Abstract
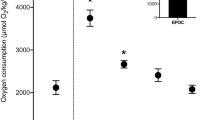
A new “less invasive” device incorporating an ultrasonic flow probe and a divided chamber, but no stitching of membranes to the fish, was employed to make the first direct measurements of ventilatory flow rate (V̇w) and % O2 utilization (%U) in juvenile rainbow trout (37 g, 8ºC) after exhaustive exercise (10-min chasing) and voluntary feeding (2.72% body mass ration). Under resting conditions, the allometrically scaled V̇w (300 ml kg−1 min−1 for a 37-g trout = 147 ml kg−1 min−1 for a 236-g trout exhibiting the same mass-specific O2 consumption rate, ṀO2) and the convection requirement for O2 (CR = 4.13 L mmol−1) were considerably lower, and the %U (67%) was considerably higher than in previous studies using surgically attached masks or the Fick principle. After exhaustive exercise, V̇w and ṀO2 approximately doubled whereas frequency (fr) and %U barely changed, so increased ventilatory stroke volume (Vsv) was the most important contributor to increased ṀO2. CR declined slightly. Values gradually returned to control conditions after 2–3 h. After voluntary feeding, short-term increases in V̇w, Vsv and ṀO2 were comparable to those after exercise, and fr again did not change. However, %U increased so CR declined even more. The initial peaks in V̇w, Vsv and ṀO2, similar to those after exercise, were likely influenced by the excitement and exercise component of voluntary feeding. However, in contrast to post-exercise fish, post-prandial fish exhibited second peaks in these same parameters at 1–3 h after feeding, and %U increased further, surpassing 85%, reflecting the true “specific dynamic action” response. We conclude that respiration in trout is much more efficient than previously believed.








Similar content being viewed by others
Data availability
From the authors, on reasonable request.
Abbreviations
- α O2 :
-
Oxygen solubility coefficient
- ANOVA:
-
Analysis of variance
- BPL:
-
Elastic balloon piece layer
- CR:
-
Convection requirement for oxygen
- EPOC:
-
Excess post-exercise oxygen consumption
- FP:
-
Ultrasonic blood flow probe
- fr:
-
Ventilatory frequency
- ID:
-
Internal diameter
- IU:
-
International units
- ṀO2 :
-
Oxygen consumption rate
- PEO2 :
-
Partial pressure of oxygen in expired water
- PiO2 :
-
Partial pressure of oxygen in inspired water
- PVC:
-
Polyvinyl chloride
- SDA:
-
Specific dynamic action
- Vsv:
-
Ventilatory stroke volume
- V̇w:
-
Ventilatory flow rate of water
- RS:
-
Rubber stopper
- %U:
-
Percentage oxygen utilization from the water
References
Alsop DH, Wood CM (1997) The interactive effects of feeding and exercise on oxygen consumption, swimming performance and protein usage in juvenile rainbow trout. J Exp Biol 200:2337–2346. https://doi.org/10.1242/jeb.200.17.2337
Altimiras J, Larsen E (2000) Non-invasive recording of heart rate and ventilation rate in rainbow trout during rest and swimming. Fish go wireless! J Fish Biol 57:197–209. https://doi.org/10.1111/j.1095-8649.2000.tb00786.x
Boutilier RG, Heming TA, Iwama GK (1984) Physicochemical parameters for use in fish respiratory physiology. In: Hoar WS, Randall DJ (eds) Fish physiology. Vol. 10A. Academic Press, New York, pp 403–430
Brett JR (1964) The respiratory metabolism and swimming performance of young sockeye salmon. J Fisher Res Bd Can 21:1183–1226. https://doi.org/10.1139/f64-103
Brett JR, Zala CA (1975) Daily pattern of nitrogen excretion and oxygen consumption of sockeye salmon (Oncorhynchus nerka) under controlled conditions. J Fisher Res Bd Can 32:2479–2486. https://doi.org/10.1139/f75-285
Bucking C, Wood CM (2006a) Water dynamics in the digestive tract of freshwater rainbow trout during the processing of a single meal. J Exp Biol 209:1883–1893. https://doi.org/10.1242/jeb.02205
Bucking C, Wood CM (2006) Gastrointestinal processing of Na+, Cl-, and K+ during digestion: implications for homeostatic balance in freshwater rainbow trout. Am J Physiol Regul Integr Comp Physiol 291:R1764–R1772. https://doi.org/10.1152/ajpregu.00224.2006
Bucking C, Wood CM (2008) The alkaline tide and ammonia excretion after voluntary feeding in freshwater rainbow trout. J Exp Biol 211:2533–2541. https://doi.org/10.1242/jeb.015610
Bucking C, Wood CM (2012) Digestion of a single meal affects gene expression of ion and ammonia transporters and glutamine synthetase activity in the gastrointestinal tract of freshwater rainbow trout. J Comp Physiol B 181:341–350
Buddington RK, Diamond JM (1986) Aristotle revisited: the function of pyloric caeca in fish. PNAS 83:8012–8014. https://doi.org/10.1073/pnas.83.20.801
Cameron JN, Cech JJ Jr (1970) Notes on the energy cost of gill ventilation in teleosts. Comp Biochem Physiol 34:447–455. https://doi.org/10.1016/0010-406X(70)90184-2
Cameron JN, Davis JC (1970) Gas exchange in rainbow trout (Salmo gairdneri) with varying blood oxygen capacity. J Fisher Res Bd Can 27:1069–1085. https://doi.org/10.1139/F70-123
Chabot D, Koenker R, Farrell AP (2016) The measurement of specific dynamic action in fishes. J Fish Biol 88(152):172. https://doi.org/10.1111/jfb.12836
Clarke A, Johnston NM (1999) Scaling of metabolic rate with body mass and temperature in teleost fish. J Anim Ecol 68:893–905. https://doi.org/10.1046/j.1365-2656.1999.00337.x
Davis JC, Cameron JN (1971) Water flow and gas exchange at the gills of rainbow trout, Salmo gairdneri. J Exp Biol 54:1–18. https://doi.org/10.1242/jeb.54.1.1
Davis JC, Watters K (1970) Evaluation of opercular catheterization as a method for sampling water expired by fish. J Fisher Res Bd Can 27:1627–1635
Dejours P (1988) Respiration in water and air: adaptations-regulations-evolution. Elsevier, Amsterdam
Edwards RRC (1971) An assessment of the energy cost of gill ventilation in the plaice (Pleuronectes platessa L.). Comp Biochem Physiol A 40:391–398. https://doi.org/10.1016/0300-9629(71)90029-6
Eom J, Wood CM (2021) Brain and gills as internal and external ammonia sensing organs for ventilatory control in rainbow trout, Oncorhynchus mykiss. Comp Biochem Physiol A 254:110896. https://doi.org/10.1016/j.cbpa.2021.110896
Eliason EJ, Farrell AP (2014) Effect of hypoxia on specific dynamic action and postprandial cardiovascular physiology in rainbow trout (Oncorhynchus mykiss). Comp Biochem Physiol Part A Mol Integr Physiol 171:44–50. https://doi.org/10.1016/j.cbpa.2014.01.021
Eliason EJ, Higgs DA, Farrell AP (2007) Effect of isoenergetic diets with different protein and lipid content on the growth performance and heat increment of rainbow trout. Aquaculture 272:723–736. https://doi.org/10.1016/j.aquaculture.2007.09.006
Eliason EJ, Higgs DA, Farrell AP (2008) Postprandial gastrointestinal blood flow, oxygen consumption and heart rate in rainbow trout (Oncorhynchus mykiss). Comp Biochem Physiol Part A Mol Integr Physiol 149:380–388. https://doi.org/10.1016/j.cbpa.2008.01.033
Eom J, Fehsenfeld S, Wood CM (2020) Is ammonia excretion affected by gill ventilation in the rainbow trout Oncorhynchus mykiss? Respir Physiol Neurobiol 275:103385. https://doi.org/10.1016/j.resp.2020.103385
Eom J, Wood CM (2020) A less invasive system for the direct measurement of ventilation in fish. Can J Fisher Aquat Sci 77:1870–1877. https://doi.org/10.1139/cjfas-2020-017
Farrell AP, Steffensen JF (1987) An analysis of the energetic cost of the branchial and cardiac pumps during sustained swimming in trout. Fish Physiol Biochem 4:73–79. https://doi.org/10.1007/BF02044316
Glass ML, Andersen NA, Kruhøffer M, Williams EM, Heisler N (1990) Combined effects of environmental PO2 and temperature on ventilation and blood gases in the carp Cyprinus carpio L. J Exp Biol 148:1–17. https://doi.org/10.1242/jeb.148.1.1
Goodrich HR, Berry AA, Montgomery DW, Davison WG, Wilson RW (2022) Fish feeds supplemented with calcium-based buffering minerals decrease stomach acidity, increase the blood alkaline tide and cost more to digest. Sci Rep 12:18468. https://doi.org/10.1038/s41598-022-22496-3
Hall FG (1931) The respiration of puffer fish. Biol Bull 61:457–467
Heath AG (1973) Ventilatory responses of teleost fish to exercise and thermal stress. Am Zool 13:491–503. https://doi.org/10.1093/icb/13.2.491
Hughes GM (1984) Scaling of respiratory areas in relation to oxygen consumption of vertebrates. Experientia 40:519–524. https://doi.org/10.1007/BF01982313
Hughes GM, Saunders RL (1970) Responses of the respiratory pumps to hypoxia in the rainbow trout (Salmo gairdneri). J Exp Biol 53:529–545. https://doi.org/10.1242/jeb.53.3.529
Iwama GK, Boutilier RG, Heming TA, Randall DJ, Mazeaud M (1987) The effects of altering gill water flow on gas transfer in rainbow trout. Can J Zool 65:2466–2470. https://doi.org/10.1139/z87-372
Janssen RG, Randall DJ (1975) The effects of changes in pH and PCO2, in blood and water on breathing in rainbow trout, Salmo gairdneri. Respir Physiol 25:235–245. https://doi.org/10.1016/0034-5687(75)90099-7
Jobling M (1981) The influences of feeding on the metabolic rate of fishes: a short review. J Fish Biol 18:385–400. https://doi.org/10.1111/j.1095-8649.1981.tb03780.x
Jones DR, Brill RW, Butler PJ, Bushnell PG, Heieis RA (1990) Measurement of ventilation volume in swimming tunas. J Exp Biol 149:491–498. https://doi.org/10.1242/jeb.149.1.491
Jones DR, Schwarzfeld T (1974) The oxygen cost to the metabolism and efficiency of breathing in trout (Salmo gairdneri). Respir Physiol 21:241–254. https://doi.org/10.1016/0034-5687(74)90097-8
Karlsson A, Eliason EJ, Mydland LT, Farrell AP, Kiessling A (2006) Postprandial changes in plasma free amino acid levels obtained simultaneously from the hepatic portal vein and the dorsal aorta in rainbow trout (Oncorhynchus mykiss). J Exp Biol 209:4885–4894. https://doi.org/10.1242/jeb.02597
Kiceniuk JW, Jones DR (1977) The oxygen transport system in trout (Salmo gairdneri) during sustained exercise. J Exp Biol 69:247–260. https://doi.org/10.1242/jeb.69.1.247
Kieffer JD (2000) Limits to exhaustive exercise in fish. Comp Biochem Physiol A 126:161–179. https://doi.org/10.1016/S1095-6433(00)00202-6
Kieffer JD (2010) Perspective—exercise in fish: 50+ years and going strong. Comp Biochem Physiol A 156:163–168. https://doi.org/10.1016/j.cbpa.2010.02.009
Kinkead R, Perry SF (1990) An investigation of the role of circulating catecholamines in the control of ventilation during acute moderate hypoxia in rainbow trout (Oncorhynchus mykiss). J Comp Physiol B 160:441–448. https://doi.org/10.1007/BF01075676
Kirsch R, Nonnotte G (1977) Cutaneous respiration in three freshwater teleosts. Respir Physiol 29:339–354. https://doi.org/10.1016/0034-5687(77)90008-1
Leonard EM, Weaver FE, Nurse CA (2022) Lactate sensing by neuroepithelial cells isolated from the gills of killifish (Fundulus heteroclitus). J Exp Biol 225:jeb245088. https://doi.org/10.1242/jeb.245088
Lomholt JP, Johansen K (1979) Hypoxia acclimation in carp. How it affects O2 uptake, ventilation and O2 extraction from water. Physiol Zool 52:38–49. https://doi.org/10.1086/physzool.52.1.30159930
McArley TJ, Morgenroth D, Zena LA, Ekström AT, Sandblom E (2021) Normoxic limitation of maximal oxygen consumption rate, aerobic scope and cardiac performance in exhaustively exercised rainbow trout (Oncorhynchus mykiss). J Exp Biol 224:jeb242614. https://doi.org/10.1242/jeb.242614
McCue MD (2006) Specific dynamic action: a century of investigation. Comp Biochem Physiol A 144:381–394. https://doi.org/10.1016/j.cbpa.2006.03.011
Medland TE, Beamish FWH (1985) The influence of diet and fish density on apparent heat increment in rainbow trout, Salmo gairdneri. Aquaculture 47:1–10. https://doi.org/10.1016/0044-8486(85)90003-1
Millidine KJ, Metcalfe NB, Armstrong JD (2008) The use of ventilation frequency as an accurate indicator of metabolic rate in juvenile Atlantic salmon (Salmo salar). Can J Fisher Aquat Sci 65:2081–2087. https://doi.org/10.1139/F08-118
Milligan CL (1996) Metabolic recovery from exhaustive exercise in rainbow trout. Comp Biochem Physiol Part A Physiol 113:51–60. https://doi.org/10.1016/0300-9629(95)02060-8
Neumann P, Holeton GF, Heisler N (1983) Cardiac output and regional blood flow in gills and muscles after exhaustive exercise in rainbow trout (Salmo gairdneri). J Exp Biol 105:1–14. https://doi.org/10.1242/jeb.105.1.1
Norin T, Malte H (2011) Repeatability of standard metabolic rate, active metabolic rate and aerobic scope in young brown trout during a period of moderate food availability. J Exp Biol 214:1668–1675. https://doi.org/10.1242/jeb.054205
Pan YK, Perry SF (2023) The control of breathing in fishes–historical perspectives and the path ahead. J Exp Biol 226(8):jeb245529. https://doi.org/10.1242/jeb.245529
Perry SF, Wood CM (1989) Control and coordination of gas transfer in fishes. Can J Zool 67:2961–2970. https://doi.org/10.1139/z89-419
Perry SF, Pan YK, Gilmour KM (2023) Insights into the control and consequences of breathing adjustments in fishes-from larvae to adults. Front Physiol 14:66. https://doi.org/10.3389/fphys.2023.1065573
Playle RC, Wood CM (1989) Water chemistry changes in the gill micro-environment of rainbow trout: experimental observations and theory. J Comp Physiol B 159:527–537. https://doi.org/10.1007/BF00694377
Playle RC, Munger RS, Wood CM (1990) Effects of catecholamines on gas exchange and ventilation in rainbow trout (Salmo gairdneri). J Exp Biol 152:353–367. https://doi.org/10.1242/jeb.152.1.353
Porteus CS, Brink DL, Milsom WK (2012) Neurotransmitter profiles in fish gills: putative gill oxygen chemoreceptors. Respir Physiol Neurobiol 184:316–325. https://doi.org/10.1016/j.resp.2012.06.019
Onukwufor JO, Wood CM (2020) Reverse translation effects of acclimation temperature and acute temperature challenges on oxygen consumption, diffusive water flux, net sodium loss rates, Q10 values and mass scaling coefficients in the rainbow trout (Oncorhynchus mykiss). J Comp Physiol B 190:205–217. https://doi.org/10.1007/s00360-020-01259-4
Randall DJ, Cameron JN (1973) Respiratory control of arterial pH as temperature changes in rainbow trout Salmo gairdneri. Am J Physiol 225:997–1002. https://doi.org/10.1152/ajplegacy.1973.225.4.997
Saunders RL (1962) The irrigation of the gills in fishes: II. Efficiency of oxygen uptake in relation to respiratory flow activity and concentrations of oxygen and carbon dioxide. Can J Zool 40:817–862. https://doi.org/10.1139/z62-077
Scarabello M, Heigenhauser GJF, Wood CM (1991) The oxygen debt hypothesis in juvenile rainbow trout after exhaustive exercise. Respir Physiol 84:245–259. https://doi.org/10.1016/0034-5687(91)90121-X
Scarabello M, Heigenhauser GJF, Wood CM (1992) Gas exchange, metabolites and EPOC after repetitive exercise in trout. J Exp Biol 167:155–169. https://doi.org/10.1242/jeb.167.1.155
Schumann D, Piiper J (1966) Der sauerstoffbedarf der atmung bei fischen nach messeungen an der narkotisierten schleie (Tinca tinca). Pflug Arch Eur J Physiol 288:15–26
Seth H, Axelsson M (2010) Sympathetic, parasympathetic and enteric regulation of the gastrointestinal vasculature in rainbow trout (Oncorhynchus mykiss) under normal and postprandial conditions. J Exp Biol 213:3118–3126. https://doi.org/10.1242/jeb.043612
Seth H, Sandblom E, Axelsson M (2009) Nutrient-induced gastrointestinal hyperemia and specific dynamic action in rainbow trout (Oncorhynchus mykiss) - importance of proteins and lipids. Am J Physiol Regul Integrat Comp Physiol 296:R345–R352. https://doi.org/10.1152/ajpregu.90571.2008
Shelton G, Jones DR, Milsom WK (1986) Control of breathing in ectothermic vertebrates. In: Fishman AP, Cherniak NS, Widdicombe JG, Geiger SR (ed) Handbook of Physiology. Sect. 3. The respiratory system. Vol. 2. Control of breathing. American Physiological Society, Bethesda, pp. 857 – 909
Smith FM, Jones DR (1982) The effect of changes in blood oxygen-carrying capacity on ventilation volume in the rainbow trout (Salmo gairdneri). J Exp Biol 97:325–334. https://doi.org/10.1242/jeb.97.1.325
Staples DJ, Nomura M (1976) Influence of body size and food ration on the energy budget of rainbow trout Salmo gairdneri Richardson. J Exp Biol 9:29–43. https://doi.org/10.1016/0305-0491(82)90202-4
Steffensen JF (1985) The transition between branchial pumping and ram ventilation in fishes: energetic consequences and dependence on water oxygen tension. J Exp Biol 114:141–150. https://doi.org/10.1242/jeb.114.1.141
Stevens ED, Randall DJ (1967a) Changes in blood pressure heart rate and breathing rate during moderate swimming activity in rainbow trout. J Exp Biol 46:307–315. https://doi.org/10.1242/jeb.46.2.307
Stevens ED, Randall DJ (1967b) Changes of gas concentrations in blood and water during moderate swimming activity in rainbow trout. J Exp Biol 46:329–337. https://doi.org/10.1242/jeb.46.2.329
Takeda T (1990) Ventilation, cardiac output and blood respiratory parameters in the carp, Cyprinus carpio, during hyperoxia. Respir Physiol 81:227–239. https://doi.org/10.1016/0034-5687(90)90048-4
Thomsen MT, Lefevre S, Nilsson GE, Wang T (2019) Bayley M (2019) Effects of lactate ions on the cardiorespiratory system in rainbow trout (Oncorhynchus mykiss). Am J Physiol Regul Integr Comp Physiol 316:R607–R620. https://doi.org/10.1152/ajpregu.00395.2018
Thorarensen H, Farrell AP (2006) Postprandial intestinal blood flow, metabolic rates, and exercise in Chinook salmon (Oncorhynchus tshawytscha). Physiol Biochem Zool 79:688–694. https://doi.org/10.1086/505512
Turner JD, Wood CM, Clark D (1983) Lactate and proton dynamics in the rainbow trout, Salmo gairdneri. J Exp Biol 104:247–268. https://doi.org/10.1242/jeb.104.1.247
van Dam L (1938) On the utilization of oxygen and regulation of breathing in some aquatic animals. Dissertation, University of Groningen
Vulesevic B, McNeill B, Perry SF (2006) Chemoreceptor plasticity and respiratory acclimation in the zebrafish Danio rerio. J Exp Biol 209:1261– 1273. https://doi.org/10.1242/jeb.02058
Wang Y, Heigenhauser GJ, Wood CM (1994) Integrated responses to exhaustive exercise and recovery in rainbow trout white muscle: acid-base, phosphogen, carbohydrate, lipid, ammonia, fluid volume and electrolyte metabolism. J Exp Biol 195:227–258. https://doi.org/10.1242/jeb.195.1.227
Watters KW, Smith LS (1973) Respiratory dynamics of the starry flounder Platichthys stellatus in response to low oxygen and high temperature. Mar Biol 19:133–148. https://doi.org/10.1007/BF00353584
Wieser W (1985) Developmental and metabolic constraints of the scope for activity in young rainbow trout (Salmo gairdneri). J Exp Biol 118:133–142. https://doi.org/10.1242/jeb.118.1.133
Wilkes PRH, Walker RL, McDonald DG, Wood CM (1981) Respiratory, ventilatory, acid-base and ionoregulatory physiology of the white sucker (Catostomus commersoni): the influence of hyperoxia. J Exp Biol 91:239–254. https://doi.org/10.1242/jeb.91.1.239
Wood CM (1991) Acid-base and ion balance, metabolism, and their interactions, after exhaustive exercise in fish. J Exp Biol 160:285–308. https://doi.org/10.1242/jeb.160.1.285
Wood CM, Bucking C (2010) The role of feeding in salt and water balance. In: Grosell M, Farrell AP, Brauner CJ (ed) The multifunctional gut of fish. Academic Press 30:165–212
Wood CM, Eom J (2021) The osmorespiratory compromise in the fish gill. Comp Biochem Physiol A 254:110895. https://doi.org/10.1016/j.cbpa.2021.110895
Wood CM, Jackson EB (1980) Blood acid-base regulation during environmental hyperoxia in the rainbow trout (Salmo gairdneri). Respir Physiol 42:351–372. https://doi.org/10.1016/0034-5687(80)90125-5
Wood CM, Munger RS (1994) Carbonic anhydrase injection provides evidence for the role of blood acid-base status in stimulating ventilation after exhaustive exercise in rainbow trout. J Exp Biol 194:225–253. https://doi.org/10.1242/jeb.194.1.225
Zhang Y, Claireaux G, Takle H, Jørgensen SM, Farrell AP (2018) A three‐phase excess post‐exercise oxygen consumption in Atlantic salmon Salmo salar and its response to exercise training. J Fish Biol 92:1385–1403. https://doi.org/10.1111/jfb.13593
Zhang Y, Gilbert MJ, Farrell AP (2019) Finding the peak of dynamic oxygen uptake during fatiguing exercise in fish. J Exp Biol 222:jeb196568. https://doi.org/10.1242/jeb.196568
Zhang Y, Gilbert MJ, Farrell AP (2020) Measuring maximum oxygen uptake with an incremental swimming test and by chasing rainbow trout to exhaustion inside a respirometry chamber yields the same results. J Fish Biol 97:28–38. https://doi.org/10.1111/jfb.14311
Zhang L, Nurse CA, Jonz MG, Wood CM (2011) Ammonia sensing by neuroepithelial cells and ventilatory responses to ammonia in rainbow trout. J Exp Biol 214:2678–2689. https://doi.org/10.1242/jeb.055541
Acknowledgements
We thank Drs. Tony Farrell, Bill Milson, and Colin Brauner for the loan of equipment.
Funding
Funded by an NSERC (Canada) Discovery Grant to CMW (RG-PIN 2017–03843).
Author information
Authors and Affiliations
Contributions
JE and CMW designed the study. JE performed the experiments. CMW obtained funding. JE and CMW wrote the paper.
Corresponding author
Ethics declarations
Ethical approval
The methodology of this study was approved by the University of British Columbia Animal Care Committee.
Competing interests
No competing interests were declared.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Eom, J., Wood, C.M. The first direct measurements of ventilatory flow and oxygen utilization after exhaustive exercise and voluntary feeding in a teleost fish, Oncorhynchus mykiss. Fish Physiol Biochem 49, 1129–1149 (2023). https://doi.org/10.1007/s10695-023-01247-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-023-01247-9