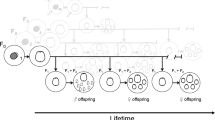
Abstract
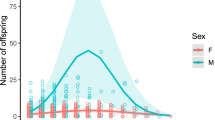
Aging is a significant internal factor influencing the sexual ability of rotifers. This study examined the effects of maternal aging on a rotifer's reproductive behavior (either sexual or asexual) and how the impacts were transmitted via generations (P − F1 − F2) in two marine species in the Brachionus plicatilis species complex: Brachionus plicatilis (large type) and Brachionus rotundiformis (super-small type). Based on the rotifer reproductive period, we established young and old lineages produced in the first and second halves of the period, respectively. The fecundity and longevity of the progeny in the two species and throughout the generations remained unaffected by maternal age. However, young amictic females in each species (especially in B. plicatilis) produced a larger ratio of mictic offspring. The multigenerational assessment revealed that the amictic offspring from old mothers produced more mictic daughters than those from young rotifers. This implies that aged rotifers may adopt an adaptive strategy (i.e., entering dormancy via sexual reproduction) for survival.





Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Alver, M. O. & A. Hagiwara, 2007. An individual-based population model for the prediction of rotifer population dynamics and resting egg production. Hydrobiologia 593: 19–26. https://doi.org/10.1007/s10750-007-9043-z.
Becks, L. & A. F. Agrawal, 2012. The evolution of sex is favored during adaptation to new environments. PLoS Biology 10: e1001317. https://doi.org/10.1371/journal.pbio.1001317.
Berkeley, S. A., C. Chapman & S. M. Sogard, 2004. Maternal age as a determinant of larval growth and survival in a marine fish, Sebastes melanops. Ecology 85: 1258–1264. https://doi.org/10.1890/03-0706.
Bock, M. J., G. C. Jarvis, E. L. Corey, E. E. Stone & K. E. Gribble, 2019. Maternal age alters offspring lifespan, fitness, and lifespan extension under caloric restriction. Scientific Reports 9: 3138. https://doi.org/10.1038/s41598-019-40011-z.
Carmona, M. J., A. Gomez & M. Serra, 1995. Mictic patterns of the rotifer Brachionus plicatilis Müller in small ponds. Hydrobiologia 313(314): 365–371. https://doi.org/10.1007/978-94-009-1583-1_47.
Clark, J., J. S. Garbutt, L. McNally & T. J. Little, 2017. Disease spread in age-structured populations with maternal age effects. Ecology Letters 20: 445–451. https://doi.org/10.1111/ele.12745.
Coakley, C. M., E. Nestoros & T. J. Little, 2018. Testing hypotheses for maternal effects in Daphnia magna. Journal of Evolutionary Biology 31: 211–216. https://doi.org/10.1111/jeb.13206.
Colinas, N., M. J. Carmona, M. Serra & E. M. García-Roger, 2023. Transgenerational effect on sexual reproduction in rotifer populations in relation to the environmental predictability of their habitats. Freshwater Biology 68: 1041–1054. https://doi.org/10.1111/fwb.14084.
Dahms, H.-U., A. Hagiwara & J.-S. Lee, 2011. Ecotoxicology, ecophysiology, and mechanistic studies with rotifers. Aquatic Toxicology 101: 1–12. https://doi.org/10.1016/j.aquatox.2010.09.006.
Declerck, S. A. J. & S. Papakostas, 2017. Monogonont rotifers as model systems for the study of micro-evolutionary adaptation and its eco-evolutionary implications. Hydrobiologia 796: 131–144. https://doi.org/10.1007/s10750-016-2782-y.
Fidhiany, L. & K. Winckler, 1998. Influence of body mass, age, and maturation on specific oxygen consumption in a freshwater cichlid fish, Cichlasoma nigrofasciatum (Guenther, 1869). Comparative Biochemistry and Physiology Part A 119: 613–619. https://doi.org/10.1016/S1095-6433(97)00474-1.
Fox, C. W., M. L. Bush & W. G. Wallin, 2003. Maternal age affects offspring lifespan of the seed beetle, Callosobruchus maculatus. Functional Ecology 17: 811–820. https://doi.org/10.1111/j.1365-2435.2003.00799.x.
Gilbert, J. J., 2003. Environmental and endogenous control of sexuality in a rotifer life cycle: developmental and population biology. Evolution & Development 5: 19–24. https://doi.org/10.1046/j.1525-142X.2003.03004.x.
Gilbert, J. J. & T. Schröder, 2004. Rotifers from diapausing, fertilized eggs: unique features and emergence. Limnology and Oceanography 49: 1341–1354. https://doi.org/10.4319/lo.2004.49.4_part_2.1341.
Gilbert, J. J. & T. Schröder, 2007. Intraclonal variation in propensity for mixis in several rotifers: variation among females and with maternal age. Hydrobiologia 593: 121–128.
Gribble, K. E., 2021. Brachionus rotifers as a model for investigating dietary and metabolic regulators of aging. Nutrition and Healthy Aging 6: 1–15. https://doi.org/10.3233/NHA-200104.
Gribble, K. E., G. Jarvis, M. Bock & D. B. Mark Welch, 2014. Maternal caloric restriction partially rescues the deleterious effects of advanced maternal age on offspring. Aging Cell 13: 623–630. https://doi.org/10.1111/acel.12217.
Hagiwara, A., M. D. Balompapueng, N. Munuswamy & K. Hirayama, 1997. Mass production and preservation of the resting eggs of the euryhaline rotifer Brachionus plicatilis and B. rotundiformis. Aquaculture 155: 223–230. https://doi.org/10.1016/S0044-8486(97)00119-1.
Hagiwara, A., W. G. Gallardo, M. Assavaaree, T. Kotani & A. B. de Araujo, 2001. Live food production in Japan: recent progress and future aspects. Aquaculture 200: 111–127. https://doi.org/10.1016/S0044-8486(01)00696-2.
Hagiwara, A., T. Kotani, T. W. Snell, M. Assava-Aree & K. Hirayama, 1995. Morphology, reproduction, genetics, and mating behavior of small, tropical marine Brachionus strains (Rotifera). Journal of Experimental Marine Biology and Ecology 194: 25–37. https://doi.org/10.1016/0022-0981(95)00081-X.
Hagiwara, A., Y. Kadota & A. Hino, 2005. Maternal effect by stem females in Brachionus plicatilis: effect of starvation on mixis induction in offspring. Hydrobiologia 546: 275–279. https://doi.org/10.1007/s10750-005-4208-0.
Han, C. & A. Hagiwara, 2023. Differing modes of growth performance induced by hypoxia and effects on oxidative defense and glycolysis metabolism in two marine Brachionus rotifer species. Aquaculture 567: 739286. https://doi.org/10.1016/j.aquaculture.2023.739286.
Han, C., H.-J. Kim, J.-S. Lee, Y. Sakakura & A. Hagiwara, 2021. Species-specific effects of iron on temperate and tropical marine rotifers in reproduction, lipid and ROS metabolisms. Chemosphere 277: 130317. https://doi.org/10.1016/j.chemosphere.2021.130317.
Han, C., H.-J. Kim, Y. Sakakura & A. Hagiwara, 2022. Species-specific ammonia tolerance in the marine rotifers Brachionus plicatilis and Brachionus rotundiformis: reproductive characteristics and its mechanisms. Aquaculture 550: 737837. https://doi.org/10.1016/j.aquaculture.2021.737837.
Hino, A. & R. Hirano, 1977. Ecological studies on the mechanism of bisexual reproduction in the rotifer Brachionus plicatilis. II. Effects of cumulative parthenogenetic generation on the frequency of bisexual reproduction. Nippon Suisan Gakkaishi 43: 1147–1155. https://doi.org/10.2331/suisan.43.1147.
Kamizono, S., E. O. Ogello, Y. Sakakura & A. Hagiwara, 2017. Effect of starvation and accumulation of generations on mixis induction in offspring of the monogonont rotifer Brachionus manjavacas hatched from resting eggs. Hydrobiologia 796: 93–97. https://doi.org/10.1007/s10750-016-3078-y.
Kim, H.-J. & A. Hagiwara, 2011. Effect of female aging on the morphology and hatchability of resting eggs in the rotifer Brachionus plicatilis Müller. Hydrobiologia 662: 107–111. https://doi.org/10.1007/s10750-010-0486-2.
Kirschner, J., D. Weber, C. Neuschl, A. Franke, M. Böttger, L. Zielke, E. Powalsky, M. Groth, D. Shagin, A. Petzold & N. Hartmann, 2012. Mapping of quantitative trait loci controlling lifespan in the short-lived fish Nothobranchius furzeri–a new vertebrate model for age research. Aging Cell 11: 252–261. https://doi.org/10.1111/j.1474-9726.2011.00780.x.
Koiso, M., M. Yoshikawa, H. Kuwada & A. Hagiwara, 2009. Effect of maternal diet on survival and life history parameters of next generations in the rotifer Brachionus plicatilis sp. complex. Nippon Suisan Gakkaishi 75: 828–833. https://doi.org/10.2331/suisan.75.828.
Lansing, A. I., 1947. A transmissible, cumulative, and reversible factor in aging. Journal of Gerontology 2: 228–239. https://doi.org/10.1093/geronj/2.3.228.
Li, W., C. Niu & S. Bian, 2022. Sex ratio in the mother’s environment affects offspring population dynamics: maternal effects on population regulation. Proceedings of the Royal Society B 289: 20212530. https://doi.org/10.1098/rspb.2021.2530.
Li, W., C. Niu & X. Li, 2019. Offspring of aged mothers of rotifer Brachionus calyciflorus shows lower sexual propensity than their elder siblings under crowded conditions. Journal of Oceanology and Limnology 37: 1604–1610. https://doi.org/10.1007/s00343-019-8203-0.
Lubzens, E., G. Minkoff & S. Marom, 1985. Salinity dependence of sexual and asexual reproduction in the rotifer Brachionus plicatilis. Marine Biology 85: 123–126. https://doi.org/10.1007/BF00397430.
Marshall, D. J. & T. Uller, 2007. When is a maternal effect adaptive? Oikos 116: 1957–1963. https://doi.org/10.1111/j.2007.0030-1299.16203.x.
McIntyre, G. & R. Gooding, 2000. Effects of maternal age on larval competitiveness in house flies. Heredity 85: 480–489. https://doi.org/10.1046/j.1365-2540.2000.00787.x.
Mills, S., J. A. Alcántara-Rodríguez, J. Ciros-Pérez, A. Gómez, A. Hagiwara, K. H. Galindo & E. J. Walsh, 2017. Fifteen species in one: deciphering the Brachionus plicatilis species complex (Rotifera, Monogononta) through DNA taxonomy. Hydrobiologia 796: 39–58. https://doi.org/10.1007/s10750-016-2725-7.
Moorad, J. A. & D. H. Nussey, 2016. Evolution of maternal effect senescence. Proceedings of the National Academy of Sciences 113: 362–367. https://doi.org/10.1073/pnas.1520494113.
Ortells, R., T. W. Snell, F. Gómez & M. Serra, 2000. Patterns of genetic differentiation in resting egg banks of a rotifer species complex in spain. Archiv Für Hydrobiologie 149: 529–551.
Plaistow, S. J., C. Shirley, H. Collin, S. J. Cornell & E. D. Harney, 2015. Offspring provisioning explains clone-specific maternal age effects on life history and life span in the water flea, Daphnia pulex. The American Naturalist 186: 376–389. https://doi.org/10.5061/dryad.3k8c0.
R Core Team 2021. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/.
Rougier, C. L. & R. Pourriot, 1977. Aging and control of the reproduction in Brachionus calyciflorus (Pallas) (Rotatoria). Experimental Gerontology 12: 137–151. https://doi.org/10.1016/0531-5565(77)90022-5.
Snell, T. W., 2014. Rotifers as models for the biology of aging. International Review of Hydrobiology 99: 84–95. https://doi.org/10.1002/iroh.201301707.
Stelzer, C. P., 2005. Evolution of rotifer life histories. Hydrobiologia 546: 335–346. https://doi.org/10.1007/s10750-005-4243-x.
Stelzer, C. P. & T. W. Snell, 2003. Induction of sexual reproduction in Brachionus plicatilis (Monogononta, Rotifera) by a density-dependent chemical cue. Limnology and Oceanography 48: 939–943. https://doi.org/10.4319/lo.2003.48.2.0939.
Sukhotin, A. A., D. Abele & H. O. Pörtner, 2002. Growth, metabolism and lipid peroxidation in Mytilus edulis: age and size effects. Marine Ecology Progress Series 226: 223–234. https://doi.org/10.3354/meps226223.
Yoshimura, K., A. Hagiwara, T. Yoshimatsu & C. Kitajima, 1996. Culture technology of marine rotifers and the implications for intensive culture of marine fish in Japan. Marine and Freshwater Research 47: 217–222. https://doi.org/10.1071/MF9960217.
Acknowledgements
The authors sincerely appreciate Hiroko Teraguchi and Masako Yokota for their technical assistance. We also sincerely thank Dr. Robert Nesta Kagali for his comments to improve the manuscript.
Funding
This research was supported by the Japan Society for the Promotion of Science KAKENHI (Grant Numbers JP20H03063 and JP20K21340) to Atsushi Hagiwara.
Author information
Authors and Affiliations
Contributions
Conceptualization: AH; Methodology: AH and CH; Formal analysis and investigation: CH; Writing—original draft preparation: CH; Writing—review and editing: CH and AH; Funding acquisition: AH; Resources: AH.
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that they have no competing financial interests or personal relationships relevant to the content of this article.
Additional information
Guest editors: Maria Špoljar, Diego Fontaneto, Elizabeth J. Walsh & Natalia Kuczyńska-Kippen / Diverse Rotifers in Diverse Ecosystems
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Han, C., Hagiwara, A. The influence of maternal aging on mixis induction in various generations of marine Brachionus rotifers. Hydrobiologia (2024). https://doi.org/10.1007/s10750-024-05480-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10750-024-05480-y