Abstract
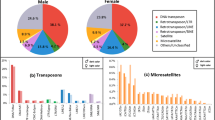
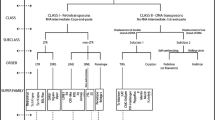
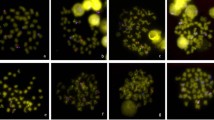
The high dynamism of repetitive DNAs is a major driver of chromosome evolution. In particular, the accumulation of repetitive DNA sequences has been reported as part of the differentiation of sex-specific chromosomes. In turn, the fish species of the genus Megaleporinus are a monophyletic clade in which the presence of differentiated ZZ/ZW sex chromosomes represents a synapomorphic condition, thus serving as a suitable model to evaluate the dynamic evolution of repetitive DNA classes. Therefore, transposable elements (TEs) and in tandem repeats were isolated and located on chromosomes of Megaleporinus obtusidens and M. reinhardti to infer their role in chromosome differentiation with emphasis on sex chromosome systems. Despite the conserved karyotype features of both species, the location of repetitive sequences - Rex 1, Rex 3, (TTAGGG)n, (GATA)n, (GA)n, (CA)n, and (A)n - varied both intra and interspecifically, being mainly accumulated in Z and W chromosomes. The physical mapping of repetitive sequences confirmed the remarkable dynamics of repetitive DNA classes on sex chromosomes that might have promoted chromosome diversification and reproductive isolation in Megaleporinus species.



Similar content being viewed by others
Data availability
No datasets were generated or analysed during the current study.
References
Bachtrog D (2008) The temporal dynamics of processes underlying Y chromosome degeneration. Genetics 179:1513–1525. https://doi.org/10.1534/genetics.107.084012
Bachtrog D, Mank JE, Peichel CL, Kirkpatrick M, Otto SP, Ashman T, Hahn MW, Kitano J, Mayrose I, Ming R, Perrin N, Ross L, Valenzuela N, Vamosi JC (2014) Sex determination: why so many ways of doing it? PLoS Biol 12:e1001899. https://doi.org/10.1371/journal.pbio.1001899
Bertollo LAC, Cioffi MB, Moreira-Filho O (2015) Direct chromosome preparation from freshwater teleost fishes. In: Ozouf-Costaz C, Pisano E, Foresti F, Almeida Toledo LF (eds) Fish Cytogenetic techniques (chondrichthyans and teleosts). CRC, Boca Raton, pp 21–26
Biscotti MA, Olmo E, Heslop-Harrison JSP (2015) Repetitive DNA in eukaryotic genomes. Chromosome Res 23:415–420. https://doi.org/10.1007/s10577-015-9499-z
Bitencourt JA, Sampaio I, Ramos RT, Affonso PRAM (2014) Chromosomal fusion in Brazilian populations of Trinectes inscriptus Gosse, 1851 (Pleuronectiformes; Achiridae) as revealed by internal telomere sequences (ITS). J Exp Mar Biol Ecol 452:101–104. https://doi.org/10.1016/j.jembe.2013.12.012
Carducci F, Barucca M, Canapa A, Biscotti MA (2018) Rex retroelements and teleost genomes: an overview. Int J Mol Sci 19:e3653. https://doi.org/10.3390/ijms19113653
Charlesworth B (1991) The evolution of sex chromosomes. Science 251:1030–1033. https://www.science.org/doi/10.1126/science.1998119
Crepaldi C, Parise-Maltempi PP (2020) Heteromorphic sex chromosomes and their DNA content in Fish: an insight through satellite DNA accumulation in Megaleporinus Elongatus. Cytogenet Genome Res 160:38–46. https://doi.org/10.1159/000506265
Deon GA, Glugoski L, Sassi FMC, Hatanaka T, Nogaroto V, Bertollo LAC, Liehr T, Al-Rikabi A, Moreira Filho O, Cioffi MB, Vicari MR (2022a) Chromosomal rearrangements and origin of the multiple XX/XY1Y2 sex chromosome system in Harttia species (Siluriformes: Loricariidae). Front Genet 13:e877522. https://doi.org/10.3389/fgene.2022.877522
Deon GA, Glugoski L, Hatanaka T, Sassi FMC, Nogaroto V, Bertollo LAC, Liehr T, Al-Rikabi A, Moreira Filho O, Cioffi MB, Vicari MR (2022b) Evolutionary breakpoint regions and chromosomal remodeling in Harttia (Siluriformes: Loricariidae) species diversification. Genet Mol Biol 45(2):e20210170. https://doi.org/10.1590/1678-4685-GMB-2021-0170
Dulz TA, Azambuja M, Nascimento VD, Lorscheider CA, Noleto RB, Moreira-Filho O, Nogaroto V, Diniz D, Affonso PRAM, Vicari MR (2020) Karyotypic diversification in two Megaleporinus species (Characiformes, Anostomidae) inferred from in situ localization of repetitive DNA sequences. Zebrafish 17:333–341. https://doi.org/10.1089/zeb.2020.1918
Ferretti AB, Milani D, Palacios-Gimenez OM, Ruiz-Ruano FJ, Cabral-de-Mello DC (2020) High dynamism for neo-sex chromosomes: satellite DNAs reveal complex evolution in a grasshopper. Heredity 125:124–137. https://doi.org/10.1038/s41437-020-0327-7
Galetti PM Jr, Foresti F, Bertollo LAC, Moreira-Filho O (1981) Heteromorphic sex chromosomes in three species of the genus Leporinus (Pisces, Anostomidae). Cytogenet Cell Genet 29:138–142. https://doi.org/10.1159/000131562
Galetti PM Jr, Lima NRW, Venere PC (1995) A monophyletic ZW sex chromosome system in Leporinus (Anostomidae, Characiformes). Cytologia 60:375–382. https://doi.org/10.1508/cytologia.60.375
Glugoski L, Nogaroto V, Deon GA, Azambuja M, Moreira-Filho O, Vicari MR (2022) Enriched tandemly repeats in chromosomal fusion points of Rineloricaria latirostris (Boulenger, 1900) (Siluriformes: Loricariidae). Genome 65:479–489. https://doi.org/10.1139/gen-2022-0043
Glugoski L, Deon GA, Nogaroto V, Moreira-Filho O, Vicari MR (2023) Robertsonian fusion site in Rineloricaria pentamaculata (Siluriformes: Loricariidae): involvement of 5S rDNA and satellite sequences. Cytogenet Genome Res 162:657–663. https://doi.org/10.1159/000530636
Haerter CAG, Blanco DR, Traldi JB, Feldberg E, Margarido VP, Lui RL (2023) Are scattered microsatellites weak chromosomal markers? Guided mapping reveals new insights into Trachelyopterus (Siluriformes: Auchenipteridae) diversity. PLoS ONE 18:e0285388. https://doi.org/10.1371/journal.pone.0285388
Ijdo JW, Wells RA, Baldini A, Reeders ST (1991) Improved telomere detection using a telomere repeat probe (TTAGGG)n generated by PCR. Nucleic Acids Res 19:4780. https://doi.org/10.1093/nar/19.17.4780
Kearse M, Moir R, Wilson A, Stones-Havas S, Cheung M, Sturrock S, Buxton S, Cooper A, Markowitz S, Duran C, Thierer T, Ashton B, Meintjes P, Drummond A (2012) Geneious Basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 28:1647–1649. https://doi.org/10.1093/bioinformatics/bts199
Kidwell MG (2002) Transposable elements and the evolution of genome size in eukaryotes. Genetica 115:49–63. https://doi.org/10.1023/A:1016072014259
Kohany O, Gentles AJ, Hankus L, Jurka J (2006) Annotation, submission and screening of repetitive elements in repbase: RepbaseSubmitter and Censor. BMC Bioinformatics 7:474. https://doi.org/10.1186/1471-2105-7-474
Kratochvíl L, Stöck M, Rovatsos M, Bullejos M, Herpin A, Jeffries DL, Peichel CL, Perrin N, Valenzuela N, Pokorná MJ (2021) Expanding the classical paradigm: what we have learnt from vertebrates about sex chromosome evolution. Philos Trans R Soc Lond B Biol Sci 376:e20200097. https://doi.org/10.1098/rstb.2020.0097
Levan A, Fredga K, Sandberg AA (1964) Nomenclature for centromeric position on chromosomes. Hereditas 52:201–220. https://doi.org/10.1111/j.1601-5223.1964.tb01953.x
López-Flores I, Garrido-Ramos MA (2012) The repetitive DNA content of eukaryotic genomes. Genome Dyn 7:1–28. https://doi.org/10.1159/000337118
Lui RL, Blanco DR, Martinez JF, Margarido VP, Venere PC, Moreira-Filho O (2013) The role of chromosomal fusion in the karyotypic evolution of the genus Ageneiosus (Siluriformes: Auchenipteridae). Neotrop Ichthyol 11:327–334. https://doi.org/10.1590/S1679-62252013005000004
Lui RL, Blanco DR, Traldi JB, Margarido VP, Moreira-Filho O (2015) Karyotypic variation of Glanidium ribeiroi Haseman, 1911 (Siluriformes, Auchenipteridae) along the Iguazu river basin. Braz J Biol 75:S215–S221. https://doi.org/10.1590/1519-6984.10714
Malimpensa GC, Traldi JB, Toyama D, Henrique-Silva F, Vicari MR, Moreira-Filho O (2018) Chromosomal mapping of repeat DNA in Bergiaria westermanni (Pimelodidae, Siluriformes): localization of 45S rDNA in B chromosomes. Cytogenet Genome Res 154:99–106. https://doi.org/10.1159/000487652
Marreta ME, Faldoni FLC, Parise-Maltempi PP (2012) Cytogenetic mapping of the W chromosome in the genus Leporinus (Teleostei, Anostomidae) using a highly repetitive DNA sequence. J Fish Biol 80:630–637. https://doi.org/10.1111/j.1095-8649.2011.03199.x
Meyne J, Baker RJ, Hobart HH, Hsu TC, Ryder OA, Ward OG, Wiley JE, Wurster-Hill DH, Yates TL, Moyzis RK (1990) Distribution of non-telomeric sites of the (TTAGGG)n telomeric sequence in vertebrate chromosomes. Chromosoma 99:3–10. https://doi.org/10.1007/BF01737283
Molina WF, Schmid M, Galetti PM Jr (1998) Heterochromatin and sex chromosomes in the neotropical fish genus Leporinus. Anostomidae) Cytobios 94:141–149. https://doi.org/10.1590/S1415-47572007000400010. Characiformes
Nakayama I, Foresti F, Tewari R, Schartl M, Chourrout D (1994) Sex chromosome polymorphism and heterogametic males revealed by two cloned DNA probes in the ZW/ZZ fish Leporinus elongatus. Chromosoma 103:31–39. https://doi.org/10.1007/BF00364723
Oliveira MLM, Paim FG, Freitas ÉAS, Oliveira C, Foresti F (2021) Cytomolecular investigations using repetitive DNA probes contribute to the identification and characterization of Characidium sp. aff. C. Vidali (Teleostei: Characiformes). Neotrop Ichthyol 19:e200045. https://doi.org/10.1590/1982-0224-2020-0045
Ozouf-Costaz C, Brandt J, Körting C, Pisano E, Bonillo C, Coutanceau J, Volff J (2004) Genome dynamics and chromosomal localization of the non-LTR retrotransposons Rex1 and Rex3 in Antarctic fish. Antarct Sci 16:51–57. https://doi.org/10.1017/S0954102004001816
Paço A, Adega F, Meštrović N, Plohl M, Chaves R (2015) The puzzling character of repetitive DNA in Phodopus genomes (Cricetidae, Rodentia). Chromosome Res 23:427–440. https://doi.org/10.1007/s10577-015-9481-9
Parise-Maltempi PP, Martins C, Oliveira C, Foresti F (2007) Identification of a new repetitive element in the sex chromosomes of Leporinus elongatus (Teleostei: Characiformes: Anostomidae): new insights into the sex chromosomes of Leporinus. Cytogenet Genome Res 116:218–223. https://doi.org/10.1159/000098190
Parise-Maltempi PP, Silva EL, Rens W, Dearden F, O’Brien PCM, Trifonov V, Ferguson-Smith MA (2013) Comparative analysis of sex chromosomes in Leporinus species (Teleostei, Characiformes) using chromosome painting. BMC Genet 14:60. https://doi.org/10.1186/1471-2156-14-60
Pinkel D, Straume T, Gray JW (1986) Cytogenetic analysis using quantitative, high-sensitivity, fluorescence hybridization. Proc Natl Acad Sci U S A 83:2934–2938. https://doi.org/10.1073/pnas.83.9.2934
Poltronieri J, Marquioni V, Bertollo LAC, Kejnovsky E, Molina WF, Liehr T, Cioffi MB (2014) Comparative chromosomal mapping of microsatellites in Leporinus species (Characiformes, Anostomidae): unequal accumulation on the W chromosomes. Cytogenet Genome Res 142:40–45. https://doi.org/10.1159/000355908
Pucci MB, Barbosa P, Nogaroto V, Almeida MC, Artoni RF, Scacchetti PC, Pansonato-Alves J, Foresti F, Moreira-Filho O, Vicari MR (2016) Chromosomal spreading of microsatellites and (TTAGGG) n sequences in the Characidium zebra and C. gomesi genomes (Characiformes: Crenuchidae). Cytogenet Genome Res 149:182–190. https://doi.org/10.1159/000447959
Ramirez JL, Birindelli JLO, Galetti PM Jr (2017a) A new genus of Anostomidae (Ostariophysi: Characiformes): diversity, phylogeny and biogeography based on cytogenetic, molecular and morphological data. Mol Phylogenet Evol 107:308–323. https://doi.org/10.1016/j.ympev.2016.11.012
Ramirez JL, Birindelli JL, Carvalho DC, Affonso PRAM, Venere PC, Ortega H, Carrillo-Avila M, Rodríguez-Pulido JA, Galetti PM Jr (2017b) Revealing hidden diversity of the underestimated neotropical ichthyofauna: DNA barcoding in the recently described Genus Megaleporinus (Characiformes: Anostomidae). Front Genet 8:149. https://doi.org/10.3389/fgene.2017.00149
Santos EO, Deon GA, Almeida RB, Oliveira EA, Nogaroto V, Silva HP, Pavanelli CS, Cestari MM, Bertollo LAC, Moreira-Filho O, Vicari MR (2019) Cytogenetics and DNA barcode reveal an undescribed Apareiodon species (Characiformes: Parodontidae). Genet Mol Biol 42:365–373. https://doi.org/10.1590/1678-4685-GMB-2018-0066
Scacchetti PC, Utsunomia R, Pansonato-Alves JC, da Costa Silva GJ, Vicari MR, Artoni RF, Oliveira C, Foresti F (2015) Repetitive DNA sequences and evolution of ZZ/ZW sex chromosomes in Characidium (Teleostei: Characiformes). PLoS ONE 10:e0137231. https://doi.org/10.1371/journal.pone.0137231
Schmid M, Steinlein C (2016) Chromosome banding in Amphibia. XXXIV. Intrachromosomal telomeric DNA sequences in Anura. Cytogenet Genome Res 148:211–226. https://doi.org/10.1159/000446298
Sember A, Nguyen P, Perez MF, Altmanová M, Ráb P, Cioffi MB (2021) Multiple sex chromosomes in teleost fishes from a cytogenetic perspective: state of the art and future challenges. Philos Trans R Soc Lond B Biol Sci 376:e20200098. https://doi.org/10.1098/rstb.2020.0098
Splendore de Borba R, Silva EL, Parise-Maltempi PP (2013) Chromosome mapping of retrotransposable elements Rex1 and Rex3 in Leporinus Spix, 1829 species (Characiformes: Anostomidae) and its relationship among heterochromatic segments and w sex chromosome. Mob Genet Elem 3:e27460. https://doi.org/10.4161/mge.27460
Traldi JB, Blanco DR, Vicari MR, Martinez JF, Lui RL, Artoni RF, Moreira-Filho O (2013) Physical mapping of (GATA)n and (TTAGGG)n sequences in species of Hypostomus (Siluriformes, Loricariidae). J Genet 92:127–130. https://doi.org/10.1007/s12041-013-0224-4
Traldi JB, Vicari MR, Martinez JF, Blanco DR, Lui RL, Azambuja M, Almeida RB, Malimpensa GC, Costa Silva GJ, Oliveira C, Pavanelli CS, Moreira-Filho O (2020) Recent Apareiodon species evolutionary divergence (Characiformes: Parodontidae) evidenced by chromosomal and molecular inference. Zool Anz 289:166–176. https://doi.org/10.1016/j.jcz.2020.10.010
Utsunomia R, Silva DMZA, Ruiz-Ruano FJ, Goes CAG, Melo S, Ramos LP, Oliveira C, Porto-Foresti F, Foresti F, Hashimoto DT (2019) Satellitome landscape analysis of Megaleporinus macrocephalus (Teleostei, Anostomidae) reveals intense accumulation of satellite sequences on the heteromorphic sex chromosome. Sci Rep 9:5856. https://doi.org/10.1038/s41598-019-42383-8
Venancio Neto S, Noleto RB, Azambuja M, Gazolla CB, Santos BR, Nogaroto V, Vicari MR (2023) Comparative cytogenetics among Boana species (Anura, Hylidae): focus on evolutionary variability of repetitive DNA. Genet Mol Biol 45:e20220203. https://doi.org/10.1590/1678-4685-GMB-2022-0203
Venere PC, Ferreira IA, Martins C, Galetti PM Jr (2004) A novel ZZ/ZW sex chromosomes system for the genus Leporinus (Pisces, Anostomidae, Characiformes). Genetica 121:75–80. https://doi.org/10.1023/B:GENE.0000019936.40970.7a
Vicari MR, Bruschi DP, Cabral-de-Mello DC, Nogaroto V (2022) Telomere organization and the interstitial telomeric sites involvement in insects and vertebrates chromosome evolution. Genet Mol Biol 45:e20220071. https://doi.org/10.1590/1678-4685-GMB-2022-0071
Volff JN, Körting C, Sweeney K, Schartl M (1999) The non-LTR retrotransposon Rex3 from the fish Xiphophorus is widespread among teleosts. Mol Biol Evol 16:1427–1438. https://doi.org/10.1093/oxfordjournals.molbev.a026055
Volff JN, Körting C, Schartl M (2000) Multiple lineages of the non-LTR retrotransposon Rex1 with varying success in invading fish genomes. Mol Biol Evol 17:1673–1684. https://doi.org/10.1093/oxfordjournals.molbev.a026266
Yano CF, Poltronieri J, Bertollo LAC, Artoni RF, Liehr T, Cioffi MB (2014) Chromosomal mapping of repetitive DNAs in Triportheus trifurcatus (Characidae, Characiformes): insights into the differentiation of the Z and W chromosomes. PLoS ONE 9:e90946. https://doi.org/10.1371/journal.pone.0100494
Ziemniczak K, Traldi JB, Nogaroto V, Almeida MC, Artoni RF, Moreira-Filho O, Vicari MR (2014) In situ localization of (GATA)n and (TTAGGG)n repeated DNAs and w sex chromosome differentiation in Parodontidae (Actinopterygii: Characiformes). Cytogenet Genome Res 144:325–332. https://doi.org/10.1159/000370297
Acknowledgements
The authors are grateful to ICMBio (Instituto Chico Mendes de Conservação da Biodiversidade) for authorizing the collection of samples (license numbers 10538-1, 15117-1, 26752-3).
Funding
This study was supported by the Fundação Araucária (Fundação Araucária de Apoio ao Desenvolvimento Científico e Tecnológico do Estado do Paraná), FAPESB (Fundação de Amparo à Pesquisa do Estado da Bahia), CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, Finance Code 001), and CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico – 305142/2019-4).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. TAD, MA, VDN, VN, and DD contributed to the cytogenetics experiments. MA and MRV performed sequence data analysis. TAD, MA, CAL, RBN, PRAMA, and MRV writing of the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors declare that there is no conflict of interests that could be perceived as prejudicial to the impartiality of the reported research.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dulz, T.A., Azambuja, M., Lorscheider, C.A. et al. Repetitive DNAs and chromosome evolution in Megaleporinus obtusidens and M. reinhardti (Characiformes: Anostomidae). Genetica (2024). https://doi.org/10.1007/s10709-024-00206-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10709-024-00206-3