Abstract
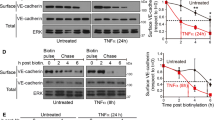
The endothelium serves as a selective barrier and controls the exchange of nutrients, hormones, and leukocytes between blood and tissues. Molecular mechanisms contributing to the pathogenesis of endothelial barrier dysfunction remain incompletely understood. Accumulating evidence implicates bone morphogenetic protein (BMP)-modulator BMPER as a key regulator in endothelial biology. Herein, we analyze the impact of BMPER in the control of endothelial barrier function. To assess the role of BMPER in vascular barrier function in mice, we measured the leakage of Evans blue dye from blood into interstitial lung tissue. BMPER+/− mice exhibited a significantly higher degree of vascular leak compared with wild-type siblings. In accordance with our in vivo observation, siRNA-based BMPER knockdown in human umbilical endothelial cells increased endothelial permeability measured by FITC-dextran passage in transwell assays. Mechanistically, BMPER knockdown reduced the expression of VE-cadherin, a pivotal component of endothelial adherens junctions. Conversely, recombinant human BMPER protein upregulated VE-cadherin protein levels and improved endothelial barrier function in transwell assays. The effects of BMPER knockdown on VE-cadherin expression and endothelial permeability were induced by enhanced BMP activity. Supporting this notion, activation of BMP4-Smad-Id1 signaling reduced VE-cadherin levels and impaired endothelial barrier function in vitro. In vivo, Evans blue dye accumulation was higher in the lungs of BMP4-treated C57BL/6 mice compared to controls indicating that BMP4 increased vascular permeability. High levels of BMPER antagonized BMP4-Smad5-Id1 signaling and prevented BMP4-induced downregulation of VE-cadherin and endothelial leakage, suggesting that BMPER exerts anti-BMP effects and restores endothelial barrier function. Taken together, this data demonstrates that BMPER-modulated BMP pathway activity regulates VE-cadherin expression and vascular barrier function.






Similar content being viewed by others
References
Mehta, D., and A.B. Malik. 2006. Signaling mechanisms regulating endothelial permeability. Physiological Reviews 86(1): 279–367.
Vandenbroucke, E., et al. 2008. Regulation of endothelial junctional permeability. Annals of the New York Academy of Sciences 1123: 134–145.
Komarova, Y., and A.B. Malik. 2010. Regulation of endothelial permeability via paracellular and transcellular transport pathways. Annual Review of Physiology 72: 463–493.
Vestweber, D. 2008. VE-cadherin: the major endothelial adhesion molecule controlling cellular junctions and blood vessel formation. Arteriosclerosis, Thrombosis, and Vascular Biology 28(2): 223–232.
Giannotta, M., M. Trani, and E. Dejana. 2013. VE-cadherin and endothelial adherens junctions: active guardians of vascular integrity. Developmental Cell 26(5): 441–454.
Vestweber, D., et al. 2009. Cell adhesion dynamics at endothelial junctions: VE-cadherin as a major player. Trends in Cell Biology 19(1): 8–15.
Dejana, E., and D. Vestweber. 2013. The role of VE-cadherin in vascular morphogenesis and permeability control. Progress in Molecular Biology and Translational Science 116: 119–144.
Garcia deVinuesa, A., et al. 2016. BMP signaling in vascular biology and dysfunction. Cytokine and Growth Factor Reviews 27: 65–79.
Cai, J., et al. 2012. BMP signaling in vascular diseases. FEBS Letters 586(14): 1993–2002.
Dyer, L.A., X. Pi, and C. Patterson. 2014. The role of BMPs in endothelial cell function and dysfunction. Trends in Endocrinology and Metabolism 25(9): 472–480.
Zhang, J.L., et al. 2008. Crystal structure analysis reveals how the Chordin family member crossveinless 2 blocks BMP-2 receptor binding. Developmental Cell 14(5): 739–750.
Zakin, L., et al. 2008. Development of the vertebral morphogenetic field in the mouse: interactions between Crossveinless-2 and Twisted Gastrulation. Developmental Biology 323(1): 6–18.
Moser, M., et al. 2003. BMPER, a novel endothelial cell precursor-derived protein, antagonizes bone morphogenetic protein signaling and endothelial cell differentiation. Molecular and Cellular Biology 23(16): 5664–5679.
Moser, M., et al. 2007. BMPER is a conserved regulator of hematopoietic and vascular development in zebrafish. Journal of Molecular and Cellular Cardiology 43(3): 243–253.
Heinke, J., et al. 2008. BMPER is an endothelial cell regulator and controls bone morphogenetic protein-4-dependent angiogenesis. Circulation Research 103(8): 804–812.
Pi, X., et al. 2012. Bmper inhibits endothelial expression of inflammatory adhesion molecules and protects against atherosclerosis. Arteriosclerosis, Thrombosis, and Vascular Biology 32(9): 2214–2222.
Helbing, T., et al. 2011. BMP activity controlled by BMPER regulates the proinflammatory phenotype of endothelium. Blood 118(18): 5040–5049.
Ishida, W., et al. 2000. Smad6 is a Smad1/5-induced smad inhibitor: characterization of bone morphogenetic protein-responsive element in the mouse Smad6 promoter. Journal of Biological Chemistry 275(9): 6075–6079.
Yuan, S.Y., and R.R. Rigor. 2010. Regulation of endothelial barrier function. San Rafael: Morgan & Claypool.
Kelley, R., et al. 2009. A concentration-dependent endocytic trap and sink mechanism converts Bmper from an activator to an inhibitor of Bmp signaling. Journal of Cell Biology 184(4): 597–609.
Valdimarsdottir, G., et al. 2002. Stimulation of Id1 expression by bone morphogenetic protein is sufficient and necessary for bone morphogenetic protein-induced activation of endothelial cells. Circulation 106(17): 2263–2270.
Miriyala, S., et al. 2006. Bone morphogenic protein-4 induces hypertension in mice: role of noggin, vascular NADPH oxidases, and impaired vasorelaxation. Circulation 113(24): 2818–2825.
Sorescu, G.P., et al. 2004. Bone morphogenic protein 4 produced in endothelial cells by oscillatory shear stress induces monocyte adhesion by stimulating reactive oxygen species production from a nox1-based NADPH oxidase. Circulation Research 95(8): 773–779.
Gavard, J. 2014. Endothelial permeability and VE-cadherin: a wacky comradeship. Cell Adhesion & Migration 8(2): 158–164.
Deanfield, J.E., J.P. Halcox, and T.J. Rabelink. 2007. Endothelial function and dysfunction: testing and clinical relevance. Circulation 115(10): 1285–1295.
Chang, K., et al. 2007. Bone morphogenic protein antagonists are coexpressed with bone morphogenic protein 4 in endothelial cells exposed to unstable flow in vitro in mouse aortas and in human coronary arteries: role of bone morphogenic protein antagonists in inflammation and atherosclerosis. Circulation 116(11): 1258–1266.
Pardali, E., and P. Ten Dijke. 2012. TGFbeta signaling and cardiovascular diseases. International Journal of Biological Sciences 8(2): 195–213.
Csiszar, A., et al. 2006. Bone morphogenetic protein-2 induces proinflammatory endothelial phenotype. American Journal of Pathology 168(2): 629–638.
Csiszar, A., et al. 2008. Differential proinflammatory and prooxidant effects of bone morphogenetic protein-4 in coronary and pulmonary arterial endothelial cells. American Journal of Physiology - Heart and Circulatory Physiology 295(2): H569–H577.
Csiszar, A., S. Lehoux, and Z. Ungvari. 2009. Hemodynamic forces, vascular oxidative stress, and regulation of BMP-2/4 expression. Antioxidants and Redox Signaling 11(7): 1683–1697.
Csiszar, A., et al. 2005. Regulation of bone morphogenetic protein-2 expression in endothelial cells: role of nuclear factor-kappaB activation by tumor necrosis factor-alpha, H2O2, and high intravascular pressure. Circulation 111(18): 2364–2372.
Helbing, T., et al. 2013. Inhibition of BMP activity protects epithelial barrier function in lung injury. Journal of Pathology 231(1): 105–116.
Helbing, T., et al. 2010. BMPER is upregulated by statins and modulates endothelial inflammation by intercellular adhesion molecule-1. Arteriosclerosis, Thrombosis, and Vascular Biology 30(3): 554–560.
Hussein, K.A., et al. 2014. Bone morphogenetic protein 2: a potential new player in the pathogenesis of diabetic retinopathy. Experimental Eye Research 125: 79–88.
Benn, A., et al. 2016. VE-cadherin facilitates BMP-induced endothelial cell permeability and signaling. Journal of Cell Science 129(1): 206–218.
Bostrom, K.I., et al. 2011. Activation of vascular bone morphogenetic protein signaling in diabetes mellitus. Circulation Research 108(4): 446–457.
Bostrom, K., et al. 1993. Bone morphogenetic protein expression in human atherosclerotic lesions. Journal of Clinical Investigation 91(4): 1800–1809.
Corriere, M.A., et al. 2008. Endothelial Bmp4 is induced during arterial remodeling: effects on smooth muscle cell migration and proliferation. Journal of Surgical Research 145(1): 142–149.
Wong, W.T., et al. 2010. Bone morphogenic protein-4 impairs endothelial function through oxidative stress-dependent cyclooxygenase-2 upregulation: implications on hypertension. Circulation Research 107(8): 984–991.
Corada, M., et al. 1999. Vascular endothelial-cadherin is an important determinant of microvascular integrity in vivo. Proceedings of the National Academy of Sciences of the United States of America 96(17): 9815–9820.
Chatterjee, A., et al. 2008. Heat shock protein 90 inhibitors attenuate LPS-induced endothelial hyperpermeability. American Journal of Physiology - Lung Cellular and Molecular Physiology 294(4): L755–L763.
Murakami, M., et al. 2008. The FGF system has a key role in regulating vascular integrity. Journal of Clinical Investigation 118(10): 3355–3366.
Medici, D., et al. 2010. Conversion of vascular endothelial cells into multipotent stem-like cells. Nature Medicine 16(12): 1400–1406.
Helbing, T., et al. 2010. Kruppel-like factor 15 regulates BMPER in endothelial cells. Cardiovascular Research 85(3): 551–559.
Acknowledgments
We are indebted to Bianca Engert for her outstanding technical assistance.
Authors’ Contributions
T.H. designed and performed the research, analyzed the data, and wrote the paper; G.W., A.H., E.K., L.A., and A.B. performed the research and analyzed the data; K.F. and P.D. performed the research; S.G. and J.S.E. analyzed the data and contributed analytical tools; C.P. and C.B. wrote the paper; and M.M. designed the research and wrote the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All procedures performed in studies involving murine or human tissues were in accordance with the ethical standards of the institution and/or national research committee and with the 1964 Helsinki declaration and its later amendments. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Conflict of Interest
The authors declare that they have no conflict of interest.
Funding
This work was supported by Deutsche Forschungsgemeinschaft Mo973/6-1 to M.M. and by intramural funds to T.H. and M.M.
ELECTRONIC SUPPLEMENTARY MATERIAL
Below is the link to the electronic supplementary material.
Supplement 1
(A) Time course of Smad1/5 phosphorylation in response to BMPER. HUVEC were incubated to BMPER (right panel, 50 ng/ml) for indicated periods. Total cells lysates were used for western blotting with an anti p-Smad1/5 antibody. B-tubulin served as a loading control. (* = P < 0.05, n = 4) (B) Smad5 dependent gene transcription was not altered by increasing BMPER concentrations. Smad5 luciferase reporter plasmid (3GC2wt-luc) was transiently co-transfected with pCMV-β-gal in HEK293 cells. After stimulation with BMPER (250 ng/ml) for additional 20 h luciferase and β-galactosidase activity were measured. Luciferase activity was normalized to β-galactosidase activity and is expressed as mean +/− SD. (* = P < 0.05 versus control, # = P < 0.05 versus BMP4, n = 4) (C) BMPER knockdown does not lead to changes in occludin expression. HUVEC were transfected with a scrsiRNA or BMPER siRNA (siBMPERI or II) for 48 h. Cell extracts were analyzed by western blot for occludin, VE-cadherin and α-tubulin. Reduction of VE-cadherin after transfection of BMPER siRNA served as a positive control. (JPG 1001 kb)
Rights and permissions
About this article
Cite this article
Helbing, T., Wiltgen, G., Hornstein, A. et al. Bone Morphogenetic Protein-Modulator BMPER Regulates Endothelial Barrier Function. Inflammation 40, 442–453 (2017). https://doi.org/10.1007/s10753-016-0490-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-016-0490-4