Abstract
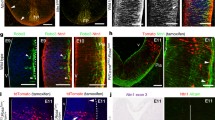
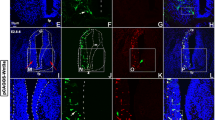
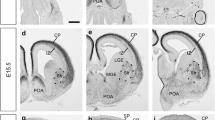
N-cadherin is a calcium-sensitive cell adhesion molecule that plays an important role in the formation of the neural circuit and the development of the nervous system. In the present study, we investigated the function of N-cadherin in cell–cell connection in vitro with HEK293T cells, and in commissural axon projections in the developing chicken spinal cord using in ovo electroporation. Cell–cell connections increased with N-cadherin overexpression in HEK293T cells, while cell contacts disappeared after co-transfection with an N-cadherin-shRNA plasmid. The knockdown of N-cadherin caused the accumulation of β-catenin in the nucleus, supporting the notion that N-cadherin regulates β-catenin signaling in vitro. Furthermore, N-cadherin misexpression perturbed commissural axon projections in the spinal cord. The overexpression of N-cadherin reduced the number of axons that projected alongside the contralateral margin of the floor plate, and formed intermediate longitudinal commissural axons. In contrast, the knockdown of N-cadherin perturbed commissural axon projections significantly, affecting the projections alongside the contralateral margin of the floor plate, but did not affect intermediate longitudinal commissural axons. Taken together, these findings suggest that N-cadherin regulates commissural axon projections in the developing chicken spinal cord.







Similar content being viewed by others
References
Arikkath J (2010) N-cadherin: stabilizing synapses. J Cell Biol 189:397–398. doi:10.1083/jcb.201004022
Avraham O, Zisman S, Hadas Y, Vald L, Klar A (2010) Deciphering axonal pathways of genetically defined groups of neurons in the chick neural tube utilizing in ovo electroporation. J Vis Exp. doi:10.3791/1792
Baum B, Georgiou M (2011) Dynamics of adherens junctions in epithelial establishment, maintenance, and remodeling. J Cell Biol 192:907–917. doi:10.1083/jcb.201009141
Bozdagi O, Shan W, Tanaka H, Benson DL, Huntley GW (2000) Increasing numbers of synaptic puncta during late-phase LTP: N-cadherin is synthesized, recruited to synaptic sites, and required for potentiation. Neuron 28:245–259
Colamarino SA, Tessier-Lavigne M (1995) The role of the floor plate in axon guidance. Annu Rev Neurosci 18:497–529. doi:10.1146/annurev.ne.18.030195.002433
Cui Y, Yamada S (2013) N-cadherin dependent collective cell invasion of prostate cancer cells is regulated by the N-terminus of α-catenin. PLoS One. doi:10.1371/journal.pone.0055069
Derycke LD, Bracke ME (2004) N-cadherin in the spotlight of cell–cell adhesion, differentiation, embryogenesis, invasion and signalling. Int J Dev Biol 48:463–476. doi:10.1387/ijdb.041793ld
Doherty P, Walsh FS (1996) CAM-FGF receptor interactions: a model for axonal growth. Mol Cell Neurosci 8:99–111. doi:10.1006/mcne.1996.0049
Flannery RJ, Bruses JL (2012) N-cadherin induces partial differentiation of cholinergic presynaptic terminals in heterologous cultures of brainstem neurons and CHO cells. Front Synaptic Neurosci 4:6. doi:10.3389/fnsyn.2012.00006.eCollection
Gartner A, Fornasiero EF, Dotti CG (2012) N-cadherin: a new player in neuronal polarity. Cell Cycle 11:2223–2224. doi:10.4161/cc.20797
Gooding JM, Yap KL, Ikura M (2004) The cadherin-catenin complex as a focal point of cell adhesion and signalling: new insights from three-dimensional structures. BioEssays 26:497–511. doi:10.1002/bies.20033
Gumbiner BM (2005) Regulation of cadherin-mediated adhesion in morphogenesis. Nat Rev Mol Cell Biol 6:622–634. doi:10.1038/nrm1699
Hamburger V, Hamilton HL (1992) A series of normal stages in the development of the chick embryo. 1951. Dev Dyn 195(4):231–272. doi:10.1002/aja.1001950404
Harris TJ, Tepass U (2010) Adherens junctions: from molecules to morphogenesis. Nat Rev Mol Cell Biol 11:502–514. doi:10.1038/nrm2927
Hatta K, Takeichi M (1986) Expression of N-cadherin adhesion molecules associated with early morphogenetic events in chick development. Nature 320(6061):447–449. doi:10.1038/320447a0
Hatta K, Takagi S, Fujisawa H, Takeichi M (1987) Spatial and temporal expression pattern of N-cadherin cell adhesion molecules correlated with morphogenetic processes of chicken embryos. Dev Biol 120:215–227
Hatta K, Nose A, Nagafuchi A, Takeichi M (1988) Cloning and expression of cDNA encoding a neural calcium-dependent cell adhesion molecule: its identity in the cadherin gene family. J Cell Biol 106:873–881
Hirata E, Park D, Sahai E (2014) Retrograde flow of cadherins in collective cell migration. Nat Cell Biol 16:621–623. doi:10.1038/ncb2995
Jungling K, Eulenburg V, Moore R, Kemler R, Lessmann V, Gottmann K (2006) N-cadherin transsynaptically regulates short-term plasticity at glutamatergic synapses in embryonic stem cell-derived neurons. J Neurosci 26:6968–6978. doi:10.1523/JNEUROSCI.1013-06.2006
Kadison SR, Kaprielian Z (2004) Diversity of contralateral commissural projections in the embryonic rodent spinal cord. J Comp Neurol 472:411–422. doi:10.1002/cne.20086
Lammens T, Swerts K, Derycke L, De Craemer A, De Brouwer S, De Preter K, Van Roy N, Vandesompele J, Speleman F, Philippe J, Benoit Y, Beiske K, Bracke M, Laureys G (2012) N-cadherin in neuroblastoma disease: expression and clinical significance. PLoS One 7:e31206. doi:10.1371/journal.pone.0031206
Le Dréau G, Garcia-Campmany L, Rabadán M, Ferronha T, Tozer S, Briscoe J, Martí E (2012) Canonical BMP7 activity is required for the generation of discrete neuronal populations in the dorsal spinal cord. Development 139:259–268. doi:10.1242/dev.074948
Lin J, Wang C, Redies C (2014) Restricted expression of classic cadherins in the spinal cord of the chicken embryo. Front Neuroanat 8:18. doi:10.3389/fnana.2014.00018
Lin J, Wang C, Yang C, Fu S, Redies C (2016) Pax3 and Pax7 interact reciprocally and regulate the expression of cadherin-7 through inducing neuron differentiation in the developing chicken spinal cord. J Comp Neurol 524(5):940–962. doi:10.1002/cne.23885
Logan CY, Nusse R (2004) The Wnt signaling pathway in development and disease. Annu Rev Cell Dev Biol 20:781–810. doi:10.1146/annurev.cellbio.20.010403.113126
Luo J, Ju MJ, Redies C (2006) Regionalized cadherin-7 expression by radial glia is regulated by Shh and Pax7 during chicken spinal cord development. Neuroscience 142(4):1133–1143. doi:10.1016/j.neuroscience.2006.07.038
Mendez P, De Roo M, Poglia L, Klauser P, Muller D (2010) N-cadherin mediates plasticity-induced long-term spine stabilization. J Cell Biol 189:589–600. doi:10.1083/jcb.201003007
Nelson WJ, Nusse R (2004) Convergence of Wnt, beta-catenin, and cadherin pathways. Science 303:1483–1487. doi:10.1126/science.1094291
Nollet F, Kools P, van Roy F (2000) Phylogenetic analysis of the cadherin superfamily allows identification of six major subfamilies besides several solitary members. J Mol Biol 299:551–572. doi:10.1006/jmbi.2000.3777
Okamura K, Tanaka H, Yagita Y, Saeki Y, Taguchi A, Hiraoka Y, Zeng L, Colman D, Miki N (2004) Cadherin activity is required for activity-induced spine remodeling. J Cell Biol 167:961–972. doi:10.1083/jcb.200406030
Ouyang M, Lu S, Kim T, Chen CE, Seong J, Leckband DE, Wang F, Reynolds AB, Schwartz MA, Wang Y (2013) N-cadherin regulates spatially polarized signals through distinct p120ctn and beta-catenin-dependent signalling pathways. Nat Commun 4:1589. doi:10.1038/ncomms2560
Pieters T, van Roy F (2014) Role of cell-cell adhesion complexes in embryonic stem cell biology. J Cell Sci 127:2603–2613. doi:10.1242/jcs.146720
Pokutta S, Weis WI (2007) Structure and mechanism of cadherins and catenins in cell–cell contacts. Annu Rev Cell Dev Biol 23:237–261. doi:10.1146/annurev.cellbio.22.010305.104241
Radice GL, Rayburn H, Matsunami H, Knudsen KA, Takeichi M, Hynes RO (1997) Developmental defects in mouse embryos lacking N-cadherin. Dev Biol 181:64–78. doi:10.1006/dbio.1996.8443
Reeber SL, Sakai N, Nakada Y, Dumas J, Dobrenis K, Johnson JE, Kaprielian Z (2008) Manipulating Robo expression in vivo perturbs commissural axon pathfinding in the chick spinal cord. J Neurosci 28:8698–8708. doi:10.1523/JNEUROSCI.1479-08.2008
Riehl R, Johnson K, Bradley R, Grunwald GB, Cornel E, Lilienbaum A, Holt CE (1996) Cadherin function is required for axon outgrowth in retinal ganglion cells in vivo. Neuron 17:837–848
Sakai N, Insolera R, Sillitoe RV, Shi SH, Kaprielian Z (2012) Axon sorting within the spinal cord marginal zone via Robo-mediated inhibition of N-cadherin controls spinocerebellar tract formation. J Neurosci 32:15377–15387. doi:10.1523/JNEUROSCI.2225-12.2012
Sandoval-Minero T, Varela-Echavarría A (2008) Cross-midline interactions between mouse commissural hindbrain axons contribute to their efficient decussation. Dev Neurobiol 68:349–364. doi:10.1002/dneu.20586
Schwabe T, Neuert H, Clandinin TR (2013) A network of cadherin-mediated interactions polarizes growth cones to determine targeting specificity. Cell 154:351–364. doi:10.1016/j.cell.2013.06.011
Shih W, Yamada S (2012) N-cadherin-mediated cell–cell adhesion promotes cell migration in a three-dimensional matrix. J Cell Sci 125:3661–3670. doi:10.1242/jcs.103861
Sotomayor M, Gaudet R, Corey DP (2014) Sorting out a promiscuous superfamily: towards cadherin connectomics. Trends Cell Biol 24:524–536. doi:10.1016/j.tcb.2014.03.007
Stan A, Pielarski KN, Brigadski T, Wittenmayer N, Fedorchenko O, Gohla A, Lessmann V, Dresbach T, Gottmann K (2010) Essential cooperation of N-cadherin and neuroligin-1 in the transsynaptic control of vesicle accumulation. Proc Natl Acad Sci USA 107:11116–11121. doi:10.1073/pnas.0914233107
Tai CY, Mysore SP, Chiu C, Schuman EM (2007) Activity-regulated N-cadherin endocytosis. Neuron 54:771–785. doi:10.1016/j.neuron.2007.05.013
Takeichi M (1995) Morphogenetic roles of classic cadherins. Curr Opin Cell Biol 7:619–627
Tepass U, Truong K, Godt D, Ikura M, Peifer M (2000) Cadherins in embryonic and neural morphogenesis. Nat Rev Mol Cell Biol 1:91–100. doi:10.1038/35040042
Togashi H, Abe K, Mizoguchi A, Takaoka K, Chisaka O, Takeichi M (2002) Cadherin regulates dendritic spine morphogenesis. Neuron 35:77–89
Wang XJ, Zhang DL, Xu ZG, Ma ML, Wang WB, Li LL, Han XL, Huo Y, Yu X, Sun JP (2014) Understanding CELSRs-Cadherin EGF LAG seven-pass G-type receptors. J Neurochem 131:699–711. doi:10.1111/jnc.12955
Wiley A, Edalat K, Chiang P, Mora M, Mirro K, Lee M, Muhr H, Elul T (2008) GSK-3beta and alpha-catenin binding regions of beta-catenin exert opposing effects on the terminal ventral optic axonal projection. Dev Dyn 237:1434–1441. doi:10.1002/dvdy.21549
Yang Z, Wu Y, Zheng L, Zhang C, Yang J, Shi M, Feng D, Wu Z, Wang YZ (2013) Conditioned medium of Wnt/β-catenin signaling-activated olfactory ensheathing cells promotes synaptogenesis and neurite growth in vitro. Cell Mol Neurobiol 33:983–990. doi:10.1007/s10571-013-9966-z
Yang C, Li X, Liu Y, Li H, Guo Z, Lin J (2015) Sonic hedgehog overexpression regulates the neuroepithelial cells proliferation in the spinal cord of dorsal regions during chicken embryo development. Neuro Endocrinol Lett 36:380–386
Zhang J, Woodhead G, Swaminathan S, Noles S, McQuinn E, Pisarek A, Stocker A, Mutch C, Funatsu N, Chenn A (2010) Cortical neural precursors inhibit their own differentiation via N-cadherin maintenance of beta-catenin signaling. Dev Cell 18:472–479. doi:10.1016/j.devcel.2009.12.025
Zhang J, Shemezis JR, McQuinn ER, Wang J, Sverdlov M, Chenn A (2013) AKT activation by N-cadherin regulates beta-catenin signaling and neuronal differentiation during cortical development. Neural Dev 8:7. doi:10.1186/1749-8104-8-7
Acknowledgments
We thank Prof. C. Redies for critical reading and correcting this manuscript.
Author contributions
Conceived and designed the experiments: Zhikun Guo, Shanting Zhao, Juntang Lin. Performed the experiments: Ciqing Yang, Xiaoying Li, Congrui Wang Sulei Fu, Han Li. Analyzed the data: Ciqing Yang.Wrote the paper: Ciqing Yang.
Funding
This work was supported by grants from the National Natural Science Foundation of China (31440049), the Science and Technology Innovation Plan of Henan (14HASTIT032), the Henan Province University youth researcher support program project (2015GGJS-133), the Scientific Research Fund (162300410232, 14B310007) and the PhD Research Startup Foundation (505090) of Xinxiang Medical University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known conflicts of interest associated with this publication.
Rights and permissions
About this article
Cite this article
Yang, C., Li, X., Wang, C. et al. N-cadherin regulates beta-catenin signal and its misexpression perturbs commissural axon projection in the developing chicken spinal cord. J Mol Hist 47, 541–554 (2016). https://doi.org/10.1007/s10735-016-9698-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-016-9698-8