Abstract
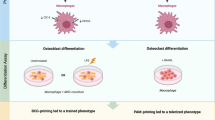
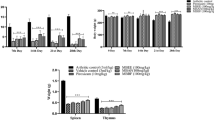
Macrophages actively participate in immunomodulatory processes throughout periodontal inflammation. Regulation of M1/M2 polarization affects macrophage chemokine and cytokine secretion, resulting in a distinct immunological status that influences prognosis. Semaphorin 3A (Sema3A), a neurite growth factor, exerts anti-inflammatory effects. In this study, we investigated the immunomodulation of Sema3A on macrophage-related immune responses in vivo and in vitro. Topical medications of Sema3A in mice with periodontitis alleviated inflammatory cell infiltration into gingival tissue and reduced areas with positive IL-6 and TNFα expression. We observed that the positive area with the M2 macrophage marker CD206 increased and that of the M1 macrophage marker iNOS decreased in Sema3A-treated mice. It has been postulated that Sema3A alleviates periodontitis by regulating alternative macrophage activation. To understand the mechanism underlying Sema3A modulation of macrophage polarization, an in vitro macrophage research model was established with RAW264.7 cells, and we demonstrated that Sema3A promotes LPS/IFNγ-induced M1 macrophages to polarize into M2 macrophages and activates the PI3K/AKT/mTOR signaling pathways. Inhibition of the PI3K signaling pathway activation might reduce anti-inflammatory activity and boost the expression of the inflammatory cytokines, iNOS, IL-12, TNFα, and IL-6. This study indicated that Sema3A might be a feasible drug to regulate alternative macrophage activation in the inflammatory response and thus alleviate periodontitis.







Similar content being viewed by others
DATA AVAILABILITY
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Change history
21 February 2023
A Correction to this paper has been published: https://doi.org/10.1007/s10753-023-01788-4
References
Tonetti, M.S., H. Greenwell, and K.S. Kornman. 2018. Staging and grading of periodontitis: Framework and proposal of a new classification and case definition. Journal of Periodontology 89 (Suppl 1): S159–S172. https://doi.org/10.1002/JPER.18-0006.
Nazir, M., A. Al-Ansari, K. Al-Khalifa, M. Alhareky, B. Gaffar, and K. Almas. 2020. Global prevalence of periodontal disease and lack of its surveillance. The Scientific World Journal 2020: 2146160. https://doi.org/10.1155/2020/2146160.
Luo, L.S., H.H. Luan, J.F. Jiang, L. Wu, C. Li, W.D. Leng, and X.T. Zeng. 2022. The spatial and temporal trends of severe periodontitis burden in Asia, 1990–2019: A population-based epidemiological study. Journal of Periodontology 93: 1615–1625. https://doi.org/10.1002/JPER.21-0625.
Lang, N.P., and P.M. Bartold. 2018. Periodontal health. Journal of Periodontology 89 (Suppl 1): S9–S16. https://doi.org/10.1002/JPER.16-0517.
Pan, W., Q. Wang, and Q. Chen. 2019. The cytokine network involved in the host immune response to periodontitis. International Journal of Oral Science 11: 30. https://doi.org/10.1038/s41368-019-0064-z.
Meyle, J., and I. Chapple. 2000. Molecular aspects of the pathogenesis of periodontitis. Periodontology 2015 (69): 7–17. https://doi.org/10.1111/prd.12104.
Hajishengallis, G., and T. Chavakis. 2021. Local and systemic mechanisms linking periodontal disease and inflammatory comorbidities. Nature reviews. Immunology 21: 426–440. https://doi.org/10.1038/s41577-020-00488-6.
Zhang, X., Q. Wang, X. Yan, Y. Shan, L. Xing, M. Li, H. Long, and W. Lai. 2020. Immune landscape of periodontitis unveils alterations of infiltrating immunocytes and molecular networks-aggregating into an interactive web-tool for periodontitis related immune analysis and visualization. Journal of Translational Medicine 18: 438. https://doi.org/10.1186/s12967-020-02616-1.
Di Stefano, M., A. Polizzi, S. Santonocito, A. Romano, T. Lombardi, and G. Isola. 2022. Impact of oral microbiome in periodontal health and periodontitis: A critical review on prevention and treatment. International Journal of Molecular Sciences 23. https://doi.org/10.3390/ijms23095142
Xue, J., S.V. Schmidt, J. Sander, A. Draffehn, W. Krebs, I. Quester, D. De Nardo, T.D. Gohel, M. Emde, L. Schmidleithner, H. Ganesan, A. Nino-Castro, M.R. Mallmann, L. Labzin, H. Theis, M. Kraut, M. Beyer, E. Latz, T.C. Freeman, T. Ulas, and J.L. Schultze. 2014. Transcriptome-based network analysis reveals a spectrum model of human macrophage activation. Immunity 40: 274–288. https://doi.org/10.1016/j.immuni.2014.01.006.
Karnes, J.M., S.D. Daffner, and C.M. Watkins. 2015. Multiple roles of tumor necrosis factor-alpha in fracture healing. Bone 78: 87–93. https://doi.org/10.1016/j.bone.2015.05.001.
Lam, R.S., N.M. O’Brien-Simpson, J.A. Holden, J.C. Lenzo, S.B. Fong, and E.C. Reynolds. 2016. Unprimed, M1 and M2 macrophages differentially interact with Porphyromonas gingivalis. PLoS ONE 11: e0158629. https://doi.org/10.1371/journal.pone.0158629.
Yang, J., Y. Zhu, D. Duan, P. Wang, Y. Xin, L. Bai, Y. Liu, and Y. Xu. 2018. Enhanced activity of macrophage M1/M2 phenotypes in periodontitis. Archives of Oral Biology 96: 234–242. https://doi.org/10.1016/j.archoralbio.2017.03.006.
Zheng, X., S. Wang, L. Xiao, P. Han, K. Xie, S. Ivanovski, Y. Xiao, and Y. Zhou. 2022. LiCl-induced immunomodulatory periodontal regeneration via the activation of the Wnt/beta-catenin signaling pathway. Journal of Periodontal Research 57: 835–848. https://doi.org/10.1111/jre.13022.
Miyashita, Y., R. Kuraji, H. Ito, and Y. Numabe. 2022. Wound healing in periodontal disease induces macrophage polarization characterized by different arginine-metabolizing enzymes. Journal of Periodontal Research 57: 357–370. https://doi.org/10.1111/jre.12965.
Nakao, Y., T. Fukuda, Q. Zhang, T. Sanui, T. Shinjo, X. Kou, C. Chen, D. Liu, Y. Watanabe, C. Hayashi, H. Yamato, K. Yotsumoto, U. Tanaka, T. Taketomi, T. Uchiumi, A.D. Le, S. Shi, and F. Nishimura. 2021. Exosomes from TNF-alpha-treated human gingiva-derived MSCs enhance M2 macrophage polarization and inhibit periodontal bone loss. Acta Biomaterialia 122: 306–324. https://doi.org/10.1016/j.actbio.2020.12.046.
Polleux, F., T. Morrow, and A. Ghosh. 2000. Semaphorin 3A is a chemoattractant for cortical apical dendrites. Nature 404: 567–573. https://doi.org/10.1038/35007001.
Hayashi, M., T. Nakashima, M. Taniguchi, T. Kodama, A. Kumanogoh, and H. Takayanagi. 2012. Osteoprotection by semaphorin 3A. Nature 485: 69–74. https://doi.org/10.1038/nature11000.
Xu, R. 2014. Semaphorin 3A: A new player in bone remodeling. Cell Adhesion & Migration 8: 5–10. https://doi.org/10.4161/cam.27752.
Li, Y., L. Yang, S. He, and J. Hu. 2015. The effect of semaphorin 3A on fracture healing in osteoporotic rats. Journal of Orthopaedic Science 20: 1114–1121. https://doi.org/10.1007/s00776-015-0771-z.
Ranganathan, P., C. Jayakumar, R. Mohamed, N.L. Weintraub, and G. Ramesh. 2014. Semaphorin 3A inactivation suppresses ischemia-reperfusion-induced inflammation and acute kidney injury. American Journal of Physiology-Renal Physiology 307: F183–F194. https://doi.org/10.1152/ajprenal.00177.2014.
Liu, L.N., X.M. Li, D.Q. Ye, and H.F. Pan. 2018. Emerging role of semaphorin-3A in autoimmune diseases. Inflammopharmacology 26: 655–665. https://doi.org/10.1007/s10787-018-0484-y.
Rienks, M., P. Carai, N. Bitsch, M. Schellings, M. Vanhaverbeke, J. Verjans, I. Cuijpers, S. Heymans, and A. Papageorgiou. 2017. Sema3A promotes the resolution of cardiac inflammation after myocardial infarction. Basic Research in Cardiology 112: 42. https://doi.org/10.1007/s00395-017-0630-5.
Vergadi, E., E. Ieronymaki, K. Lyroni, K. Vaporidi, and C. Tsatsanis. 2017. Akt signaling pathway in macrophage activation and M1/M2 polarization. Journal of Immunology (Baltimore, Md. : 1950) 198:1006–1014. https://doi.org/10.4049/jimmunol.1601515
Braun, C., K. Katholnig, C. Kaltenecker, M. Linke, N. Sukhbaatar, M. Hengstschlager, and T. Weichhart. 2021. p38 regulates the tumor suppressor PDCD4 via the TSC-mTORC1 pathway. Cell Stress 5:176–182. https://doi.org/10.15698/cst2021.12.260
Souza, J.A.C., A.V.B. Nogueira, P.P.C. Souza, G. Oliveira, M.C. Medeiros, G.P. Garlet, J.A. Cirelli, and C.J. Rossa. 2017. Suppressor of cytokine signaling 1 expression during LPS-induced inflammation and bone loss in rats. Brazilian Oral Research 31: e75. https://doi.org/10.1590/1807-3107BOR-2017.vol31.0075.
Petean, I.B.F., L.A. Almeida-Junior, M.F.M. Arnez, A.M. Queiroz, R.A.B. Silva, L.A.B. Silva, L.H. Faccioli, and F.W.G. Paula-Silva. 2021. Celecoxib treatment dampens LPS-induced periapical bone resorption in a mouse model. International Endodontic Journal 54: 1289–1299. https://doi.org/10.1111/iej.13472.
Mukherjee, S., R. Hussaini, R. White, D. Atwi, A. Fried, S. Sampat, L. Piao, Q. Pan, and P. Banerjee. 2018. TriCurin, a synergistic formulation of curcumin, resveratrol, and epicatechin gallate, repolarizes tumor-associated macrophages and triggers an immune response to cause suppression of HPV+ tumors. Cancer Immunology, Immunotherapy 67: 761–774. https://doi.org/10.1007/s00262-018-2130-3.
Yang, B., X. Pang, Z. Li, Z. Chen, and Y. Wang. 2021. Immunomodulation in the treatment of periodontitis: Progress and perspectives. Frontiers in immunology 12: 781378. https://doi.org/10.3389/fimmu.2021.781378.
Wang, W., C. Zheng, J. Yang, and B. Li. 2021. Intersection between macrophages and periodontal pathogens in periodontitis. Journal of Leukocyte Biology 110: 577–583. https://doi.org/10.1002/JLB.4MR0421-756R.
Liu, H., J. Xia, Y. Chen, J. Ai, T. Wang, and G. Tan. 2021. Immunosuppressive regulation of dendritic cells and T cells in allergic rhinitis by semaphorin 3A. American Journal of Rhinology & Allergy 35: 846–853. https://doi.org/10.1177/19458924211005592.
Casazza, A., D. Laoui, M. Wenes, S. Rizzolio, N. Bassani, M. Mambretti, S. Deschoemaeker, J.A. Van Ginderachter, L. Tamagnone, and M. Mazzone. 2013. Impeding macrophage entry into hypoxic tumor areas by Sema3A/Nrp1 signaling blockade inhibits angiogenesis and restores antitumor immunity. Cancer Cell 24: 695–709. https://doi.org/10.1016/j.ccr.2013.11.007.
Teng, Y., Z. Yin, J. Li, K. Li, X. Li, and Y. Zhang. 2017. Adenovirus-mediated delivery of Sema3A alleviates rheumatoid arthritis in a serum-transfer induced mouse model. Oncotarget 8: 66270–66280. https://doi.org/10.18632/oncotarget.19915
Preshaw, P.M., and J.J. Taylor. 2011. How has research into cytokine interactions and their role in driving immune responses impacted our understanding of periodontitis? Journal of Clinical Periodontology 38 (Suppl 11): 60–84. https://doi.org/10.1111/j.1600-051X.2010.01671.x.
Naruishi, K., and T. Nagata. 2018. Biological effects of interleukin-6 on gingival fibroblasts: Cytokine regulation in periodontitis. Journal of Cellular Physiology 233: 6393–6400. https://doi.org/10.1002/jcp.26521.
Luo, G., F. Li, X. Li, Z.G. Wang, and B. Zhang. 2018. TNF-alpha and RANKL promote osteoclastogenesis by upregulating RANK via the NF-kappaB pathway. Molecular Medicine Reports 17: 6605–6611. https://doi.org/10.3892/mmr.2018.8698.
Marahleh, A., H. Kitaura, F. Ohori, A. Kishikawa, S. Ogawa, W.R. Shen, J. Qi, T. Noguchi, Y. Nara, and I. Mizoguchi. 2019. TNF-alpha Directly enhances osteocyte RANKL expression and promotes osteoclast formation. Frontiers in immunology 10: 2925. https://doi.org/10.3389/fimmu.2019.02925.
Metcalfe, S., N. Anselmi, A. Escobar, M.B. Visser, and J.G. Kay. 2021. Innate phagocyte polarization in the oral cavity. Frontiers in immunology 12: 768479. https://doi.org/10.3389/fimmu.2021.768479.
Ruffell, B., and L.M. Coussens. 2015. Macrophages and therapeutic resistance in cancer. Cancer Cell 27: 462–472. https://doi.org/10.1016/j.ccell.2015.02.015.
Afik, R., E. Zigmond, M. Vugman, M. Klepfish, E. Shimshoni, M. Pasmanik-Chor, A. Shenoy, E. Bassat, Z. Halpern, T. Geiger, I. Sagi, and C. Varol. 2016. Tumor macrophages are pivotal constructors of tumor collagenous matrix. The Journal of Experimental Medicine 213: 2315–2331. https://doi.org/10.1084/jem.20151193.
Olmsted-Davis, E., J. Mejia, E. Salisbury, Z. Gugala, and A.R. Davis. 2021. A Population of M2 macrophages associated with bone formation. Frontiers in immunology 12: 686769. https://doi.org/10.3389/fimmu.2021.686769.
Miao, Y., L. He, X. Qi, and X. Lin. 2020. Injecting immunosuppressive M2 macrophages alleviates the symptoms of periodontitis in mice. Frontiers in Molecular Biosciences 7: 603817. https://doi.org/10.3389/fmolb.2020.603817.
Oliveira Costa, F., L.O. Miranda Cota, E.J. Pereira Lages, T.C. Medeiros Lorentz, A.M. Soares Dutra Oliveira, P.A. Dutra Oliveira, and J.E. Costa. 2011. Progression of periodontitis in a sample of regular and irregular compliers under maintenance therapy: A 3-year follow-up study. Journal of Periodontology 82:1279–87. https://doi.org/10.1902/jop.2011.100664
Schwendicke, F., M. Stolpe, A. Plaumann, and C. Graetz. 2016. Cost-effectiveness of regular versus irregular supportive periodontal therapy or tooth removal. Journal of Clinical Periodontology 43: 940–947. https://doi.org/10.1111/jcpe.12595.
Yamashita, N., M. Yamane, F. Suto, and Y. Goshima. 2016. TrkA mediates retrograde semaphorin 3A signaling through plexin A4 to regulate dendritic branching. Journal of Cell Science 129: 1802–1814. https://doi.org/10.1242/jcs.184580.
Xing, Q., J. Feng, and X. Zhang. 2021. Glucocorticoids suppressed osteoblast differentiation by decreasing Sema3A expression via the PIK3/Akt pathway. Experimental Cell Research 403: 112595. https://doi.org/10.1016/j.yexcr.2021.112595.
Fang, S., X. Wan, X. Zou, S. Sun, X. Hao, C. Liang, Z. Zhang, F. Zhang, B. Sun, H. Li, and B. Yu. 2021. Arsenic trioxide induces macrophage autophagy and atheroprotection by regulating ROS-dependent TFEB nuclear translocation and AKT/mTOR pathway. Cell Death & Disease 12: 88. https://doi.org/10.1038/s41419-020-03357-1.
Wei, Y., M. Liang, L. Xiong, N. Su, X. Gao, and Z. Jiang. 2021. PD-L1 induces macrophage polarization toward the M2 phenotype via Erk/Akt/mTOR. Experimental Cell Research 402: 112575. https://doi.org/10.1016/j.yexcr.2021.112575.
ACKNOWLEDGEMENTS
Dr. Leyi Chen especially thanks Dr. Jingying Chen (0000-0002-7617-5902) for his help with slide scanning and instructions on immunohistochemistry.
Funding
This study was supported by the Oral Infectious Disease Mechanism Research and Clinical Translation Application Innovation Team of Guangdong Province of China (2021KCXTD033 to Buling Wu) and the President Foundation of Shenzhen Stomatology Hospital (Pingshan), Southern Medical University (2022A001 to Buling Wu).
Author information
Authors and Affiliations
Contributions
TT, WAX, and BLW conceived and designed the experiments. LYC completed most of the animal experiments and wrote the original draft. TT finished the in vitro experiment and drafted the final manuscript and review. ZTW collected and analyzed the data. TT and MQZ helped with some of the immunochemistry staining. All the authors reviewed and approved the final draft of the manuscript.
Corresponding authors
Ethics declarations
Ethics Approval
Animal experiments were approved and conducted in accordance with the guidelines established by Shenzhen TOP Biotechnology Company (TOP-IACUC-2021–0107).
Consent to Participate
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Summary
Sema3A exerts an ameliorative effect on LPS-induced periodontitis in mice by regulating the alternative polarization of macrophages.
The original online version of this article was revised: The statement "Tian Tian and Leyi Chen are the co-first authors who contributed equally to this work and share first authorship." should be captured as article note.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tian, T., Chen, L., Wang, Z. et al. Sema3A Drives Alternative Macrophage Activation in the Resolution of Periodontitis via PI3K/AKT/mTOR Signaling. Inflammation 46, 876–891 (2023). https://doi.org/10.1007/s10753-022-01777-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-022-01777-z