Abstract
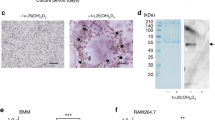
Bone is a metabolically active organ subjected to continuous remodeling process that involves resorption by osteoclast and subsequent formation by osteoblasts. Osteoclast involvement in this physiological event is regulated by macrophage colony-stimulating factor (M-CSF) and receptor activator of nuclear factor κB ligand (RANKL). Fusion of mono-nuclear pre-osteoclasts is a critical event for osteoclast differentiation and for bone resorption. Here we show that PBMCs can be successfully fused with polyethylenglicol (PEG) in order to generated viable osteoclast-like cells that exhibit tartrate-resistant acid phosphatase (TRAP) and bone resorptive activities. PEG-fused PBMCs expressed additional markers compatible with osteoclastogenic differentiation such as carbonic anhydrase II (CAII), calcitonin receptor (CR), cathepsin K (Cat K), vacuolar ATPase (V-ATPase) subunit C1 (V-ATPase), integrin β3, RANK and cell surface aminopeptidase N/CD13. Actin redistribution in PEG-fused cells was found to be affected by cell cycle synchronization at G0/G1 or G2/M phases. PEG-induced fusion also led to expression of tyrosine kinases c-Src and Syk in their phosphorylated state. Scanning electron microscopy images showed morphological features typical of osteoclast-like cells. The results here shown allow concluding that PEG-induced fusion of PBMCs provides a suitable model system for understanding the mechanisms involved in osteoclastogenesis and for assaying new therapeutic strategies.







Similar content being viewed by others
References
Berton G, Mócsai A, Lowell CA (2005) Src and Syk kinases: key regulators of phagocytic cell activation. Trends Immunol 26:208–214. doi:10.1016/j.it.2005.02.002
Biosse Duplan M, Zalli D, Stephens S et al (2014) Microtubule dynamic instability controls podosome patterning in osteoclasts through EB1, cortactin, and Src. Mol Cell Biol 34:16–29. doi:10.1128/MCB.00578-13
Boyce BF, Yoneda T, Lowe C et al (1992) Requirement of pp60c-src expression for osteoclasts to form ruffled borders and resorb bone in mice. J Clin Invest 90:1622–1627. doi:10.1172/JCI116032
Boyle WJ, Simonet WS, Lacey DL (2003) Osteoclast differentiation and activation. Nature 423:337–342. doi:10.1038/nature01658
Brunton VG, MacPherson IRJ, Frame MC (2004) Cell adhesion receptors, tyrosine kinases and actin modulators: a complex three-way circuitry. Biochim Biophys Acta - Mol. Cell Res 1692:121–144. doi:10.1016/j.bbamcr.2004.04.010
Campbell AM (1984) Fusion procedures. In: Burdon RH, Knippenberg PH (eds) Laboratory techniques in biochemistry and molecular biology: monoclonal antibody technology. Elsevier, Amsterdam, pp 120–134
Chambers TJ (2000) Regulation of the differentiation and function of osteoclasts. J Pathol 192:4–13. doi:10.1002/1096-9896(2000)9999:9999<::AID-PATH645>3.0.CO;2-Q
Collin O, Tracqui P, Stephanou A et al (2006) Spatiotemporal dynamics of actin-rich adhesion microdomains: influence of substrate flexibility. J Cell Sci 119:1914–1925. doi:10.1242/jcs.02838
D’Amico L, Roato I (2012) Cross-talk between T cells and osteoclasts in bone resorption. Bonekey Rep 1:82. doi:10.1038/bonekey.2012.82
Destaing O, Saltel F, Géminard J-C et al (2003) Podosomes display actin turnover and dynamic self- organization in osteoclasts expressing actin-green fluorescent protein V. Mol Biol Cell 14:407–416. doi:10.1091/mbc.E02
Edwards JR, Mundy GR (2011) Advances in osteoclast biology: old findings and new insights from mouse models. Nat Rev Rheumatol 7:235–243. doi:10.1038/nrrheum.2011.23
Faust J, Lacey DL, Hunt P et al (1999) Osteoclast markers accumulate on cells developing from human peripheral blood mononuclear precursors. J Cell Biochem 72:67–80
Gil-Henn H, Destaing O, Sims NA et al (2007) Defective microtubule-dependent podosome organization in osteoclasts leads to increased bone density in Pyk2(–/–) mice. J Cell Biol 178:1053–1064. doi:10.1083/jcb.200701148
Goldberg RD, Michelassi F, Montag AG (1991) Osteoclast-like giant cell tumor of the pancreas: Immunophenotypic similarity to giant cell tumor of bone. Hum Pathol 22:618–622. doi:10.1016/0046-8177(91)90243-I
Granot-Attas S, Luxenburg C, Finkelshtein E, Elson A (2009) Protein tyrosine phosphatase epsilon regulates integrin-mediated podosome stability in osteoclasts by activating Src. Mol Biol Cell 20:4324–4334. doi:10.1091/mbc.E08-11-1158
Heinemann C, Heinemann S, Worch H et al (2011) Development of an osteoblast/osteoclast co-culture derived by human bone marrow stromal cells and human monocytes for biomaterials testing. Eur Cell Mater 21:80–93 [pii]
Hirayama T, Sabokbar A, Athanasou NA (2000) Humoral factors act directly on circulating osteoclast precursors to control osteoclast formation. Calcif Tissue Int 66:(1) 212
Holtrop ME, Raisz LG, Simmons HA (1974) The effects of parathyroid hormone, colchicine, and calcitonin on the ultrastructure and the activity of osteoclasts in organ culture. J Cell Biol 60:346–355
Home WC, Neff L, Chatterjee D et al (1992) Osteoclasts express high levels of pp60c-src in association with intracellular membranes. J Cell Biol 119:1003–1013
Horton MA, Rimmer EF, Lewis D et al (1984) Cell surface characterization of the human osteoclast: Phenotypic relationship to other bone marrow-derived cell types. J Pathol 144:281–294. doi:10.1002/path.1711440410
Hu S, Planus E, Georgess D et al (2011) Podosome rings generate forces that drive saltatory osteoclast migration. Mol Biol Cell 22:3120–3126. doi:10.1091/mbc.E11-01-0086
Imai Y, Youn M-Y, Inoue K et al (2013) Nuclear receptors in bone physiology and diseases. Physiol Rev 93:481–523. doi:10.1152/physrev.00008.2012
Izawa T, Zou W, Chappel JC et al (2012) c-Src links a RANK/αvβ3 integrin complex to the osteoclast cytoskeleton. Mol Cell Biol 32:2943–2953. doi:10.1128/MCB.00077-12
Jurdic P, Saltel F, Chabadel A, Destaing O (2006) Podosome and sealing zone: specificity of the osteoclast model. Eur J Cell Biol 85:195–202. doi:10.1016/j.ejcb.2005.09.008
Kleinhans C, Schmid FF, Schmid FV, Kluger PJ (2015) Comparison of osteoclastogenesis and resorption activity of human osteoclasts on tissue culture polystyrene and on natural extracellular bone matrix in 2D and 3D. J Biotechnol 205:101–110. doi:10.1016/j.jbiotec.2014.11.039
Koga T, Inui M, Inoue K et al (2004) Costimulatory signals mediated by the ITAM motif cooperate with RANKL for bone homeostasis. Nature 428:758–763. doi:10.1038/nature02444
Kudo O, Fujikawa Y, Itonaga I et al (2002) Proinflammatory cytokine (TNFalpha/IL-1alpha) induction of human osteoclast formation. J Pathol 198:220–227. doi:10.1002/path.1190
Lee S-H, Rho J, Jeong D et al (2007) v-ATPase V0 subunit d2–deficient mice exhibit impaired osteoclast fusion and increased bone formation. Nat Med 12:1403–1409. doi:10.1038/nm1514
Lee YG, Chain BM, Cho JY (2009) Distinct role of spleen tyrosine kinase in the early phosphorylation of inhibitor of κBα via activation of the phosphoinositide-3-kinase and Akt pathways. Int J Biochem Cell Biol 41:811–821. doi:10.1016/j.biocel.2008.08.011
Lee J-O, Jeong D, Kim M-Y, Cho JY (2015) ATP-binding pocket-targeted suppression of Src and Syk by luteolin contributes to its anti-inflammatory action. Mediators Inflamm 967053. doi:10.1155/2015/967053
Linder S (2009) Invadosomes at a glance. J Cell Sci 122:3009–3013. doi:10.1242/jcs.032631
Long MA, Rossi FMV (2011) Targeted cell fusion facilitates stable heterokaryon generation in vitro and in vivo. PLoS ONE 6:e26381. doi:10.1371/journal.pone.0026381
Lorenzo JA, Sousa SL, Alander C et al (1987) Comparison of the bone-resorbing activity in the supernatants from phytohemagglutinin-stimulated human peripheral blood mononuclear cells with that of cytokines through the use of an antiserum to inter leukin 1. Endocrinology 121:1164–1170. doi:10.1210/endo-121-3-1164
Luo J, Yang Z, Ma Y et al (2016) LGR4 is a receptor for RANKL and negatively regulates osteoclast differentiation and bone resorption. Nat Med 22:539–546
Luxenburg C, Parsons JT, Addadi L, Geiger B (2006) Involvement of the Src-cortactin pathway in podosome formation and turnover during polarization of cultured osteoclasts. J Cell Sci 119:4878–4888. doi:10.1242/jcs.03271
Luxenburg C, Geblinger D, Klein E et al (2007) The architecture of the adhesive apparatus of cultured osteoclasts: From podosome formation to sealing zone assembly. PLoS ONE 2:e179. doi:10.1371/journal.pone.0000179
Manrique E, Castillo LM, Lazala O, et al (2016) Bone resorptive activity of human peripheral blood mononuclear cells after fusion with polyethylene glycol. J Bone Miner Metab 1–15. doi:10.1007/s00774-016-0744-0
Matayoshi A, Brown C, DiPersio JF et al (1996) Human blood-mobilized hematopoietic precursors differentiate into osteoclasts in the absence of stromal cells. Proc Natl Acad Sci USA 93:10785–10790
Matsumoto T, Nagase Y, Hirose J et al (2013) Regulation of bone resorption and sealing zone formation in osteoclasts occurs through protein kinase b–mediated microtubule stabilization. J Bone Miner Res 28:1191–1202. doi:10.1002/jbmr.1844
Mercer WE, Schlegel RA (1979) Phytohemagglutinin enhancement of cell fusion reduces polyethylene glycol cytotoxicity. Exp Cell Res 120:417–421. doi:10.1016/0014-4827(79)90403-8
Miyazaki T, Tanaka S, Sanjay A, Baron R (2006) The role of c-Src kinase in the regulation of osteoclast function. Mod Rheumatol 16:68. doi:10.1007/s10165-006-0460-z
Mócsai A, Humphrey MB, Van Ziffle JAG et al (2004) The immunomodulatory adapter proteins DAP12 and Fc receptor γ-chain (FcRγ) regulate development of functional osteoclasts through the Syk tyrosine kinase. Proc Natl Acad Sci USA 101:6158–6163. doi:10.1073/pnas.0401602101
Mulari MTK, Zhao H, Lakkakorpi PT, Väänänen HK (2003) Osteoclast ruffled border has distinct subdomains for secretion and degraded matrix uptake. Traffic 4:113–125. doi:10.1034/j.1600-0854.2003.40206.x
Murillo A, Guerrero CA, Acosta O, Cardozo CA (2010) Bone resorptive activity of osteoclast-like cells generated in vitro by PEG-induced macrophage fusion. Biol Res 43:205–224. doi:10.4067/S0716-97602010000200009
Nakamura I, Takahashi N, Sasaki T et al (1996) Chemical and physical properties of the extracellular matrix are required for the actin ring formation in osteoclasts. J Bone Miner Res 11:1873–1879. doi:10.1002/jbmr.5650111207
Nicholson GC, Malakellis M, Collier FM et al (2000) Induction of osteoclasts from CD14-positive human peripheral blood mononuclear cells by receptor activator of nuclear factor kappaB ligand (RANKL). Clin Sci 99:133–140
Okumura S, Mizoguchi T, Sato N et al (2006) Coordination of microtubules and the actin cytoskeleton is important in osteoclast function, but calcitonin disrupts sealing zones without affecting microtubule networks. Bone 39:684–693. doi:10.1016/j.bone.2006.04.010
Pfaff M, Jurdic P (2001) Podosomes in osteoclast-like cells: structural analysis and cooperative roles of paxillin, proline-rich tyrosine kinase 2 (Pyk2) and integrin alphaVbeta3. J Cell Sci 114:2775–2786
Raggatt LJ, Partridge NC (2010) Cellular and molecular mechanisms of bone remodeling. J Biol Chem 285:25103–25108. doi:10.1074/jbc.R109.041087
Reeve JL, Zou W, Liu Y et al (2009) SLP-76 couples Syk to the osteoclast cytoskeleton. J Immunol 183:1804–1812. doi:10.4049/jimmunol.0804206
Saltel F, Chabadel A, Bonnelye E, Jurdic P (2008) Actin cytoskeletal organisation in osteoclasts: a model to decipher transmigration and matrix degradation. Eur J Cell Biol 87:459–468. doi:10.1016/j.ejcb.2008.01.001
Sanjay A, Houghton A, Neff L et al (2001) Cbl associates with Pyk2 and Src to regulate Src kinase activity, α(v)β(3) integrin-mediated signaling, cell adhesion, and osteoclast motility. J Cell Biol 152:181–196
Schratt G, Philippar U, Berger J et al (2002) Serum response factor is crucial for actin cytoskeletal organization and focal adhesion assembly in embryonic stem cells. J Cell Biol 156:737–750. doi:10.1083/jcb.200106008
Susa M, Luong-Nguyen N-H, Cappellen D et al (2004) Human primary osteoclasts: in vitro generation and applications as pharmacological and clinical assay. J Transl Med 2:6. doi:10.1186/1479-5876-2-6
Tehrani S, Faccio R, Chandrasekar I et al (2006) Cortactin has an essential and specific role in osteoclast actin assembly. Mol Biol Cell 17:2882–2895. doi:10.1091/mbc.E06-03-0187
Teitelbaum SL (2007) Osteoclasts: what do they do and how do they do it?. Am J Pathol 170:427–435. doi:10.2353/ajpath.2007.060834
Teitelbaum SL, Ross FP (2003) Genetic regulation of osteoclast development and function. Nat Rev Genet 4:638–649. doi:10.1038/nrg1122
Vielreicher M, Harms G, Butt E et al (2007) Dynamic interaction between Src and C-terminal Src kinase in integrin αIIbβ3-mediated signaling to the cytoskeleton. J Biol Chem 282:33623–33631. doi:10.1074/jbc.M704107200
Wilson SR, Peters C, Saftig P, Brömme D (2009) Cathepsin K activity-dependent regulation of osteoclast actin ring formation and bone resorption. J Biol Chem 284:2584–2592. doi:10.1074/jbc.M805280200
Xing L, Xiu Y, Boyce BF (2012) Osteoclast fusion and regulation by RANKL-dependent and independent factors. World J Orthop 3:212–222. doi:10.5312/wjo.v3.i12.212
Yagi M, Miyamoto T, Sawatani Y et al (2005) DC-STAMP is essential for cell–cell fusion in osteoclasts and foreign body giant cells. J Exp Med 202:345–351. doi:10.1084/jem.20050645
Zhao C, Irie N, Takada Y et al (2006) Bidirectional ephrinB2-EphB4 signaling controls bone homeostasis. Cell Metab 4:111–121. doi:10.1016/j.cmet.2006.05.012
Zhuang Y, Mwangi W, Brown WC et al (2006) Characterization of a phenotypically unique population of CD13(+) dendritic cells resident in the spleen. Clin Vaccine Immunol 13:1064–1069. doi:10.1128/CVI.00178-06
Zou W, Kitaura H, Reeve J et al (2007) Syk, c-Src, the αvβ3 integrin, and ITAM immunoreceptors, in concert, regulate osteoclastic bone resorption. J Cell Biol 176:877–888. doi:10.1083/jcb.200611083
Zou W, Reeve JL, Liu Y et al (2008) DAP12 couples c-Fms activation to the osteoclast cytoskeleton by recruitment of Syk. Mol Cell 31:422–431. doi:10.1016/j.molcel.2008.06.023
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Castillo, L.M., Guerrero, C.A. & Acosta, O. Expression of typical osteoclast markers by PBMCs after PEG-induced fusion as a model for studying osteoclast differentiation. J Mol Hist 48, 169–185 (2017). https://doi.org/10.1007/s10735-017-9717-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-017-9717-4