y, Liu B (2018) Inhibition of histone deacetylase reduces lipopolysaccharide-induced-inflammation in primary mammary epithelial cells by regulating ROS-NF-кB signaling pathways. Int Immunopharmacol 56:230
Yamazaki H, Hiramatsu N, Hayakawa K, Tagawa Y, Okamura M, Ogata R, Huang T, Nakajima S, Yao J, Paton AW (2009) Activation of the Akt-NF-κB pathway by subtilase cytotoxin through the ATF6 branch of the unfolded protein response. J Immunol 183:1480–1487
Yang Z, Yin R, Cong Y, Yang Z, Zhou E, Wei Z, Liu Z, Cao Y, Zhang N (2014) Oxymatrine lightened the inflammatory response of LPS-induced mastitis in mice through affecting NF-κB and MAPKs signaling pathways. Inflammation 37:2047–2055
Zhang K, Kaufman RJ (2008) From endoplasmic-reticulum stress to the inflammatory response. Nature 454:455–462. https://doi.org/10.1038/nature07203
Zhou E, Fu Y, Wei Z, Cao Y, Zhang N, Yang Z (2014) Cepharanthine attenuates lipopolysaccharide-induced mice mastitis by suppressing the NF-κB signaling pathway. Inflammation 37:331–337
Acknowledgements
This work was supported by the Anhui Agricultural University College Students Innovation and Entrepreneurship Training Program Innovative Training Project Foundation (Grant Numbers 201710364061 and 201710364017).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Su, S., Li, X., Li, S. et al. Rutin protects against lipopolysaccharide-induced mastitis by inhibiting the activation of the NF-κB signaling pathway and attenuating endoplasmic reticulum stress. Inflammopharmacol 27, 77–88 (2019). https://doi.org/10.1007/s10787-018-0521-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-018-0521-x