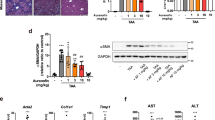
Abstract—
Chronic tissue injury resulting in fibrosis of multiple organs, responsible for one-third of the death globally. Liver fibrosis is a common pathway/condition involved in all chronic liver diseases. Thioacetamide (TAA), a hepatotoxicant, was used to induce hepatic fibrosis. Anti-diabetic drug glibenclamide (GLB) possesses anti-inflammatory properties and inhibits NACHT, LRR, and PYD domains-containing protein 3 (NLRP3) inflammasome activation. Dimethyl fumarate (DMF), a multiple sclerosis drug, activates the nuclear factor erythroid 2-related factor 2 (Nrf2)/antioxidant response element (ARE) pathway and maintains the antioxidant status in the cell. The present study was designed to investigate (i) role of NLRP3 inflammasome and Nrf2/ARE pathway in TAA-induced hepatotoxicity and liver fibrosis, (ii) mechanism involved in GLB and DMF mediated hepatoprotection against TAA-induced hepatotoxicity, and (iii) additional/synergistic hepatoprotective effect of combination treatment with NLRP3 inhibition + Nrf2 activation or GLB + DMF or MCC950 + 4OI to reverse/ameliorate the experimental liver fibrosis completely. TAA was administered intraperitoneally to mice for seven consecutive weeks, and treatments of GLB, DMF, GLB + DMF, MCC950, 4OI, and MCC950 + 4OI were provided for the last three consecutive weeks. The intervention with GLB, DMF, GLB + DMF, MCC950, 4OI, and MCC950 + 4OI significantly protected TAA-induced oxidative stress and inflammatory conditions by improving biochemical, histological, and immunoexpression changes in mice. The GLB, DMF, and GLB + DMF intervention exhibited a better protective effect compared with MCC950, 4OI, and MCC950 + 4OI, which revealed that this specific inhibitor/activator possesses only NLRP3 inflammasome inhibitory/Nrf2 activatory properties. In contrast, the clinical drug GLB and DMF have several other beneficial effects, which are independent of NLRP3 inhibition and Nrf2 activation.








Similar content being viewed by others
Availability of Data and Material
All data and materials support their published claims and comply with field standards. Data will be available on demand.
Code Availability
It is not applicable in present research.
References
Asrani, Sumeet K., Harshad Devarbhavi, John Eaton, and Patrick S. Kamath. 2019. Burden of liver diseases in the world. Journal of hepatology 70: 151–171. https://doi.org/10.1016/j.jhep.2018.09.014.
Sepanlou, Sadaf G., Saeid Safiri, Catherine Bisignano, Kevin S. Ikuta, Shahin Merat, Mehdi Saberifiroozi, Hossein Poustchi, et al. 2020. The global regional and national burden of cirrhosis by cause in 195 countries and territories 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. The Lancet gastroenterology & hepatology 5: 245–266. https://doi.org/10.1016/S2468-1253(19)30349-8.
Wallace, M.C., K. Hamesch, M. Lunova, Y. Kim, R. Weiskirchen, P. Strnad, and S.L. Friedman. 2015. Standard operating procedures in experimental liver research: Thioacetamide model in mice and rats. Laboratory animals 49: 21–29. https://doi.org/10.1177/0023677215573040.
Li, Sha, Hor-Yue. Tan, Ning Wang, Zhang-Jin. Zhang, Lixing Lao, Chi-Woon. Wong, and Yibin Feng. 2015. The role of oxidative stress and antioxidants in liver diseases. International Journal of Molecular Sciences 16: 26087–26124. https://doi.org/10.3390/ijms161125942.
Dixon, Laura J., Chris A. Flask, Bettina G. Papouchado, Ariel E. Feldstein, and Laura E. Nagy. 2013. Caspase-1 as a central regulator of high fat diet-induced non-alcoholic steatohepatitis. PLoS ONE 8: e56100. https://doi.org/10.1371/journal.pone.0056100.
Zhang, Gensheng, Xiuhui Lin, Shufang Zhang, Huiqing Xiu, Chuli Pan, and Wei Cui. 2017. A protective role of glibenclamide in inflammation-associated injury. Mediators of inflammation 2017: 3578702. https://doi.org/10.1155/2017/3578702.
Lamkanfi, Mohamed, James L. Mueller, Alberto C. Vitari, Shahram Misaghi, Anna Fedorova, Kurt Deshayes, Wyne P. Lee, Hal M. Hoffman, and Vishva M. Dixit. 2009. Glyburide inhibits the Cryopyrin/Nalp3 inflammasome. The Journal of Cell Biology 187: 61–70. https://doi.org/10.1083/jcb.200903124.
Koh, Gavin C K W., Tassili A. Weehuizen, Katrin Breitbach, Kathrin Krause, Hanna K. de Jong, Liesbeth M. Kager, Arjan J. Hoogendijk, et al. 2013. Glyburide reduces bacterial dissemination in a mouse model of melioidosis. PLoS neglected tropical diseases 7: e2500. https://doi.org/10.1371/journal.pntd.0002500.
Sharma, Ritu S., David J. Harrison, Dorothy Kisielewski, Diane M. Cassidy, Alison D. McNeilly, Jennifer R. Gallagher, Shaun V. Walsh, et al. 2018. Experimental nonalcoholic steatohepatitis and liver fibrosis are ameliorated by pharmacologic activation of Nrf2 (NF-E2 p45-related factor 2). Cellular and molecular gastroenterology and hepatology 5: 367–398. https://doi.org/10.1016/j.jcmgh.2017.11.016.
Dwivedi, D.K., Gopabandhu Jena, and Vinod Kumar. 2020. Dimethyl fumarate protects thioacetamide-induced liver damage in rats: Studies on Nrf2 NLRP3 and NF-κB. Journal of biochemical and molecular toxicology 34: e22476. https://doi.org/10.1002/jbt.22476.
Coll, Rebecca C., Avril A B. Robertson, Jae Jin Chae, Sarah C. Higgins, Raúl. Muñoz-Planillo, Marco C. Inserra, Irina Vetter, Lara S. Dungan, Brian G. Monks, and Andrea Stutz. 2015. A small-molecule inhibitor of the NLRP3 inflammasome for the treatment of inflammatory diseases. Nature medicine 21: 248.
Mridha, Auvro R., Alexander Wree, Avril A B. Robertson, Matthew M. Yeh, Casey D. Johnson, Derrick M. Van Rooyen, Fahrettin Haczeyni, et al. 2017. NLRP3 inflammasome blockade reduces liver inflammation and fibrosis in experimental NASH in mice. Journal of hepatology 66: 1037–1046. https://doi.org/10.1016/j.jhep.2017.01.022.
Green, Jack P., Yu. Shi, Fatima Martin-Sanchez, Pablo Pelegrin, Gloria Lopez-Castejon, Catherine B. Lawrence, and David Brough. 2018. Chloride regulates dynamic NLRP3-dependent ASC oligomerization and inflammasome priming. Proceedings of the National Academy of Sciences of the United States of America 115: E9371–E9380. https://doi.org/10.1073/pnas.1812744115.
Mills, Evanna L., Dylan G. Ryan, Hiran A. Prag, Dina Dikovskaya, Deepthi Menon, Zbigniew Zaslona, Mark P. Jedrychowski, Ana S H. Costa, Maureen Higgins, and Emily Hams. 2018. Itaconate is an anti-inflammatory metabolite that activates Nrf2 via alkylation of KEAP1. Nature 556: 113.
Tang, Chun, Shengyu Tan, Yiqing Zhang, Lini Dong, and Xu. Yan. 2019. Activation of Keap1-Nrf2 signaling by 4-octyl itaconate protects human umbilical vein endothelial cells from high glucose. Biochemical and biophysical research communications 508: 921–927. https://doi.org/10.1016/j.bbrc.2018.12.032.
Kim, Yong Ook, Yury Popov, and Detlef Schuppan. 2017. Optimized mouse models for liver fibrosis. Methods in Molecular Biology 1559: 279–296. https://doi.org/10.1007/978-1-4939-6786-5_19.
Dwivedi, D.K., and G.B. Jena. 2019. NLRP3 inhibitor glibenclamide attenuates high-fat diet and streptozotocin-induced non-alcoholic fatty liver disease in rat: Studies on oxidative stress inflammation DNA damage and insulin signalling pathway. Naunyn-Schmiedeberg’s Archives of Pharmacology 393: 705–716. https://doi.org/10.1007/s00210-019-01773-5.
Lin, Yu-Ju, I-Chun Lin, Hong-Ren Yu, Jiunn-Ming Sheen, Li-Tung Huang, and You-Lin Tain. 2018. Early postweaning treatment with dimethyl fumarate prevents prenatal dexamethasone- and postnatal high-fat diet-induced programmed hypertension in male rat offspring. Oxidative medicine and cellular longevity 2018: 5343462. https://doi.org/10.1155/2018/5343462.
Ahmed, Danish, Vikas Kumar, Amita Verma, Manju Sharma, and Manju Sharma. 2015. Antidiabetic, antioxidant, antihyperlipidemic effect of extract of Euryale ferox salisb. With enhanced histopathology of pancreas, liver and kidney in streptozotocin induced diabetic rats. SpringerPlus 4:315. https://doi.org/10.1186/s40064-015-1059-7
Dwivedi, D.K., and G.B. Jena. 2020. Diethylnitrosamine and thioacetamide-induced hepatic damage and early carcinogenesis in rats: Role of Nrf2 activator dimethyl fumarate and NLRP3 inhibitor glibenclamide. Biochemical and Biophysical Research Communications 522: 381–387. https://doi.org/10.1016/j.bbrc.2019.11.100.
Fu, Qun, Jing Li, Lili Qiu, Jiaping Ruan, Mingjie Mao, Shuming Li, and Qinghong Mao. 2020. Inhibiting NLRP3 inflammasome with MCC950 ameliorates perioperative neurocognitive disorders suppressing neuroinflammation in the hippocampus in aged mice. International immunopharmacology 82: 106317. https://doi.org/10.1016/j.intimp.2020.106317.
Wang, Hao, Xuming Sun, Hunter S. Hodge, Carlos M. Ferrario, and Leanne Groban. 2019. NLRP3 inhibition improves heart function in GPER knockout mice. Biochemical and biophysical research communications 514: 998–1003. https://doi.org/10.1016/j.bbrc.2019.05.045.
Yi, Zhongjie, Meihong Deng, Melanie J. Scott, Fu. Guang, Patricia A. Loughran, Zhao Lei, Shilai Li, et al. 2020. IRG1/itaconate activates Nrf2 in hepatocytes to protect against liver ischemia-reperfusion injury. Hepatology 72: 1394–1411. https://doi.org/10.1002/hep.31147.
Tian, Feng, Zhe Wang, Junqiu He, Zhihao Zhang, and Ninghua Tan. 2020. 4-Octyl itaconate protects against renal fibrosis via inhibiting TGF-beta/Smad pathway, autophagy and reducing generation of reactive oxygen species. European journal of pharmacology 873: 172989. https://doi.org/10.1016/j.ejphar.2020.172989.
Pandey, Surya Narayan, Mohit Kwatra, Durgesh Kumar Dwivedi, Priyansha Choubey, Mangala Lahkar, and Ashok Jangra. 2020. 7,8-Dihydroxyflavone alleviated the high-fat diet and alcohol-induced memory impairment: Behavioral biochemical and molecular evidence. Psychopharmacology (Berl) 237: 1827–1840. https://doi.org/10.1007/s00213-020-05502-2.
Rahman, Z., D.K. Dwivedi, and G.B. Jena. 2019. Ethanol-induced gastric ulcer in rats and intervention of tert-butylhydroquinone: Involvement of Nrf2/HO-1 signalling pathway. Human & Experimental Toxicology 39: 547–562. https://doi.org/10.1177/0960327119895559.
Ishak, K., A. Baptista, L. Bianchi, F. Callea, J. De Groote, F. Gudat, H. Denk, V. Desmet, G. Korb, and R.N. MacSween. 1995. Histological grading and staging of chronic hepatitis. Journal of hepatology 22: 696–699. https://doi.org/10.1016/0168-8278(95)80226-6.
Wang, Huafeng, Huan Zhang, Zimu Zhang, Biao Huang, Xixi Cheng, Dan Wang, Zha la Gahu, et al. 2016. Adiponectin-derived active peptide ADP355 exerts anti-inflammatory and anti-fibrotic activities in thioacetamide-induced liver injury. Scientific Reports 6: 19445. https://doi.org/10.1038/srep19445.
Chittaranjan, Sahu, Aarzoo Charaya, Shivani Singla, Durgesh K. Dwivedi, and Gopabandhu Jena. 2020. Zinc deficient diet increases the toxicity of bisphenol A in rat testis. Journal of biochemical and molecular toxicology 34: e22549. https://doi.org/10.1002/jbt.22549.
Sahu, C., D.K. Dwivedi, and G.B. Jena. 2020. Zinc and selenium combination treatment protected diabetes-induced testicular and epididymal damage in rat. Human & experimental toxicology 39: 1235–1256. https://doi.org/10.1177/0960327120914963.
Chen, Yuan-Li., Xu. Guo, Xiao Liang, Juan Wei, Jing Luo, Guan-Nan. Chen, Xiao-Di. Yan, Xue-Ping. Wen, Ming Zhong, and Xin Lv. 2016. Inhibition of hepatic cells pyroptosis attenuates CLP-induced acute liver injury. American journal of translational research 8: 5685–5695.
Abdelrahman, Rehab S., and Noha Abdel-Rahman. 2019. Dimethyl fumarate ameliorates acetaminophen-induced hepatic injury in mice dependent of Nrf-2/HO-1 pathway. Life sciences 217: 251–260. https://doi.org/10.1016/j.lfs.2018.12.013.
Wang, Jinglin, Haozhen Ren, Xianwen Yuan, Hucheng Ma, Xiaolei Shi, and Yitao Ding. 2018. Interleukin-10 secreted by mesenchymal stem cells attenuates acute liver failure through inhibiting pyroptosis. Hepatology research: The official journal of the Japan Society of Hepatology 48: E194–E202. https://doi.org/10.1111/hepr.12969.
de David, Cintia, Graziella Rodrigues, Silvia Bona, Luise Meurer, Javier Gonzalez-Gallego, Maria Jesus Tunon, and Norma Possa Marroni. 2011. Role of quercetin in preventing thioacetamide-induced liver injury in rats. Toxicologic pathology 39: 949–957. https://doi.org/10.1177/0192623311418680.
Kapoor, Radhika, and Poonam Kakkar. 2014. Naringenin accords hepatoprotection from streptozotocin induced diabetes in vivo by modulating mitochondrial dysfunction and apoptotic signaling cascade. Toxicology reports 1: 569–581. https://doi.org/10.1016/j.toxrep.2014.08.002.
Schupp, Nicole, Reinhard Schinzel, August Heidland, and Helga Stopper. 2005. Genotoxicity of advanced glycation end products: Involvement of oxidative stress and of angiotensin II type 1 receptors. Annals of the New York Academy of Sciences 1043: 685–695. https://doi.org/10.1196/annals.1333.079.
Ma, Xingyu, Yang Zhou, Bingke Qiao, Songhong Jiang, Qian Shen, Yuzhu Han, Anfang Liu, et al. 2020. Androgen aggravates liver fibrosis by activation of NLRP3 inflammasome in CCl4 induced liver injury mouse model. American Journal of Physiology-Endocrinology and Metabolism 318: E817–E829. https://doi.org/10.1152/ajpendo.00427.2019.
Wree, Alexander, Akiko Eguchi, Matthew D. McGeough, Carla A. Pena, Casey D. Johnson, Ali Canbay, Hal M. Hoffman, and Ariel E. Feldstein. 2014. NLRP3 inflammasome activation results in hepatocyte pyroptosis liver inflammation and fibrosis in mice. Hepatology 59: 898–910. https://doi.org/10.1002/hep.26592.
Watanabe, Azuma, Muhammad Adnan Sohail, Dawidson Assis Gomes, Ardeshir Hashmi, Jun Nagata, Fayyaz Shiraz Sutterwala, Shamail Mahmood, et al. 2009. Inflammasome-mediated regulation of hepatic stellate cells. American journal of physiology Gastrointestinal and Liver Physiology 296: G1248–G1257. https://doi.org/10.1152/ajpgi.90223.2008.
Boaru, Sorina Georgiana, Erawan Borkham-Kamphorst, Eddy Van de Leur, Eric Lehnen, Christian Liedtke, and Ralf Weiskirchen. 2015. NLRP3 inflammasome expression is driven by NF-kappaB in cultured hepatocytes. Biochemical and biophysical research communications 458: 700–706. https://doi.org/10.1016/j.bbrc.2015.02.029.
Arriola Benitez, Paula Constanza, Ayelen Ivana Pesce Viglietti, Marco Tulio R. Gomes, Sergio Costa Oliveira, Jorge Fabian Quarleri, Guillermo Hernan Giambartolomei, and Maria Victoria Delpino. 2019. Brucella abortus infection elicited hepatic stellate cell-mediated fibrosis through inflammasome-dependent IL-1beta production. Frontiers in immunology 10: 3036. https://doi.org/10.3389/fimmu.2019.03036.
Garstkiewicz, Martha, Gerhard E. Strittmatter, Serena Grossi, Jennifer Sand, Gabriele Fenini, Sabine Werner, Lars E. French, and Hans-Dietmar. Beer. 2017. Opposing effects of Nrf2 and Nrf2-activating compounds on the NLRP3 inflammasome independent of Nrf2-mediated gene expression. European journal of immunology 47: 806–817. https://doi.org/10.1002/eji.201646665.
Miglio, Gianluca, Eleonora Veglia, and Roberto Fantozzi. 2015. Fumaric acid esters prevent the NLRP3 inflammasome-mediated and ATP-triggered pyroptosis of differentiated THP-1 cells. International immunopharmacology 28: 215–219. https://doi.org/10.1016/j.intimp.2015.06.011.
Giustina, Amanda Della, Sandra Bonfante, Graciela Freitas Zarbato, Lucineia Gainski Danielski, Khiany Mathias, Aloir Neri Jr de Oliveira, Leandro Garbossa, et al. 2018. Dimethyl fumarate modulates oxidative stress and inflammation in organs after sepsis in rats. Inflammation 41:315-327. https://doi.org/10.1007/s10753-017-0689-z
Zhang, Ning-Ping., Xue-Jing. Liu, Li. Xie, Xi-Zhong. Shen, and Wu. Jian. 2019. Impaired mitophagy triggers NLRP3 inflammasome activation during the progression from nonalcoholic fatty liver to nonalcoholic steatohepatitis. Laboratory investigation: A journal of technical methods and pathology 99: 749–763. https://doi.org/10.1038/s41374-018-0177-6.
Liao, Shan-Ting., Chao Han, Xu. Ding-Qiao, Fu. Xiao-Wei, Jun-Song. Wang, and Ling-Yi. Kong. 2019. 4-Octyl itaconate inhibits aerobic glycolysis by targeting GAPDH to exert anti-inflammatory effects. Nature communications 10: 5091. https://doi.org/10.1038/s41467-019-13078-5.
Dwivedi, D.K., and G.B. Jena. 2018. Glibenclamide protects against thioacetamide-induced hepatic damage in Wistar rat: Investigation on NLRP3 MMP-2 and stellate cell activation. Naunyn-Schmiedeberg’s Archives of Pharmacology 391: 1257–1274. https://doi.org/10.1007/s00210-018-1540-2.
Schulze-Topphoff, Ulf, Michel Varrin-Doyer, Kara Pekarek, Collin M. Spencer, Aparna Shetty, Sharon A. Sagan, Bruce A. C. Cree, et al. 2016. Dimethyl fumarate treatment induces adaptive and innate immune modulation independent of Nrf2. Proceedings of the National Academy of Sciences of the United States of America 113: 4777–4782. https://doi.org/10.1073/pnas.1603907113.
Acknowledgements
The authors are thankful to the National Institute of Pharmaceutical Education and Research, SAS Nagar, India, for providing financial assistance to carry out the present experimentation. The authors are grateful to the anonymous reviewers for their critical suggestions to improve the quality and scientific clarity of the manuscript.
Funding
The authors are thankful to the National Institute of Pharmaceutical Education and Research, SAS Nagar, India, for providing financial assistance to carry out the present experimentation. Durgesh K. Dwivedi had presented this study at the virtual annual meeting of the Society of Toxicology, USA, on 17 March 2021.
Author information
Authors and Affiliations
Contributions
Durgesh K. Dwivedi conceived, conducted the experiments, analyzed the data, and wrote the manuscript. G. B. Jena conceived the idea, reviewed the manuscript, and administered the project. Finally, both the authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Ethical Approval
All institutional and national guidelines for the care and use of laboratory animals were followed. The animal studies were approved by the Ethics Committee of the NIPER SAS Nagar (IAEC/19/15).
Informed Consent
Informed consent is not applicable in the present study.
Plant Reproducibility
It is not applicable in present research.
Clinical Trials Registration
It is not applicable in present research, as the experiments were performed in mice.
Consent to Participate
It is not applicable in present research.
Consent for Publication
Both authors are willing to publish their research work.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dwivedi, D.K., Jena, G.B. Simultaneous Modulation of NLRP3 Inflammasome and Nrf2/ARE Pathway Rescues Thioacetamide-Induced Hepatic Damage in Mice: Role of Oxidative Stress and Inflammation. Inflammation 45, 610–626 (2022). https://doi.org/10.1007/s10753-021-01571-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-021-01571-3