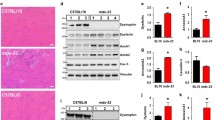
Abstract
Dysferlin is a sarcolemmal muscle protein associated with the processes of membrane repair, trafficking, and fusion of intracellular vesicles and muscle regeneration. Mutations in the DYSF gene cause clinically distinct forms of muscular dystrophies. The dysferlin-deficient SJL/J mouse model presents a reduction of 85% of the protein but shows mild weakness and discrete histopathological alterations. To study the effect of dysferlin deficiency in the muscle regenerative process, we used a model of electrical injury by electroporation to induce muscle degeneration/regeneration in the SJL/J mouse. The relative expression of the genes Pax7, MyoD, Myf5, and Myog was accompanied by the histopathological evaluation during muscle recovery at different time points after injury. We also investigated the effects of dysferlin deficiency in the expression of genes encoding FAM65B and HDAC6 proteins, recently described as forming a tricomplex with dysferlin at the beginning of myoblast differentiation. We observed an altered time course through the process of degeneration and regeneration in dysferlin-deficient mice, with remarkable regenerative capacity characterized by a faster and effective response in the first days after injury, as compared to the WT mice. Also, dysferlin deficiency seems to significantly alter the gene expression of Fam65b and Hdac6 during regeneration, since higher levels of expression of both genes were observed in dysferlin-deficient mice. These results need further attention to define their relevance in the disease mechanism.





Similar content being viewed by others
References
Almeida CF, Vainzof M. Skeletal muscle injury by electroporation—a model to study degeneration/regeneration pathways in muscle. Nucleic Acid Detection and Structural Investigations: methods and Protocols, Methods in Molecular Biology—in press, 2019
Anderson JE, Mitchell CM, McGeachie JK, Grounds MD (1995) The time course of basic fibroblast growth factor expression in crush-injured skeletal muscles of SJL/J and BALB/c mice. Exp Cell Res 216:325–334. https://doi.org/10.1006/excr.1995.1041
Balasubramanian A, Kawahara G, Gupta VA, Rozkalne A, Beauvais A, Kunkel LM et al (2014) Fam65b is important for formation of the HDAC6-dysferlin protein complex during myogenic cell differentiation. FASEB J 28:2955–2969
Bansal D, Miyake K, Vogel SS, Groh S, Chen C-C, Williamson R et al (2003) Defective membrane repair in dysferlin-deficient muscular dystrophy. Nature 423:168–172. https://doi.org/10.1038/nature01573
Bashir R, Britton S, Strachan T, Keers S, Vafiadaki E, Lako M et al (1998) A gene related to Caenorhabditis elegans spermatogenesis factor fer-1 is mutated in limb-girdle muscular dystrophy type 2B. Nat Genet 20:37–42. https://doi.org/10.1038/1689
Biondi O, Villemeur M, Marchand A, Chretien F, Bourg N, Gherardi RK et al (2013) Dual effects of exercise in dysferlinopathy. Am J Pathol 182:2298–2309. https://doi.org/10.1016/j.ajpath.2013.02.045
Cai C, Weisleder N, Ko J-K, Komazaki S, Sunada Y, Nishi M et al (2009) Membrane repair defects in muscular dystrophy are linked to altered interaction between MG53, caveolin-3, and dysferlin. J Biol Chem 284:15894–15902. https://doi.org/10.1074/jbc.M109.009589
Calyjur PC, Almeida CF, Ayub-Guerrieri D, Ribeiro AF Jr, de Fernandes SA, Ishiba R et al (2016) The mdx mutation in the 129/Sv background results in a milder phenotype: transcriptome comparative analysis searching for the protective factors. PLoS ONE 11:e0150748
Ceco E, McNally EM (2013) Modifying muscular dystrophy through transforming growth factor-beta. FEBS J 280:4198–4209. https://doi.org/10.1111/febs.12266
Chiu Y-H, Hornsey MA, Klinge L, Jorgensen LH, Laval SH, Charlton R et al (2009) Attenuated muscle regeneration is a key factor in dysferlin-deficient muscular dystrophy. Hum Mol Genet 18:1976–1989. https://doi.org/10.1093/hmg/ddp121
Cohen TV, Cohen JE, Partridge TA (2012) Myogenesis in dysferlin-deficient myoblasts is inhibited by an intrinsic inflammatory response. Neuromuscul Disord 22(7):648–658. https://doi.org/10.1016/j.nmd.2012.03.002
Czerwinska AM, Streminska W, Ciemerych MA, Grabowska I (2012) Mouse gastrocnemius muscle regeneration after mechanical or cardiotoxin injury. Folia Histochem Cytobiol 50:144–153. https://doi.org/10.5603/FHC.2012.0021
De Luna N, Gallardo E, Soriano M, Dominguez-Perles R, de La Torre C, Rojas-Garcia R et al (2006) Absence of dysferlin alters myogenin expression and delays human muscle differentiation “in vitro”. J Biol Chem 281:17092–17098. https://doi.org/10.1074/jbc.M601885200
Demonbreun AR, Quattrocelli M, Barefield DY, Allen MV, Swanson KE, McNally EM (2016) An actin-dependent annexin complex mediates plasma membrane repair in muscle. J Cell Biol 213:705–718. https://doi.org/10.1083/jcb.201512022
Di Fulvio S, Azakir BA, Therrien C, Sinnreich M (2011) Dysferlin interacts with histone deacetylase 6 and increases alpha-tubulin acetylation. PLoS ONE 6:e28563. https://doi.org/10.1371/journal.pone.0028563
Glover L, Brown RH (2007) Dysferlin in membrane trafficking and patch repair. Traffic 8:785–794. https://doi.org/10.1111/j.1600-0854.2007.00573.x
Grounds M, McGeachie J (1989) A comparison of muscle precursor replication in crush-injured skeletal muscle of Swiss and BALBc mice. Cell Tissue Res 255:385–391. https://doi.org/10.1007/BF00224122
Grounds MD, Terrill JR, Radley-Crabb HG, Robertson T, Papadimitriou J, Spuler S, Shavlakadze T (2014) Lipid acumulation in dysferlin-deficient muscles. Am J Pathol 84(6):1668–1676. https://doi.org/10.1016/j.ajpath.2014.02.005
Hardy D, Besnard A, Latil M, Jouvion G, Briand D, Thepenier C et al (2016) Comparative study of injury models for studying muscle regeneration in mice. PLoS ONE 11:e0147198. https://doi.org/10.1371/journal.pone.0147198
Hofhuis J, Bersch K, Büssenschütt R, Drzymalski M, Liebetanz D, Nikolaev VO et al (2017) Dysferlin mediates membrane tubulation and links T-tubule biogenesis to muscular dystrophy. J Cell Sci 130:841–852. https://doi.org/10.1242/jcs.198861
Huang Y, Verheesen P, Roussis A, Frankhuizen W, Ginjaar I, Haldane F et al (2005) Protein studies in dysferlinopathy patients using llama-derived antibody fragments selected by phage display. Eur J Hum Genet 13:721–730. https://doi.org/10.1038/sj.ejhg.5201414
Huang Y, Laval SH, van Remoortere A, Baudier J, Benaud C, Anderson LVB et al (2007) AHNAK, a novel component of the dysferlin protein complex, redistributes to the cytoplasm with dysferlin during skeletal muscle regeneration. FASEB J 21:732–742. https://doi.org/10.1096/fj.06-6628com
Illa I, Serrano-Munuera C, Gallardo E, Lasa A, Rojas-García R, Palmer J et al (2001) Distal anterior compartment myopathy: a dysferlin mutation causing a new muscular dystrophy phenotype. Ann Neurol 49:130–134
Kerr JP, Ward CW, Bloch RJ (2014) Dysferlin at transverse tubules regulates Ca(2 +) homeostasis in skeletal muscle. Front Physiol 5:89. https://doi.org/10.3389/fphys.2014.00089
Kobayashi K, Izawa T, Kuwamura M, Yamate J (2012) Dysferlin and animal models for dysferlinopathy. J Toxicol Pathol 25:135–147. https://doi.org/10.1293/tox.25.135
Lennon NJ, Kho A, Bacskai BJ, Perlmutter SL, Hyman BT, Brown RH Jr (2003) Dysferlin interacts with annexins A1 and A2 and mediates sarcolemmal wound-healing. J Biol Chem 278:50466–50473. https://doi.org/10.1074/jbc.M307247200
Liu J, Aoki M, Illa I, Wu C, Fardeau M, Angelini C et al (1998) Dysferlin, a novel skeletal muscle gene, is mutated in Miyoshi myopathy and limb girdle muscular dystrophy. Nat Genet 20:31–36. https://doi.org/10.1038/1682
Maley MA, Fan Y, Beilharz MW, Grounds MD (1994) Intrinsic differences in MyoD and myogenin expression between primary cultures of SJL/J and BALB/C skeletal muscle. Exp Cell Res 211:99–107. https://doi.org/10.1006/excr.1994.1064
Matsuda C, Hayashi YK, Ogawa M, Aoki M, Murayama K, Nishino I et al (2001) The sarcolemmal proteins dysferlin and caveolin-3 interact in skeletal muscle. Hum Mol Genet 10:1761–1766. https://doi.org/10.1093/hmg/10.17.1761
McGeachie JK, Grounds MD (1995) Retarded myogenic cell replication in regenerating skeletal muscles of old mice: an autoradiographic study in young and old BALBc and SJL/J mice. Cell Tissue Res 280(2):277–282. https://doi.org/10.1007/BF00307799
Mitchell CA, McGeachie JK, Grounds MD (1992) Cellular differences in the regeneration of murine skeletal muscle: a quantitative histological study in SJL/J and BALB/c mice. Cell Tissue Res 269:159–166. https://doi.org/10.1007/BF00384736
Mitchell CA, Grounds MD, Papadimitriou JM (1995) The genotype of bone marrow-derived inflammatory cells does not account for differences in skeletal muscle regeneration between SJL/J and BALB/c mice. Cell Tissue Res 280(2):407–413. https://doi.org/10.1007/BF00307814
Rawat R, Cohen TV, Ampong B, Francia D, Henriques-Pons A, Hoffman EP et al (2010) Inflammasome up-regulation and activation in dysferlin-deficient skeletal muscle. Am J Pathol 176:2891–2900. https://doi.org/10.2353/ajpath.2010.090058
Roberts P, McGeachie JK, Grounds MD (1997) The host environment determines strain-specific differences in the timing of skeletal muscle regeneration: cross-transplantation studies between SJL/J and BALB/c mice. J Anat 191(Pt 4):585–594. https://doi.org/10.1046/j.1469-7580.1997.19140585.x
Roche JA, Lovering RM, Bloch RJ (2008) Impaired recovery of dysferlin-null skeletal muscle after contraction-induced injury in vivo. NeuroReport 19:1579–1584. https://doi.org/10.1097/WNR.0b013e328311ca35
Roche JA, Lovering RM, Roche R, Ru LW, Reed PW, Bloch RJ (2010) Extensive mononuclear infiltration and myogenesis characterize recovery of dysferlin-null skeletal muscle from contraction-induced injuries. Am J Physiol Cell Physiol 298:312. https://doi.org/10.1152/ajpcell.00122.2009
Rudnicki MA, Le Grand F, McKinnell I, Kuang S (2008) The molecular regulation of muscle stem cell function. Cold Spring Harb Symp Quant Biol 73:323–331. https://doi.org/10.1101/sqb.2008.73.064
Sabourin LA, Rudnicki MA (2000) The molecular regulation of myogenesis. Clin Genet 57:16–25. https://doi.org/10.1034/j.1399-0004.2000.570103.x
Sellers SL, Milad N, White Z, Pascoe C, Chan R, Payne GW, Seow C, Rossi F, Seidman MA, Bernatchez P (2018) Increased nonHDL cholesterol levels cause muscle wasting and ambulatory dysfunction in the mouse model of LGMD2B. J Lipid Res 59(2):261–272
Terrill JR, Radley-Crabb HG, Iwasaki T, Lemckert FA, Arthur PG, Grounds MD (2013) Oxidative stress and pathology in muscular dystrophies: focus on protein thiol oxidation and dysferlinopathies. FEBS J 280(17):4149–4164. https://doi.org/10.1111/febs.12142
Vainzof M, Anderson LVB, McNally EM, Davis DB, Faulkner G, Valle G et al (2001) Dysferlin protein analysis in limb-girdle muscular dystrophies. J Mol Neurosci 17:71–80. https://doi.org/10.1385/JMN:17:1:71
Vainzof M, Ayub-Guerrieri D, Onofre PCG, Martins PCM, Lopes VF, Zilberztajn D et al (2008) Animal models for genetic neuromuscular diseases. J Mol Neurosci 34:241–248. https://doi.org/10.1007/s12031-007-9023-9
Weller AH, Magliato SA, Bell KP, Rosenberg NL (1997) Spontaneous myopathy in the SJL/J mouse. Pathol Strength Loss Muscle Nerve 20:72–82. https://doi.org/10.1002/(SICI)1097-4598(199701)20:1%3c72:AID-MUS10%3e3.0.CO;2-3
Acknowledgements
The authors would like to thank the scientific and technical support of the team of the Human Genome Research Center.
Funding
This work was supported by Conselho Nacional de Desenvolvimento Cientifico e Tecnológico (CNPq projetos 404664/2012-1 e 456522/2014-0), Fundação de Amparo à Pesquisa do Estado de São Paulo—Centro de Pesquisa, Inovação e Difusão (FAPESP-CEPID- projeto 2013/08028-1), CNPq-INCT (Projeto no: 610031/2009-0), Financiadora de Estudos e Projetos (FINEP—projeto 01.08.0579.00) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they do not have any competing or financial interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ishiba, R., Santos, A.L.F., Almeida, C.F. et al. Faster regeneration associated to high expression of Fam65b and Hdac6 in dysferlin-deficient mouse. J Mol Hist 50, 375–387 (2019). https://doi.org/10.1007/s10735-019-09834-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-019-09834-y