Abstract
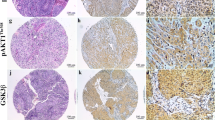
Phosphatidylinositol 3,4,5-trisphosphate-dependent Rac exchanger (P-Rex) proteins control many fundamental cellular functions including cell migration, actin cytoskeletal rearrangement and adhesion in many cancers. However, P-Rex1 expression and its prognostic effect and possible clinical value are not clearly elucidated in human oral squamous cell carcinoma (OSCC). Here, OSCC tissue microarrays were used to verify the expression levels of P-Rex1, coinhibitory immune checkpoints and tumor associated macrophage (TAM) markers, and to analyze the relationship between PREX1 expression levels and clinicopathological characteristics in OSCC. The study found that P-Rex1 expression was elevated in OSCC compared to dysplasia and normal mucosa (P < 0.0001). In addition, patients who expressed high PREX1 had a poorer prognosis than those who expressed low PREX1 (P = 0.0070). Furthermore, positive correlations were found between P-Rex1 expression and the immune checkpoints PD-L1, Galectin-9 and B7-H4, and the TAM markers CD68, CD206 and CD163. In short, these findings implicated that overexpression of P-Rex1 may predict a poor prognosis in human OSCC.




Similar content being viewed by others
References
Ali J, Sabiha B, Jan HU, Haider SA, Khan AA, Ali SS (2017) Genetic etiology of oral cancer. Oral Oncol 70:23–28. https://doi.org/10.1016/j.oraloncology.2017.05.004
Barrio-Real L, Benedetti LG, Engel N, Tu Y, Cho S, Sukumar S, Kazanietz MG (2014) Subtype-specific overexpression of the Rac-GEF P-REX1 in breast cancer is associated with promoter hypomethylation. Breast Cancer Res 16(5):441. https://doi.org/10.1186/s13058-014-0441-7
Bavle RM, Venugopal R, Konda P, Muniswamappa S, Makarla S (2016) Molecular classification of oral squamous cell carcinoma. J Clin Diagn Res 10(9):ZE18–ZE21. https://doi.org/10.7860/JCDR/2016/19967.8565
Budczies J, Klauschen F, Sinn BV, Gyorffy B, Schmitt WD, Darb-Esfahani S, Denkert C (2012) Cutoff Finder: a comprehensive and straightforward Web application enabling rapid biomarker cutoff optimization. PLoS ONE 7(12):e51862. https://doi.org/10.1371/journal.pone.0051862
Callejas-Valera JL, Iglesias-Bartolome R, Amornphimoltham P, Palacios-Garcia J, Martin D, Califano JA, Molinolo AA, Gutkind JS (2016) mTOR inhibition prevents rapid-onset of carcinogen-induced malignancies in a novel inducible HPV-16 E6/E7 mouse model. Carcinogenesis 37(10):1014–1025. https://doi.org/10.1093/carcin/bgw086
Chi AC, Day TA, Neville BW (2015) Oral cavity and oropharyngeal squamous cell carcinoma–an update. CA Cancer J Clin 65(5):401–421. https://doi.org/10.3322/caac.21293
Donald S, Hill K, Lecureuil C, Barnouin R, Krugmann S, John Coadwell W, Andrews SR, Walker SA, Hawkins PT, Stephens LR, Welch HC (2004) P-Rex2, a new guanine-nucleotide exchange factor for Rac. FEBS Lett 572(1–3):172–176. https://doi.org/10.1016/j.febslet.2004.06.096
Dong X, Mo Z, Bokoch G, Guo C, Li Z, Wu D (2005) P-Rex1 is a primary Rac2 guanine nucleotide exchange factor in mouse neutrophils. Curr Biol 15(20):1874–1879. https://doi.org/10.1016/j.cub.2005.09.014
Eisen MB, Spellman PT, Brown PO, Botstein D (1998) Cluster analysis and display of genome-wide expression patterns. Proc Natl Acad Sci USA 95(25):14863–14868. https://doi.org/10.1073/pnas.95.25.14863
Ferris RL (2015) Immunology and Immunotherapy of Head and Neck Cancer. J Clin Oncol 33(29):3293–3304. https://doi.org/10.1200/JCO.2015.61.1509
Goel HL, Pursell B, Shultz LD, Greiner DL, Brekken RA, Vander Kooi CW, Mercurio AM (2016) P-Rex1 promotes resistance to VEGF/VEGFR-targeted therapy in prostate cancer. Cell Rep 14(9):2193–2208. https://doi.org/10.1016/j.celrep.2016.02.016
Gulubova M, Ananiev J, Yovchev Y, Julianov A, Karashmalakov A, Vlaykova T (2013) The density of macrophages in colorectal cancer is inversely correlated to TGF-beta1 expression and patients’ survival. J Mol Histol 44(6):679–692. https://doi.org/10.1007/s10735-013-9520-9
Gupta S, Kong W, Peng Y, Miao Q, Mackillop WJ (2009) Temporal trends in the incidence and survival of cancers of the upper aerodigestive tract in Ontario and the United States. Int J Cancer 125(9):2159–2165. https://doi.org/10.1002/ijc.24533
Hill K, Krugmann S, Andrews SR, Coadwell WJ, Finan P, Welch HC, Hawkins PT, Stephens LR (2005) Regulation of P-Rex1 by phosphatidylinositol (3,4,5)-trisphosphate and Gbetagamma subunits. J Biol Chem 280(6):4166–4173. https://doi.org/10.1074/jbc.M411262200
Huang SH, O’Sullivan B (2017) Overview of the 8th Edition TNM classification for head and neck cancer. Curr Treat Options Oncol 18(7):40. https://doi.org/10.1007/s11864-017-0484-y
Inagi K, Takahashi H, Okamoto M, Nakayama M, Makoshi T, Nagai H (2002) Treatment effects in patients with squamous cell carcinoma of the oral cavity. Acta Otolaryngol 547:25–29. https://doi.org/10.1080/000164802760057527
Jaffe AB, Hall A (2005) Rho GTPases: biochemistry and biology. Annu Rev Cell Dev Biol 21:247–269. https://doi.org/10.1146/annurev.cellbio.21.020604.150721
Leemans CR, Braakhuis BJ, Brakenhoff RH (2011) The molecular biology of head and neck cancer. Nat Rev Cancer 11(1):9–22. https://doi.org/10.1038/nrc2982
Lindsay CR, Lawn S, Campbell AD, Faller WJ, Rambow F, Mort RL, Timpson P, Li A, Cammareri P, Ridgway RA, Morton JP, Doyle B, Hegarty S, Rafferty M, Murphy IG, McDermott EW, Sheahan K, Pedone K, Finn AJ, Groben PA, Thomas NE, Hao H, Carson C, Norman JC, Machesky LM, Gallagher WM, Jackson IJ, Van Kempen L, Beermann F, Der C, Larue L, Welch HC, Ozanne BW, Sansom OJ (2011) P-Rex1 is required for efficient melanoblast migration and melanoma metastasis. Nat Commun 2:555. https://doi.org/10.1038/ncomms1560
Liu JF, Ma SR, Mao L, Bu LL, Yu GT, Li YC, Huang CF, Deng WW, Kulkarni AB, Zhang WF, Sun ZJ (2017) T-cell immunoglobulin mucin 3 blockade drives an antitumor immune response in head and neck cancer. Mol Oncol 11(2):235–247. https://doi.org/10.1002/1878-0261.12029
Montero JC, Seoane S, Ocana A, Pandiella A (2011) P-Rex1 participates in Neuregulin-ErbB signal transduction and its expression correlates with patient outcome in breast cancer. Oncogene 30(9):1059–1071. https://doi.org/10.1038/onc.2010.489
Rhodes DR, Yu J, Shanker K, Deshpande N, Varambally R, Ghosh D, Barrette T, Pandey A, Chinnaiyan AM (2004) ONCOMINE: a cancer microarray database and integrated data-mining platform. Neoplasia 6(1):1–6. https://doi.org/10.1016/s1476-5586(04)80047-2
Rivera C, Venegas B (2014) Histological and molecular aspects of oral squamous cell carcinoma (Review). Oncol Lett 8(1):7–11. https://doi.org/10.3892/ol.2014.2103
Rosenfeldt H, Vazquez-Prado J, Gutkind JS (2004) P-REX2, a novel PI-3-kinase sensitive Rac exchange factor. FEBS Lett 572(1–3):167–171. https://doi.org/10.1016/j.febslet.2004.06.097
Rossman KL, Der CJ, Sondek J (2005) GEF means go: turning on RHO GTPases with guanine nucleotide-exchange factors. Nat Rev Mol Cell Biol 6(2):167–180. https://doi.org/10.1038/nrm1587
Ryan MB, Finn AJ, Pedone KH, Thomas NE, Der CJ, Cox AD (2016) ERK/MAPK signaling drives overexpression of the Rac-GEF, PREX1, in BRAF- and NRAS-mutant melanoma. Mol Cancer Res 14(10):1009–1018. https://doi.org/10.1158/1541-7786.MCR-16-0184
Saldanha AJ (2004) Java Treeview–extensible visualization of microarray data. Bioinformatics 20(17):3246–3248. https://doi.org/10.1093/bioinformatics/bth349
Shingaki S, Takada M, Sasai K, Bibi R, Kobayashi T, Nomura T, Saito C (2003) Impact of lymph node metastasis on the pattern of failure and survival in oral carcinomas. Am J Surg 185(3):278–284. https://doi.org/10.1016/s0002-9610(02)01378-8
Sosa MS, Lopez-Haber C, Yang C, Wang H, Lemmon MA, Busillo JM, Luo J, Benovic JL, Klein-Szanto A, Yagi H, Gutkind JS, Parsons RE, Kazanietz MG (2010) Identification of the Rac-GEF P-Rex1 as an essential mediator of ErbB signaling in breast cancer. Mol Cell 40(6):877–892. https://doi.org/10.1016/j.molcel.2010.11.029
Srijakotre N, Man J, Ooms LM, Lucato CM, Ellisdon AM, Mitchell CA (2017) P-Rex1 and P-Rex2 RacGEFs and cancer. Biochem Soc Trans 45(4):963–977. https://doi.org/10.1042/BST20160269
Sun ZJ, Zhang L, Hall B, Bian Y, Gutkind JS, Kulkarni AB (2012) Chemopreventive and Chemotherapeutic Actions of mTOR Inhibitor in Genetically Defined Head and Neck Squamous Cell Carcinoma Mouse Model. Clin Cancer Res 18(19):5304–5313. https://doi.org/10.1158/1078-0432.CCR-12-1371
Vigil D, Cherfils J, Rossman KL, Der CJ (2010) Ras superfamily GEFs and GAPs: validated and tractable targets for cancer therapy? Nat Rev Cancer 10(12):842–857. https://doi.org/10.1038/nrc2960
Welch HC, Coadwell WJ, Ellson CD, Ferguson GJ, Andrews SR, Erdjument-Bromage H, Tempst P, Hawkins PT, Stephens LR (2002) P-Rex1, a PtdIns(3,4,5)P3- and Gbetagamma-regulated guanine-nucleotide exchange factor for Rac. Cell 108(6):809–821. https://doi.org/10.1016/s0092-8674(02)00663-3
Welch HC, Condliffe AM, Milne LJ, Ferguson GJ, Hill K, Webb LM, Okkenhaug K, Coadwell WJ, Andrews SR, Thelen M, Jones GE, Hawkins PT, Stephens LR (2005) P-Rex1 regulates neutrophil function. Curr Biol 15(20):1867–1873. https://doi.org/10.1016/j.cub.2005.09.050
Welch HC (2015) Regulation and function of P-Rex family Rac-GEFs. Small GTPases 6(2):49–70. https://doi.org/10.4161/21541248.2014.973770
Wertheimer E, Gutierrez-Uzquiza A, Rosemblit C, Lopez-Haber C, Sosa MS, Kazanietz MG (2012) Rac signaling in breast cancer: a tale of GEFs and GAPs. Cell Signal 24(2):353–362. https://doi.org/10.1016/j.cellsig.2011.08.011
Wu CC, Li H, Xiao Y, Yang LL, Chen L, Deng WW, Wu L, Zhang WF, Sun ZJ (2018) Over-expression of IQGAP1 indicates poor prognosis in head and neck squamous cell carcinoma. J Mol Histol 49(4):389–398. https://doi.org/10.1007/s10735-018-9779-y
Wu JS, Sheng SR, Liang XH, Tang YL (2017) The role of tumor microenvironment in collective tumor cell invasion. Future Oncol 13(11):991–1002. https://doi.org/10.2217/fon-2016-0501
Wu L, Deng WW, Yu GT, Mao L, Bu LL, Ma SR, Liu B, Zhang WF, Sun ZJ (2016) B7-H4 expression indicates poor prognosis of oral squamous cell carcinoma. Cancer Immunol Immunother 65(9):1035–1045. https://doi.org/10.1007/s00262-016-1867-9
Wu L, Deng WW, Huang CF, Bu LL, Yu GT, Mao L, Zhang WF, Liu B, Sun ZJ (2017b) Expression of VISTA correlated with immunosuppression and synergized with CD8 to predict survival in human oral squamous cell carcinoma. Cancer Immunol Immunother 66(5):627–636. https://doi.org/10.1007/s00262-017-1968-0
Yoshizawa M, Kawauchi T, Sone M, Nishimura YV, Terao M, Chihama K, Nabeshima Y, Hoshino M (2005) Involvement of a Rac activator,P-Rex1, in neurotrophin-derived signaling and neuronal migration. J Neurosci 25(17):4406–4419. https://doi.org/10.1523/JNEUROSCI.4955-04.2005
Yu GT, Bu LL, Huang CF, Zhang WF, Chen WJ, Gutkind JS, Kulkarni AB, Sun ZJ (2015) PD-1 blockade attenuates immunosuppressive myeloid cells due to inhibition of CD47/SIRPalpha axis in HPV negative head and neck squamous cell carcinoma. Oncotarget 6(39):42067–42080. https://doi.org/10.18632/oncotarget.5955
Funding
This work was funded by the National Nature Science Foundation of China 81874131, 81672668. Z. J. Sun was supported by Fundamental Research Funds for the Central Universities of China 2042017kf0171 (Outstanding Young Scholars) and Hubei Province Nature Science Funds for Distinguished Young Scholar 2017CFA062.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study was authorized by the Medical Ethics Committee of the School and Hospital of Stomatology of Wuhan University, and informed consent was provided by each patient before surgery.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wan, SC., Wu, H., Li, H. et al. Overexpression of PREX1 in oral squamous cell carcinoma indicates poor prognosis. J Mol Hist 51, 531–540 (2020). https://doi.org/10.1007/s10735-020-09901-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-020-09901-9