Abstract
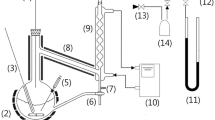
In this work, the isothermal vapor–liquid equilibrium (VLE) of trans-1,3,3,3-tetrafluoropropene (HFO-1234ze(E)) and 1,1,1,3,3-pentafluoropropane (HFC245fa) binary system, along with the saturated vapor pressures HFO-1234ze(E) and HFC-245fa, were measured by static-analytic apparatus. The VLE measurements of HFO-1234ze(E) + HFC-245fa binary system are performed at temperatures from (293.28 to 333.09) K, while the measurements for vapor pressures of HFO1234ze(E) and HFC-245fa were conducted in the temperature range of (263.08 to 363.01) K. The vapor pressures were correlated by the Peng-Robinson (PR) equation of state (EoS) associated with the Mathias-Copeman (MC) alpha function, and the VLE data were correlated to the PRMC-vdW model where the MC alpha function and the van der Waals (vdW) mixing rules were combined with the PR EoS. The experimental VLE data were compared with the predictive E-PPR78 model. The modeling results of the PRMC-vdW model had good agreement with the experimental VLE data, while the E-PPR78 model shows negative bias in the prediction of equilibrium pressures. The experimental VLE data were also compared with literature data for consistency checks. The two sets of VLE data were basically consistent, while a discrepancy was found when analyzing the relationship between binary interaction parameter kij and temperature.










Similar content being viewed by others
Abbreviations
- HFCs:
-
Hydrofluorocarbons
- VLE:
-
Vapor–liquid equilibrium
- HFOs:
-
Hydrofluoro-olefins
- vdW:
-
Van der Waals mixing rules
- PR:
-
Peng-Robinson equation of state
- MC:
-
Mathias-Copeman alpha function
- MRDP :
-
Average relative deviation of pressure
- MADy :
-
Average absolute deviation of vapor-composition
- HFC-245fa:
-
1,1,1,3,3-Pentafluoropropane
- HFO-1234ze(E):
-
Trans-1,3,3,3-tetrafluoropropene
- EoS:
-
Equation of state
- a :
-
Energy parameter of the equation of state
- b :
-
Covolume parameter of the equation of state
- GE :
-
Excess Gibbs energy
- q :
-
Coefficient of MHV2 mixing rule
- N :
-
Number of data points
- p :
-
Pressure (MPa)
- R :
-
Molar gas constant (8.314472 J·mol−1·K−1)
- T :
-
Temperature (K)
- u :
-
Combined uncertainty
- U :
-
Expanded uncertainty
- x :
-
Liquid phase mole fraction
- y :
-
Vapor phase mole fraction
- n :
-
Molar mass
- m1, 2, 3 :
-
Adjustable parameters
- k ij :
-
Binary interaction parameter
- OF:
-
Objective function
- α :
-
Temperature-dependent alpha function
- τ :
-
Dimensionless interaction parameters
- δ :
-
Relative deviation
- Δ :
-
Absolute deviation
- ω:
-
Acentric factor
- i, j:
-
Molecular species
- c:
-
Critical property
- exp:
-
Experimental
- cal:
-
Calculated
References
M.O. McLinden, J.S. Brown, R. Brignoli, A.F. Kazakov, P.A. Domanski, Limited options for low-global-warming-potential refrigerants. Nat. Commun. 8, 1–9 (2017). https://doi.org/10.1038/ncomms14476
S. Bobbo, G. Di Nicola, C. Zilio, J.S. Brown, L. Fedele, Low GWP halocarbon refrigerants: a review of thermophysical properties. Int. J. Refrig. 90, 181–201 (2018). https://doi.org/10.1016/j.ijrefrig.2018.03.027
C. Coquelet, D. Richon, Experimental determination of phase diagram and modeling: application to refrigerant mixtures. Int. J. Refrig. 32, 1604–1614 (2009). https://doi.org/10.1016/j.ijrefrig.2009.03.013
M. Hillert, Phase Equilibria Phase Diagrams and Phase Transformations (Cambridge University Press, Cambridge, 2007). https://doi.org/10.1017/cbo9780511812781
W. Zhang, Z.Q. Yang, J. Lu, J. Lu, Vapor pressures of 2-chloro-3,3,3-trifluoropropene (HCFO-1233xf). J. Chem. Eng. Data. 58, 2307–2310 (2013). https://doi.org/10.1021/je400407z
Z.Q. Yang, L.G. Kou, W. Mao, J. Lu, W. Zhang, J. Lu, Isothermal vapor-liquid equilibrium for the binary system of 2,3,3,3-tetrafluoropropene and 2-chloro-1,1,1,2-tetrafluoropropane. J. Chem. Eng. Data. 60, 1153–1156 (2015). https://doi.org/10.1021/je501130r
Z.Q. Yang, L.G. Kou, W. Mao, J. Lu, W. Zhang, J. Lu, Experimental study of saturated pressure measurements for 2,3,3,3-tetrafluoropropene (HFO-1234yf) and 2-chloro-1,1,1,2-tetrafluoropropane (HCFC-244bb). J. Chem. Eng. Data. 59, 157–160 (2014). https://doi.org/10.1021/je400970y
Z.Q. Yang, L.G. Kou, J. Lu, W. Zhang, W. Mao, J. Lu, Isothermal vapor-liquid equilibria measurements for binary systems of 2,3,3,3-tetrafluoropropene (HFO-1234yf) + 2-chloro-3,3,3-trifluoropropene (HCFO-1233xf) and 2-chloro-3,3,3-trifluoropropene (HCFO-1233xf) + 2-chloro-1,1,1,2-tetrafluoropropane (HCFC-244. Fluid Phase Equilib. 414, 143–148 (2016). https://doi.org/10.1016/j.fluid.2016.01.027
Z. Qiang Yang, L. Gang Kou, S. Han, C. Li, Z. Jun Hao, W. Mao, W. Zhang, J. Lu, Vapor-liquid equilibria of 2,3,3,3-tetrafluoropropene (HFO-1234yf) + 1,1,1,2,2-pentafluoropropane (HFC-245cb) system. Fluid Phase Equilib. 427, 390–393 (2016). https://doi.org/10.1016/j.fluid.2016.07.031
A. Mota-Babiloni, J. Navarro-Esbrí, Á. Barragán, F. Molés, B. Peris, Drop-in energy performance evaluation of R1234yf and R1234ze(E) in a vapor compression system as R134a replacements. Appl. Therm. Eng. 71, 259–265 (2014). https://doi.org/10.1016/j.applthermaleng.2014.06.056
G.M. Rusch, A. Tveit, H. Muijser, M.M. Tegelenbosch-Schouten, G.M. Hoffman, The acute, genetic, developmental and inhalation toxicology of trans-1,3,3,3-tetrafluoropropene (HFO-1234ze). Drug Chem. Toxicol. 36, 170–180 (2013). https://doi.org/10.3109/01480545.2012.661738
H. Groult, F.R. Leroux, A. Tressaud, Modern Synthesis Processes and Reactivity of Fluorinated Compounds: Progress in Fluorine Science (Elsevier, London, 2017)
W. Mao, Y. Bai, Z. Jia, Z. Yang, Z. Hao, J. Lu, Highly efficient gas-phase dehydrofluorination of 1,1,1,3,3-pentafluoropropane to 1,3,3,3-tetrafluoropropene over mesoporous nano-aluminum fluoride prepared from a polyol mediated sol-gel process. Appl. Catal. A Gen. 564, 147–156 (2018). https://doi.org/10.1016/j.apcata.2018.07.028
T. Katsuyuki, Measurements of vapor pressure and saturated liquid density for HFO-1234ze(E) and HFO-1234ze(Z). J. Chem. Eng. Data. 61, 1645–1648 (2016). https://doi.org/10.1021/acs.jced.5b01039
Y. Higashi, K. Tanaka, T. Ichikawa, Critical parameters and saturated densities in the critical region for trans -1,3,3,3-Tetrafluoropropene (HFO-1234ze(E)). J. Chem. Eng. Data. 55, 1594–1597 (2010). https://doi.org/10.1021/je900696z
K. Tanaka, G. Takahashi, Y. Higashi, Measurements of the isobaric specific heat capacities for trans -1,3,3,3-tetrafluoropropene (HFO-1234ze(E)) in the liquid phase. J. Chem. Eng. Data. 55, 2267–2270 (2010). https://doi.org/10.1021/je900799e
K. Zhang, H. Chen, Z. Yang, Y. Duan, Experimental pvT property for the liquid HFO1234ze(E) using the isochoric method. J. Chem. Thermodyn. 149, 106160 (2020). https://doi.org/10.1016/j.jct.2020.106160
T. Sotani, H. Kubota, S. Matsuo, Y. Tanaka, Vapor pressures and compressed liquid PVT properties of 1,1,1,3,3-pentafluoropropane(HFC-245fa). Rev. High Press. Sci. Technol. 7, 1195–1197 (1998). https://doi.org/10.4131/jshpreview.7.1195
Y. Higashi, S. Hayasaka, C. Shirai, R. Akasaka, Measurements of PρT properties, vapor pressures, saturated densities, and critical parameters for R 1234ze(Z) and R 245fa. Int. J. Refrig. 52, 100–108 (2015). https://doi.org/10.1016/j.ijrefrig.2014.12.007
X. Li, Q. Pang, J. Liu, Q. Ning, G. He, Isothermal vapour-liquid equilibrium data for environmentally friendly binary system {R1234ze(E) + R245fa}. J. Chem. Thermodyn. 175, 106894 (2022). https://doi.org/10.1016/j.jct.2022.106894
P.M. Mathias, T.W. Copeman, Extension of the Peng-Robinson equation of state to complex mixtures: evaluation of the various forms of the local composition concept. Fluid Phase Equilib. (1983). https://doi.org/10.1016/0378-3812(83)80084-3
H. Orbey, S.I. Sandler, Modelling Vapor-Liquid Equilibria. Cubic Equation of State and Their Mixing Rules (Cambridge University Press, Cambridge, 1998)
J.M. Smith, H.C. Van Ness, M.M. Abbot, M.T. Swihart, Introduction to Chemical Engineering Thermodynamics, 8th edn. (McGraw Hill, New York, 2018)
X. Xu, S. Lasala, R. Privat, J.N. Jaubert, E-PPR78: a proper cubic EoS for modelling fluids involved in the design and operation of carbon dioxide capture and storage (CCS) processes. Int. J. Greenh. Gas Control. 56, 126–154 (2017). https://doi.org/10.1016/j.ijggc.2016.11.015
Z. Yang, X. Tang, J. Wu, J. Lu, Experimental measurements of saturated vapor pressure and isothermal vapor-liquid equilibria for 1,1,1,2-Tetrafluoroethane (HFC-134a) + 3,3,3-trifluoropropene (HFO-1243zf) binary system. Fluid Phase Equilib. 498, 86–93 (2019). https://doi.org/10.1016/j.fluid.2019.06.020
L. Kou, Z. Yang, X. Tang, W. Zhang, J. Lu, Experimental measurements and correlation of isothermal vapor-liquid equilibria for HFC-32 + HFO-1234ze (E) and HFC-134a + HFO-1234ze (E) binary systems. J. Chem. Thermodyn. 139, 105798 (2019). https://doi.org/10.1016/j.jct.2019.04.020
E.W. Lemmon, I.H. Bell, M.L. Huber, M.O. McLinden, NIST Standard Reference Database 23: Reference Fluid Thermodynamic and Transport Properties-REFPROP, Version 10.0, National Institute of Standards and Technology, Stand. Ref. Data Progr. Gaithersbu (2018). https://doi.org/10.18434/T4/1502528.
S. Dahl, M.L. Michelsen, High-pressure vapor-liquid equilibrium with a UNIFAC-based equation of state. AIChE J. 36, 1829–1836 (1990). https://doi.org/10.1002/aic.690361207
R. Privat, F. Mutelet, Predicting the phase equilibria of synthetic petroleum fluids with the PPR78 approach. Thermodynamics 56, 3225–3235 (2010). https://doi.org/10.1002/aic
R. Privat, F. Mutelet, J.N. Jaubert, Addition of the hydrogen sulfide group to the PPR 78, Model (Predictive 1978, Peng-Robinson equation of state with temperature dependent kij-calculated through a group contribution method). Ind. Eng. Chem. Res. 47, 10041–10052 (2008). https://doi.org/10.1021/ie800799z
X. Xu, R. Privat, P. Duchet-suchaux, F. Bran, Predicting binary-interaction parameters of cubic equations of state for petroleum fluids containing pseudo-components. Ind. Eng. Chem. Res. 54, 2816–2824 (2015). https://doi.org/10.1021/ie504920g
J.W. Qian, R. Privat, J.N. Jaubert, C. Coquelet, D. Ramjugernath, Fluid-phase-equilibrium prediction of fluorocompound-containing binary systems with the predictive E-PPR78 model. Int. J. Refrig. 73, 65–90 (2017). https://doi.org/10.1016/j.ijrefrig.2016.09.013
R. Akasaka, Y. Zhou, E.W. Lemmon, A fundamental equation of state for 1,1,1,3,3-pentafluoropropane (R-245fa). J. Phys. Chem. Ref. Data. 44, 013104 (2015). https://doi.org/10.1063/1.4913493
W. Wagner, J. Ewers, W. Pentermann, New vapour-pressure measurements and a new rational vapour-pressure equation for oxygen. J. Chem. Thermodyn. 8, 1049–1060 (1976). https://doi.org/10.1016/0021-9614(76)90136-1
M.O. McLinden, M. Thol, E.W. Lemmon, Thermodynamic properties of trans-1,3,3,3-tetrafluoropropene [R1234ze(E)]: measurements of density and vapor pressure and a comprehensive equation of state, in Proc. 13th Int. Refrig. Air Cond. Conf. Purdue (2010)
K. Tanaka, G. Takahashi, Y. Higashi, Measurements of the vapor pressures and p ρ T properties for trans -1,3,3,3-tetrafluoropropene (HFO-1234ze(E)). J. Chem. Eng. Data. 55, 2169–2172 (2010). https://doi.org/10.1021/je900756g
G. Di Nicola, J.S. Brown, L. Fedele, S. Bobbo, C. Zilio, Saturated pressure measurements of trans-1,3,3,3-tetrafluoroprop-1-ene (R1234ze(E)) for reduced temperatures ranging from 0.58 to 0.92. J. Chem. Eng. Data. 57, 2197–2202 (2012). https://doi.org/10.1021/je300240k
M. Gong, H. Zhang, H. Li, Q. Zhong, X. Dong, J. Shen, J. Wu, Vapor pressures and saturated liquid densities of HFO1234ze(E) at temperatures from 253.343 to 293.318 K. Int. J. Refrig. 64, 168–175 (2016). https://doi.org/10.1016/j.ijrefrig.2016.01.007
M. Thol, E.W. Lemmon, Equation of state for the thermodynamic properties of trans-1,3,3,3-tetrafluoropropene [R-1234ze(E)]. Int. J. Thermophys. 37, 1–16 (2016). https://doi.org/10.1007/s10765-016-2040-6
P.H. van Konynenburg, R.L. Scott, Critical lines and phase equilibria in binary van der Waals mixtures. Philos. Trans. R. Soc. London. Ser. A 298, 495–540 (1980). https://doi.org/10.1098/rsta.1980.0266
R. Privat, J.N. Jaubert, Classification of global fluid-phase equilibrium behaviors in binary systems. Chem. Eng. Res. Des. 91, 1807–1839 (2013). https://doi.org/10.1016/j.cherd.2013.06.026
Acknowledgements
This work was supported by the open fund from the Key Laboratory of Sulfur Hexafluoride of China Southern Power Grid Co., Ltd. (No. GDDKY2021KF04) and the National Science Foundation of China (No.51976164)
Author information
Authors and Affiliations
Contributions
NT and ZY wrote the main manuscript text; XT and DS performed the measurement. ZY modeled the results and WG prepared the Tables and Figures. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical Approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tang, N., Gu, W., Sun, D. et al. Isothermal Vapor–Liquid Equilibrium for the Binary System of Trans-1,3,3,3-tetrafluoropropene and 1,1,1,3,3-Pentafluoropropane. Int J Thermophys 44, 117 (2023). https://doi.org/10.1007/s10765-023-03219-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10765-023-03219-w