Abstract
Evidence of freshwater biodiversity decline, species extinction and severe alterations in the biological structure and function of freshwater ecosystems is accumulating. Here we explore the role of macrophyte diversity in lowland streams for the abundance, taxon richness and composition of macroinvertebrate communities also including the abundance of different functional feeding groups. We applied a controlled in situ experiment in four small lowland stream reaches situated in rural landscapes in Denmark. We were able to explain a major fraction of the variability in the macroinvertebrate communities by differences in total macrophyte surface areas among treatments, but the number of macrophyte species in the experimental treatment also played a highly significant role (i.e. one, two or three plant species) for both the abundance, richness and composition of the macroinvertebrate community. We strongly encourage managers to protect plant diversity in streams, not only because the plants themselves constitute an important part of the biodiversity, but also because they positively affect the macroinvertebrate community. Moreover, macrophyte-friendly management can be seen as a nature-based solution to mitigate the degraded physical conditions characterising many streams in agricultural catchments.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Evidence of freshwater biodiversity decline, species extinction and severe alterations in the biological structure and function of freshwater ecosystems has been accumulating (Strayer & Dudgeon, 2010; Reid et al., 2010; WWF, 2020, Haase et al. 2023). Despite this evident crisis in freshwater biodiversity, efforts to address it lag behind, resulting in insufficient measures to initiate the necessary changes. This is not only a result of political delays but also from the intricate nature of freshwater habitats, posing challenges in pinpointing precisely where to initiate efforts to reverse adverse trends. The complexity encompasses both the many different organisms and their interplay as well as multiple ecosystem stressors acting alone and in concert (Birk et al., 2020). In the present study, we focus on benthic macroinvertebrates that for more than 100 years have acted as sentinels of environmental perturbations (Friberg, 2014). Macroinvertebrates are a diverse and ubiquitous group of organisms in freshwaters (Voelz & Mcarthur, 2000; Strayer, 2006) that affect important ecosystem processes like detritus breakdown, nutrient mineralisation, and even primary production via grazing. Macroinvertebrates have a range of feeding traits, or functional feeding groups, which based on their morphological characteristics and behavior are categorised into shredders, filter feeders, gatherers, scrapers, and predators (Cummins & Klug, 1979; Vannote et al., 1980). Macroinvertebrates are also important food for higher trophic levels in the stream, as well as for terrestrial consumers as they also have an emergent adult stage (e.g. spiders and birds).
Macroinvertebrates can be very abundant, but this is highly dependent on habitat conditions and the amounts of energy available for fuelling the stream food web. With specific focus on the role of aquatic plants, Iversen et al. (1985) found that 95% of all macroinvertebrates in a small lowland stream were found within the stream vegetation, and Shupryt and Stelzer (2009) recorded almost three times higher numbers of macroinvertebrates in plant beds than in inorganic stream bed substrates. Plants also have strong indirect effects on macroinvertebrates as they modulate habitat conditions within the streams. They shape the physical environment by altering current velocities, sedimentation rates and substrate conditions, thereby creating a range of habitats that would not be present under the same geomorphic conditions without them (Gurnell et al., 2012). Hence, a number of studies have observed significant relationships between the morphology of the macrophytes and structural characteristics of the invertebrate assemblages (McAbendroth et al., 2005; Thomaz et al., 2008; Ferreiro et al., 2011; Mormul et al., 2011; Wolters et al., 2018). In general, macrophytes with a high structural complexity support a higher macroinvertebrate species richness and larger numbers of individuals compared to plants with a simpler architecture and low complexity (e.g. Rooke, 1986; Jeffries, 1993; Cheruvelil et al., 2002; Warfe & Barmuta, 2006; Germ et al. 2024). However, other studies have not demonstrated a higher abundance and richness of structurally complex species, reflecting that a number of other factors may affect these relationships (Stoner & Lewis, 1985; Chilton, 1990; Attrillet al., 2000; Taniguchi et al., 2003; Cremona et al., 2008; Verdonschot et al., 2012). For instance, macroinvertebrates of different body sizes might perceive the same macrophyte species to have different levels of structural complexity. That is, smaller-bodied organisms are favoured where gaps within the vegetation are smaller and more complex, whereas larger-bodied invertebrates find it more difficult to move around (McAbendroth et al., 2005). On top of this, macrophytes with different shapes may modify the food supply in different ways. For example, macrophytes with dissected leaves may trap higher amounts of detritus and support a higher epiphytic biomass and production than macrophytes with streamlined leaves (Cattaneo & Kalff, 1980; Gregg & Rose, 1982), and the combined effects of food supply, predation risk and available microhabitats provided by the macrophytes may therefore all contribute to shaping the macroinvertebrate community in addition to factors that operate at larger spatial scales (Verdonschot et al., 2012).
Although earlier studies have examined the importance of macrophyte morphology in creating suitable habitat for different organism groups (e.g. Beckett et al., 1992; Warfe & Barmuta, 2006; Bell et al., 2013) these have mainly focused on single-species beds of macrophytes. In the present study, we looked deeper into how plant diversity per se might shape the structural characteristics of the macroinvertebrate community. Specifically, we wanted to explore if and how macrophyte diversity can affect the number and richness of macroinvertebrate taxa, the abundance of different functional feeding groups as well as the number and richness of mayflies (Ephemeroptera), stoneflies (Plecoptera) and caddisflies (Trichoptera) that are particularly sensitive to changes in physical conditions as well as water quality. We conducted our study in four small lowland streams situated in catchments with predominantly agricultural land use. The riparian zone along these streams is quite narrow (2 m) and within-stream habitat diversity is mainly formed by beds of macrophytes (Sand-Jensen, 1997). Based on the knowledge, we expected that macrophyte beds supporting multiple species would foster a richer, more diverse, and functionally complex macroinvertebrate community compared to single-species macrophyte beds. To investigate this, we employed an in situ experimental approach, involving various macrophyte species with differing morphotypes. Specifically, we hypothesised that macrophyte beds consisting of several species representing different morphotypes would support a higher macroinvertebrate abundance and taxon richness compared to single-species plant beds. Moreover, we hypothesised that multi-species macrophyte beds would support more different functional feeding groups as multi-species beds likely create a larger range of feeding niches. To test these hypotheses, we introduced similar types of single- and multi-species experimental treatments into four stream reaches in different combinations, allowing us to directly examine the role of single- and multi-species treatments in situ for macroinvertebrate communities under controlled conditions.
Materials and methods
Experimental streams
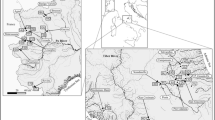
The experiment was conducted in four small (3–5 m wide) streams: Ejstrup Bæk (EJ), Grydeå (GR), Hjøllund Bæk (HJ) and Sunds Nørreå (SU) situated in Jutland in the western part of Denmark that is dominated by sandy outwash plains. The streams are meandering and unshaded from riparian vegetation. Physical and chemical characteristics of the streams are given in Table S1. There was no significant impact on water quality from effluents in the streams. All plant and invertebrate collection and use was in accordance with all the relevant guidelines and necessary permissions were obtained.
Plant habitats
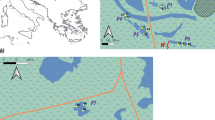
In each of the four study streams, seven different plant habitats were constructed using three species of plants with different shoot morphologies: Potamogeton alpinus Balbis (hereafter Potamogeton), Sparganium emersum Rehman (Sparganium) and Ranunculus peltatus Schrank (Ranunculus) (all species identified by A. Baattrup-Pedersen). Ranunculus has a high morphological complexity, plant perimeter and branching per area, while Potamogeton has medium complexity and Sparganium low complexity (Fig. 1). The plants were used alone to create single-species habitats (Sparganium, Potamogeton and Ranunculus) and in various combinations to create multi-species habitats (SP, SR, PR and SPR). Shoots of Sparganium, Potamogeton and Ranunculus were collected in April. A total of six 6–10 cm long shoots were planted in net pots (21 × 21 × 18 cm) with the lower 2 cm being carefully pushed into the sediment. The shoots were mixed in multi-species pots. The sediment was natural stream sediment rinsed and enriched with inorganic nutrients by adding slow-releasing fertiliser to support luxurious growth of the macrophytes. Before being transferred to the experimental stream sites, the plants were grown outdoors in water cultures (~ 500 L basins) enriched with CO2 (approximately 100 µM free-CO2) to resemble in situ growth conditions. The water was renewed regularly. Following 45 days of growth, total plant shoot density was adjusted to a similar level in all pots, and the density of individual species was adjusted to similar levels in the multi-species pots.
Experimental set-up used to study linkages between plant diversity and macroinvertebrate community characteristics. In each of the four study stream a total of seven different plant beds were constructed using three species of plants with different shapes: Potamogeton alpinus Balbis (P), Sparganium emersum Rehman (S) and Ranunculus peltatus Schrank (R). These species were used alone to create single-species habitats (S, P and R) and in various combinations to create multi-species habitats (SP, SR, PR and SPR). Two complete sets of treatments were placed in each of the four experimental streams with a minimum distance of 20 m between each replicate set of pots. The position of the pots was randomised both within replicate sets and among streams. The distance between the pots was approximately 40 cm to minimise interference between pots
Experimental streams
In May, two complete sets of treatments were placed in plant-free sections in each of the four experimental stream with a minimum of 20 m between each replicate set of pots (Fig. 1). The position of the pots was randomised both within the replicate sets and among streams. The distance between the pots was approximately 40 cm to minimise interference between pots. The depth position varied between 24 and 45 cm in EJ, between 39 and 55 cm in GR, between 26 and 39 cm in HJ and between 33 and 60 cm in SU. Following 7 days of exposure, the biomass of Potamogeton was significantly reduced in one of the streams (SU), probably due to herbivory from the caddisfly Anabolia nervosa (Curtis, 1834), an efficient grazer on Potamogeton species in Danish streams (Jacobsen & Friberg, 1995). To counteract the loss of Potamogeton within the timeframe of the experiment, these pots were supplemented with Elodea canadensis Michaux (Elodea), chosen because it establishes easily and grows fast. Therefore, treatments with Potamogeton in stream S consisted of a mixture of Potamogeton and Elodea.
Plant and associated macroinvertebrate sampling
Macroinvertebrates were sampled in the various treatments after 42 days in the streams using a 200 µm mesh net bag. The net was swiftly pushed vertically down through the water column, enclosing all plant material belonging to each pot. Following enclosure, the net bag was sealed at the base, and the plants were cut just above the sediment with a pair of scissors. The net bag was kept sealed and carefully moved on to the bank where the net was inverted to transfer plant biomass and associated macroinvertebrates into a sample container. Once returned to the laboratory, the plant samples were rinsed over a 200 µm net to remove macroinvertebrates. Only a few specimens were still attached after rinsing [primarily Simuliidae pupae and Acroloxus lacustris Linnaeus, 1758)], and these were removed using forceps. The samples were preserved in 70% alcohol in the laboratory until identification (invertebrates were identified by K. Bertram-Friis). Identification was undertaken to species or genus level for most taxa, but some taxa were identified to phylum (Nematoda), class (Oligochaeta and Copepoda), suborder (Cladocera), subfamily (Chironomidae) or genera (e.g., Hydrozoa, Bivalvia, Ostracoda and among the Diptera Limoniidae, Ceratopogonidae and Brachycera, being a suborder of Diptera) using up to 100 × magnification (M16, Leica, Mannheim, Germany) and appropriate identification keys.
Fresh and dry weight (after 48 h at 60 °C) of species-specific plant material was determined for each pot. For each pot, the surface area of each plant species was determined by first scanning each plant individual using a scanner (600 DPI; Genius HR6X; Paris, France), and then use a free software (ImageJ 4.0.4 for Windows, National Institutes of Health, Bethesda, MD) to calculate surface area. The surface area was then multiplied by two to count for surface area for both sides of the plants.
Data analysis
Initially we calculated a number of indices to characterise the macroinvertebrate community following taxonomic adjustment. These included total abundance, EPT abundance that combines the abundance of Ephemeroptera, Plecoptera and Trichoptera, taxonomic richness including all taxa and EPT taxa and, finally, the abundance of taxa in different functional feeding groups including: shredders (Shr), scrapers (Scr), gatherers (Gat), filter feeders (Fil) and predators (Pre) according to Dall et al. (1995) and unpublished material (see table S2). To test the hypotheses, we then conducted a series of mixed-effects models using Mixed Model in SAS (SAS Institute Inc. version 9.3, Cary, North Carolina, USA). Total abundance and EPT abundance were log (x + 1) transformed prior the analyses. Firstly, we tested the effect of stream site on total plant surface area (SA) treating block as random factor and the effect of treatment on SA treating stream and block as random factors. We then tested i) the linear effect of surface area and ii) the number of species in the treatments (1, 2 and 3) on community metrics followed by a one-way ANOVA with Tukey’s post hoc tests to test for effects of plant species composition (Sparganium, Potamogeton, Ranunculus, Sparganium-Potamogeton, Sparganium-Ranunuculus, Potamogeton-Ranunculus, Sparganium-Potamogeton-Ranunculus) on community metrics. To test for the effect of the number of plant species, surface area (SA) was entered as covariate in the model.
Results
Macrophyte treatments and macroinvertebrate community responses
We found significant differences among streams investigated in terms of surface areas obtained across the seven treatments after 42 days of growth, (Fig. 2; ANOVA, F = 9.66, P < 0.0001), indicating that site specific conditions influenced overall macrophyte growth. However, despite these stream specific patterns, we also found differences in the surface areas obtained among treatments (Fig. 2; ANOVA, F = 4.20, P < 0.05), and pairwise comparisons revealed that surface areas in the treatment with only Sparganium and Potamogeton were lower than in the Sparganium-Ranunculus treatment. In contrast, the total surface area did not vary with number of plant species in treatments (ANOVA, F = 2.29, P > 0.05).
The total surface areas of macrophytes in the seven treatments in the four study streams. The bars represent the minimum and maximum surface area obtained for each treatment and stream. White line in bars indicates mean values. See Fig. 1 for description of treatment abbreviations
Overall, we saw a high degree of variability in the macroinvertebrate community metrics among treatments (Fig. 3 and Fig. 4). The most obvious pattern was that Sparganium treatments, being the least complex morphotype, had the lowest number of associated macroinvertebrate taxa (median 13 taxa) compared to the other treatments (median values between 17 and 29 taxa) and also the lowest number of EPT taxa (median 3 taxa) compared to the other treatments (median values between 4 and 11 taxa; Fig. 3). The total abundance of macroinvertebrates varied less among the different treatments, but the abundance of EPT taxa was particularly high in the Sparganium-Potamogeton-Ranunculus treatment (median 120 individuals) and the Sparganium-Ranunculus treatment (median 135 individuals) (Fig. 3). For the functional feeding groups, the clearest response was that the abundance of filter feeders was particularly high in treatments with Sparganium (Fig. 4).
Box-whisker plots displaying total abundance (number of individuals) (A), abundance of EPT taxa (number of individuals) (B), number of taxa (C) and number of EPT taxa (D) for the four study streams. The box represents the upper and lower quartile, the white line the median and the bars the minimum and maximum. See Fig. 1 for description of treatment abbreviations. Different letters indicate significant differences among treatments (P < 0.05)
Box- whisker plots displaying the abundances (number of individuals) of shredders (A), filter feeders (B), gatherers (C), scrapers (D) and predators (E). The box represents the upper and lower quartile, the white line the median and the bars the minimum and maximum. See Fig. 1 for description of treatment abbreviations. Different letters indicate significant differences among treatments (P < 0.05)
Linkages between treatment characteristics and macroinvertebrate community characteristics
A large part of the variability found in the macroinvertebrate abundance and richness could be explained by differences in the total surface area among the treatments (Table 1; Fig. 5). We observed that total abundance and taxa richness increased significantly with the surface area (Fig. 5; Table 1; F = 23.45; P < 0.0001 and F = 12.01; P < 0.01, respectively) and so did the EPT abundance and taxa richness (Fig. 5; Table 1; F = 53.10; P < 0.0001 and F = 13.99; P < 0.001, respectively). The same pattern appeared when looking at the abundance of the functional feeding groups (Table 1). Here we observed that the abundance of both shredders (F = 16.22; P < 0.05), gatherers (F = 43.68; P < 0.0001), scrapers (F = 103.17; P < 0.0001) and predators (F = 39.57; P < 0.05) increased with increasing surface area, but also that this was not the case for the abundance of filter feeders (F = 2.35; P > 0.05).
Log–log plots of total macroinvertebrate abundance and abundance of EPT taxon groups (left) and log–log plots of taxon richness and richness of EPT taxon groups (right) against total surface area of the seven treatments including single-species habitats (Potamogeton, Sparganium and Ranunculus) and multi-species habitats (Sparganium-Potamogeton, Sparganium-Ranunculus, Potamogeton-Ranunculus and Sparganium-Potamogeton-Ranunculus). EPT includes combined Ephemeroptera, Plecoptera and Trichoptera in the samples
In addition to surface area, we found that the number of macrophyte species present in the treatments also played a significant role (i.e. one, two or three plant species; Table 2) for both the abundance and richness of the macroinvertebrate community and, consequently, that some of the observed variability in Fig. 5 could be attributed to macrophyte bed characteristics. When treating surface area as a covariate in the mixed model, we observed that total abundance and taxa richness increased significantly with the number of plant species (F = 12.01; P < 0.05 and F = 11.07; P < 0.05, respectively) and so did the EPT abundance and taxa richness (F = 13.99; P < 0.05 and F = 8.80; P < 0.05, respectively). The same pattern occurred when looking at the abundance of the functional feeding groups (Table 2). Here the abundance of both shredders (F = 10.12; P < 0.05), gatherers (F = 14.33; P < 0.05), scrapers (F = 9.84; P < 0.05) and predators (F = 13.56; P < 0.05) increased with the number of plant species, but this was not the case for the abundance of filter feeders (F = 1.98; P > 0.05).
Discussion
We observed that the compositional patterns of the macrophyte bed played a key role for the macroinvertebrate community in our four streams. The number and richness of macroinvertebrates increased with increasing surface area of the bed, which is well known (Cyr & Downing, 1988; Cheruvelil et al., 2002), but in addition, the number of plant species forming the bed also affected both the abundance and richness of macroinvertebrates thereby explaining some of the observed variability in the relationship between surface area and the abundance and richness of the macroinvertebrate community. These findings clearly demonstrate that the interplay between macrophytes and macroinvertebrates can be an important feature of lowland stream ecosystems and add to the current understanding that some plant species support greater numbers, higher diversity and greater biomass of organisms than other species (McGaha, 1952; Krull, 1970; Chilton, 1990). We were only partly able to confirm our hypotheses, however, since the stimulating effect of multi-species beds compared to single-species beds on macroinvertebrate abundance and taxon richness depended on the species forming the beds. Ranunculus alone, or in combinations with other macrophytes, gave a very similar response of the macroinvertebrate community and hence overruled the effect of treatments with multiple morphotypes. Our findings therefore support other studies in the value of complex-shaped Ranunculus as ecosystem engineers in lowland streams (Cheruvelil et al., 2002). However, when accounting for differences in surface area among the treatments, we found that both taxon richness and the abundance of different functional feeding groups (shredders, gatherers, scrapers and predators) can be similarly high in beds consisting of a combination of Potamogeton and Sparganium as in beds consisting of only Ranunculus.
Many previous studies have revealed that macrophytes with high structural complexity support a higher macroinvertebrate species richness and larger numbers of individuals compared to plants with a more simple architecture such as stream-lined leaves or stems (e.g. Cheruvelil et al., 2002; Warfe & Barmuta, 2006). Here we show that a similarly high abundance and richness may occur in beds consisting of two more simple-shaped macrophytes as in complex-shaped macrophytes, likely displaying that many joint effects associated with macrophyte shape and space can affect the macroinvertebrate community. Whilst many studies have tried to identify the mechanisms underlying differences in abundance and richness patterns in relation to differences in the complexity of single species of macrophytes, less is known about mixed beds of macrophytes. What we do know, however, is that macrophyte structural complexity influences several processes in freshwater ecosystems, which may in turn affect the macroinvertebrate community. For instance, macrophytes with a high structural complexity can provide a higher number of microhabitats for colonisation (McNett & Rypstra, 2000; McAbendroth et al., 2005), and this can lead to a higher degree of surface convolution and refuge space that may influence the abundance and diversity patterns of the macroinvertebrate community (Warfe et al., 2008). The physical structure of the macrophyte can also affect habitat characteristics like current velocity patterns, which may again affect both the taxonomic and functional characteristics of the macroinvertebrate community (Wolters et al., 2018). Thus, macrophytes directly affect flow velocity patterns by redirecting water around and through their structures and different morphotypes may create intricate channelling effects, diverting the flow in particular directions. This redirection can cause alterations in local flow velocities, creating areas of acceleration and deceleration around the plants (Sand-Jensen and Mebus, 1996). Irrespective of the underlying mechanisms, our results highlight that not only plant structural complexity but also the diversity of the plant community can play a role for the abundance and richness of the macroinvertebrate community and that a combination of simple- to medium-complex species can support just as many individuals and taxa as structurally complex species.
The different functional feeding groups were represented in all treatments independent of the identity and number of macrophyte species forming the habitat, and the abundance of both shredders, gatherers, scrapers and predators increased with both increasing surface area and the number of macrophyte species forming the bed. The only exception was abundance of filter feeders, which was similar among the treatments. Several species in the family Simuliidae were an abundant filter feeder in our streams, and they were often found on Sparganium as they provide suitable surface for attachment. The linkages between macrophyte communities and food supply seem obvious for some of the functional feeding groups. For example, filter feeders are more likely to be directly influenced by current velocity that is directly coupled to the availability of the food resources, than by the identity or the complexity of the macrophyte species, whereas the abundance of gatherers can be stimulated in complex macrophyte species like Ranunculus that acts as a good filter and trap detritus, which can be utilised as a food source (Rooke, 1984; 1986). The latter may be explained by the comparable high abundances of gatherers in treatments with Ranunculus present both alone and in combination with other species. Similarly, high complexity plants also provide more surface area available for periphyton growth, and this may increase the density of scrapers feeding on the periphyton and again explain the comparable high abundances of scrapers in treatments with Ranunculus alone and in combination with other species. However, when we take differences in surface area among treatments into account in the analyses, we still see a stimulating effect of complexity on the abundance of scrapers, so it is not only a matter of available surface area but also of the species forming the habitats. Hence, complex morphotypes may also stimulate the variation in food availability as also previously suggested by Hinojosa-Garro et al. (2010), who observed that plant complexity can stimulate the development of more heterogeneous algal assemblages.
Linkages between the macrophyte community and predator abundance can be more intricate. Complex habitats can provide a better environment for escaping predation by increasing the availability of hiding places and/or reducing predator–prey encounters, and the predation success can therefore be higher in spatially simple habitats where prey may be more easily detectable and susceptible to attacks by predators (Gilinsky, 1984). On the other hand, the body size of the macroinvertebrates will, to some extent, determine the habitat characteristics as animals perceive and use the habitat proportionally to their own body size (McAbendroth et al., 2005). In the present study, we found that predator communities were most abundant in the treatments with either high-complexity Ranunculus or in treatments with a combination of low- and medium-complexity species, indicating that predation success is just as high in high complexity single-species habitats with Ranunculus as in multi-species habitats, which was also observed previously for Coleoptera, Hemiptera and Odonata by Hinojosa-Garro et al. (2010) in multiple-species habitats with low- and medium-complexity Potamogeton and Sparganium. These findings could well be linked to the higher abundance and richer macroinvertebrate communities in these treatments, providing a more abundant and varied food supply for predators. However, more work is needed to establish realised feeding links of macroinvertebrates in macrophyte stands, for example by undertaking a thorough analysis of food web structure using gut contents or stable isotopes (e.g. Woodward, 2009).
Perspectives
Macrophytes play a crucial role in lowland streams by providing habitats, stabilising sediments, improving water quality, and enhancing biodiversity. Despite their crucial role, vegetation clearance is common in many lowland streams within agricultural landscapes, leading to significant ecological repercussions. We hope that our findings can help pave the way for more macrophyte-friendly management measures, that consider the stimulating role of macrophytes for other organism groups as shown in our study, not least in stream in agricultural catchments that are often characterised by a degraded physical environment.
Data availability
The datasets used and/or analysed during the current study are available from the corresponding author on request.
References
Attrill, M. J., J. A. Strong & A. A. Rowden, 2000. Are macroinvertebrate communities influenced by seagrass structural complexity? Ecography 23: 114–121.
Bell, N., T. Riis, A. M. Suren & A. Baattrup-Pedersen, 2013. Distribution of invertebrates within beds of two morphological contrasting stream macrophyte species. Fundamental and Applied Limnology 183: 309–321.
Birk, S., D. Chapman, L. Carvalho, B. M. Spears, H. E. Andersen, C. Argillier, & D. Hering, 2020. Impacts of multiple stressors on freshwater biota across spatial scales and ecosystems. Nature Ecology & Evolution 4(8): 1060–1068.
Cattaneo, A. & J. Kalff, 1980. The relative contribution of aquatic macrophytes and their epiphytes to the production of macrophyte beds. Limnology and Oceanography 25: 280–289.
Cheruvelil, K., P. Soranno, J. Madsen & M. Roberson, 2002. Plant architecture and epiphytic macroinvertebrate communities: the role of an exotic dissected macrophyte. Journal of the North American Benthological Society 21: 261–277.
Chilton, E. W., 1990. Macroinvertebrate communities associated with 3 aquatic Macrophytes (Ceratophyllum demersum, Myriophyllum spicatum, and Vallisneria americana) in Lake Onalaska, Wisconsin. Journal of Freshwater Ecology 5: 455–466.
Cremona, F., D. Planas & M. Lucotte, 2008. Biomass and composition of macroinvertebrate communities associated with different types of macrophyte architectures and habitats in a large fluvial lake. Fundamental and Applied Limnology 171: 119–130.
Cummins, K. W. & M. J. Klug, 1979. Feeding ecology of stream invertebrates. Annual Review of Ecology and Systematics 10: 147–172.
Cyr, H. & J. Downing, 1988. The abundance of phytophilous invertebrates on different species of submerged macrophytes. Freshwater Biology 20: 365–374.
Dall, P. C., T. M. Iversen, J. Kirkegaard, C. Lindegaard & J. Thorup, 1995. En oversigt over danske ferskvandsinvertebrater til brug ved bedømmelse af forurening i søer og vandløb, Københavns Universitet, København, Ferskvandsbiologisk Laboratorium:
Ferreiro, N., C. Feijoó, A. Giorgi, & L. Leggieri, 2011. Effects of macrophyte heterogeneity and food availability on structural parameters of the macroinvertebrate community in a Pampean stream. Hydrobiologia 664: 199–211.
Friberg, N., 2014. Impacts and indicators of change in lotic ecosystems. Wiley Interdisciplinary Reviews Water 1(6): 513–531.
Germ, M., Ž Tertinek & I. Zelnik, 2024. Diversity of macrophytes and macroinvertebrates in different types of standing waters in the drava field. Water 16: 1130.
Gilinsky, E., 1984. The role of fish predation and spatial heterogeneity in determining benthic community structure. Ecology 65: 455–468.
Gregg, W. W. & F. L. Rose, 1982. The effects of aquatic macrophytes on the stream micro-environment. Aquatic Botany 14: 309–324.
Gurnell, A. G., W. Bertoldi & D. Corenblit, 2012. Changing river channels: the roles of hydrological processes, plants and pioneer fluvial landforms in humid temperate, mixed load, gravel bed rivers. Earth Science Reviews 111: 129–141.
Haase, P., D. E. Bowler, N. J. Baker, N. Bonada, S. Domisch, J. R. Garcia Marquez & E. A. Welti, 2023. The recovery of European freshwater biodiversity has come to a halt. Nature 620(7974): 582–588.
Hinojosa-Garro, D., C. F. Mason & G. J. C. Underwood, 2010. Influence of macrophyte spatial architecture on periphyton and macroinvertebrate community structure in shallow water bodies under contrasting land management. Fundamental and Applied Limnology 177: 19–37.
Iversen, T. M., J. Thorup, T. Hansen, J. Lodal & J. Olsen, 1985. Quantitative estimates and community structure in a macrophyte rich stream. Archiv Für Hydrobiologie 102: 291–301.
Jacobsen, D. & N. Friberg, 1995. Food preference of the Trichopteran larva Anabolia nervosa from 2 streams with different food availability. Hydrobiologia 308: 139–144.
Jeffries, M., 1993. Invertebrate colonization of artificial pondweeds of differing fractal dimension. Oikos 67: 142–148.
McAbendroth, L., P. M. Ramsay, A. Foggo, S. D. Rundle & D. T. Bilton, 2005. Does macrophyte structural complexity drive invertebrate diversity, biomass and body size distributions? Oikos 111: 279–290.
Mcnett, B. J., & A. L. Rypstra, 2000. Habitat selection in a large orb‐weaving spider: vegetational complexity determines site selection and distribution. Ecological Entomology 25(4): 423–432.
Mormul, R. P., S. M. Thomaz, A. M. Takeda, & R. D. Behrend, 2011. Structural complexity and distance from source habitat determine invertebrate abundance and diversity. Biotropica 43(6): 738–745.
Reid, H. E., G. J. Brierley & I. K. G. Boothroyd, 2010. Influence of bed heterogeneity and habitat type on macroinvertebrate uptake in peri-urban streams. International Journal of Sediment Research 25: 203–220.
Rooke, J. B., 1984. The invertebrate fauna of 4 macrophytes in a lotic system. Freshwater Biology 14: 507–513.
Rooke, J. B., 1986. Macroinvertebrates associated with macrophytes and plastic imitations in the Ermaosa River, Ontario, Canada. Archiv Für Hydrobiologie 106: 307–325.
Sand-Jensen, K. & J. R. Mebus, 1996. Fine-scale patterns of water velocity within macrophyte patches in streams. Oikos 76: 169–180.
Sand-Jensen, K. (1997). Macrophytes as biological engineers in the ecology of Danish streams. In: Freshwater biology. Priorities and development in Danish research. Gad, København, pp 74–101
Shupryt, M. P. & R. S. Stelzer, 2009. Macrophyte beds contribute disproportionately to benthic invertebrate abundance and biomass in a sand plains stream. Hydrobiol 632: 329–339.
Strayer, D. L. 2006. Challenges for freshwater invertebrate conservation. Journal of the North American Benthological Society 25(2): 271–287.
Strayer, D. L., & D. Dudgeon, 2010. Freshwater biodiversity conservation: recent progress and future challenges. Journal of the North American Benthological Society 29(1): 344–358.
Stoner, A. W. & F. G. Lewis, 1985. The influence of quantitative and qualitative aspects of habitat complexity in tropical sea-grass meadows. Journal of Experimental Marine Biology and Ecology 94: 19–40.
Taniguchi, H., S. Nakano & M. Tokeshi, 2003. Influences of habitat complexity on the diversity and abundance of epiphytic invertebrates on plants. Freshwater Biology 48: 718–728.
Thomaz, S. D., E. D. Dibble, L. R. Evangelista, J. Higuti & L. M. Bini, 2008. Influence of aquatic macrophyte habitat complexity on invertebrate abundance and richness in tropical lagoons. Freshwater Biology 53: 358–367.
Vannote, R. L., G. W. Minshall, K. W. Cummins, J. R. Sedell & C. E. Cushing, 1980. The river continuum concept. Canadian Journal of Fisheries and Aquatic Sciences 37(1): 130–137.
Verdonschot, R. C. M., K. Didderen & P. F. M. Verdonschot, 2012. Importance of habitat structure as a determinant of the taxonomic and functional composition of lentic macroinvertebrate assemblages. Limnologica 42: 31–42.
Voelz, N. J., & J. V. McArthur, 2000. An exploration of factors influencing lotic insect species richness. Biodiversity & Conservation 9: 1543–1570.
Warfe, M. D. & L. A. Barmuta, 2006. Habitat structural complexity mediates food web dynamics in a freshwater macrophyte community. Oecologia 150: 141–154.
Warfe, D. M., L. A. Barmuta & S. Wotherspoon, 2008. Quantifying habitat structure: surface convolution and living space for species in complex environments. Oikos 117: 1764–1773.
WWF, 2020. Living Planet Report 2020 - Bending the curve of biodiversity loss. Almond, R.E.A., Grooten M. and Petersen, T. (Eds). WWF, Gland, Switzerland.
Wolters, J. W., R. C. M. Verdonschot, J. Schoelynck, P. F. M. Verdonschot & P. Meire, 2018. The role of macrophyte structural complexity and water flow velocity in determining the epiphytic macroinvertebrate community composition in a lowland stream. Hydrobiologia 806: 157–173.
Woodward, G., 2009. Biodiversity, ecosystem functioning and food webs in fresh waters: assembling the jigsaw puzzle. Freshwater Biology 54: 2171–2187.
Acknowledgements
The authors are grateful to the European Union 7th Framework Project REFORM (Grant agreement ID 282656; N. Friberg, A. Baattrup-Pedersen), and the Horizon 2020 project MERLIN (Grant agreement ID 101036337; A. Baattrup-Pedersen) for financial support. We thank Tinna Christensen for figure layout, Mette Bundgaard-Larsen and Anne Mette Poulsen for manuscript editing.
Funding
Open access funding provided by Aarhus Universitet. Data used in the manuscript were generated as part of a research project funded by the European Union 7th Framework Project REFORM (Grant agreement ID 282656; N. Friberg, A. Baattrup-Pedersen), and the Horizon 2020 project MERLIN (Grant agreement ID 101036337; A. Baattrup-Pedersen).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Data collection and analysis were performed by Annette Baattrup-Pedersen, Klaus Bertram Friis, and Nikolai Friberg. The first draft of the manuscript was written by Annette Baattrup-Pedersen and Tenna Riis and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest.
Additional information
Handling editor: Sally A. Entrekin
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Baattrup-Pedersen, A., Friis, K.B., Friberg, N. et al. Inter-linkages between in-stream plant diversity and macroinvertebrate communities. Hydrobiologia (2024). https://doi.org/10.1007/s10750-024-05700-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10750-024-05700-5