Abstract
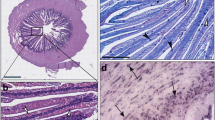
The study aimed to investigate the effects of dietary sodium butyrate (NaBT) supplementation on the gut health of largemouth bass (Micropterus salmoides) fed with a high soybean meal diet. Three isonitrogenous and isolipidic diets were formulated: a high fishmeal group (Control); a high soybean meal group (SBM), in which the 30% fishmeal protein in the Control diet was replaced by soy protein; and an NaBT group, in which 0.2% NaBT was added to the SBM diet. Each diet was fed to triplicate tanks (20 fish in each tank). After 8 weeks of feeding trial, the distal intestine and intestinal digesta of the fish in each treatment were sampled. The results showed that fishmeal replacement and NaBT supplementation did not affect fish growth performance. Dietary 0.2% NaBT supplementation improved intestinal morphology, increasing the villus width and villus height and reducing the width of lamina propria. The distal intestine of fish in the control and NaBT groups demonstrated lower activities of total superoxide dismutase (T-SOD) and glutathione peroxidase (GPx) and a lower malondialdehyde (MDA) content, compared with the fish in the SBM group. Moreover, the addition of 0.2% NaBT in the feed significantly decreased the expression of tumor necrosis factor α (TNF-α) and interleukin 1β (IL-1β) compared to the SBM diet. PCoA and UPGMA analyses based on weighted UniFrac distances demonstrated that intestinal microbial communities in the NaBT group were closer to those in the control group than to those in the SBM group. In addition, dietary 0.2% NaBT supplementation significantly increased the abundance of Firmicutes and Bacteroidetes and decreased the abundance of Tenericutes at the phylum level. Furthermore, the abundance of Bacteroides, Lachnospiraceae_unclassified, and Lachnospiraceae_uncultured was significantly increased, while that of Mycoplasma was significantly decreased in fish intestine at NaBT group at the genus level. In conclusion, dietary NaBT supplementation had beneficial roles in protecting the gut health of largemouth bass from the impairments caused by soybean meal.






Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Code availability
Not applicable.
References
AOAC (1995) Official methods of analysis, 16th edn. Association of official Analytical Chemests, Arlington, VA
Baeverfjord G, Krogdahl A (1996) Development and regression of soybean meal induced enteritis in Atlantic salmon, Salmo salar L., distal intestine: a comparison with the intestines of fasted fish. J Fish Dis 19:375–387. https://doi.org/10.1046/j.1365-2761.1996.d01-92.x
Bedford A, Gong J (2018) Implications of butyrate and its derivatives for gut health and animal production. Anim Nutr 4:151–159. https://doi.org/10.1016/j.aninu.2017.08.010
Biagi G, Piva A, Moschini M, Vezzali E, Roth FX (2007) Performance, intestinal microflora, and wall morphology of weanling pigs fed sodium butyrate. J Anim Sci 85:1184–1191. https://doi.org/10.2527/jas.2006-378
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Butt RL, Volkoff H (2019) Gut microbiota and energy homeostasis in fish. Front Endocrinol 10:12. https://doi.org/10.3389/fendo.2019.00009
Chen G, Ran X, Li B, Li Y, He D, Huang B, Fu S, Liu J, Wang W (2018) Sodium butyrate inhibits inflammation and maintains epithelium barrier integrity in a TNBS-induced inflammatory bowel disease mice model. EBioMedicine 30:317–325. https://doi.org/10.1016/j.ebiom.2018.03.030
Chen W, Li D, Paulus B, Wilson I, Chadwick VS (2001) High prevalence of mycoplasma pneumoniae in intestinal mucosal biopsies from patients with inflammatory bowel disease and controls. Dig Dis Sci 46:2529–2535. https://doi.org/10.1023/A:1012352626117
China Fishery Stastical Yearbook (2019) China fishery statistical yearbook. China Agriculture Press, Beijing, China
Chomczynski P, Sacchi N (2006) The single-step method of RNA isolation by acid guanidinium thiocyanate–phenol–chloroform extraction: twenty-something years on. Nat Protoc 1:581. https://doi.org/10.1038/nprot.2006.83
Czerwiński J, Højberg O, Smulikowska S, Engberg RM, Mieczkowska A (2012) Effects of sodium butyrate and salinomycin upon intestinal microbiota, mucosal morphology and performance of broiler chickens. Arch Anim Nutr 66:102–116. https://doi.org/10.1080/1745039X.2012.663668
Del Rio D, Stewart AJ, Pellegrini N (2005) A review of recent studies on malondialdehyde as toxic molecule and biological marker of oxidative stress. Nutr Metab Cardiovasc Dis 15:316–328. https://doi.org/10.1016/j.numecd.2005.05.003
Esterbauer H, Cheeseman KH (1990) Determination of aldehydic lipid peroxidation products: malonaldehyde and 4-hydroxynonenal. Methods Enzymol 186:407–421. https://doi.org/10.1016/0076-6879(90)86134-H
Fachi JL, Felipe JS, Pral LP, da Silva BK, Corrêa RO, de Andrade MCP, da Fonseca DM, Basso PJ, Câmara NOS, de Sales ESÉ, Dos Santos MF, Guima SES, Thomas AM, Setubal JC, Magalhães YT, Forti FL, Candreva T, Rodrigues HG, de Jesus MB, Consonni SR, Farias ADS, Varga-Weisz P, Vinolo MAR (2019) Butyrate protects mice from clostridium difficile-induced colitis through an HIF-1-dependent mechanism. Cell Rep 27:750-761.e757. https://doi.org/10.1016/j.celrep.2019.03.054
Fang W, Xue H, Chen X, Chen K, Ling W (2019) Supplementation with sodium butyrate modulates the composition of the gut microbiota and ameliorates high-fat diet-induced obesity in mice. J Nutr 149:747–754. https://doi.org/10.1093/jn/nxy324
Francis G, Makkar HPS, Becker K (2001) Antinutritional factors present in plant-derived alternate fish feed ingredients and their effects in fish. Aquaculture 199:197–227. https://doi.org/10.1016/s0044-8486(01)00526-9
Gallo BD, Farrell JM, Leydet BF (2020) Fish gut microbiome: a primer to an emerging discipline in the fisheries sciences. Fisheries 45:271–282. https://doi.org/10.1002/fsh.10379
Gao Y, Storebakken T, Shearer KD, Penn M, Øverland M (2011) Supplementation of fishmeal and plant protein-based diets for rainbow trout with a mixture of sodium formate and butyrate. Aquaculture 311:233–240. https://doi.org/10.1016/j.aquaculture.2010.11.048
Gnanadurai R, Fifer H (2020) Mycoplasma genitalium: a review. Microbiology 166:21–29. https://doi.org/10.1099/mic.0.000830
Hamer HM, Jonkers D, Venema K, Vanhoutvin S, Troost FJ, Brummer RJ (2008) Review article: the role of butyrate on colonic function. Aliment Pharmacol Ther 27:104–119. https://doi.org/10.1111/j.1365-2036.2007.03562.x
Han D, Shan X, Zhang W, Chen Y, Wang Q, Li Z, Zhang G, Xu P, Li J, Xie S, Mai K, Tang Q, De Silva SS (2018) A revisit to fishmeal usage and associated consequences in Chinese aquaculture. Rev Aquac 10:493–507. https://doi.org/10.1111/raq.12183
Hardy RW (2010) Utilization of plant proteins in fish diets: effects of global demand and supplies of fishmeal. Aquacult Res 41:770–776. https://doi.org/10.1111/j.1365-2109.2009.02349.x
He M, Li X, Poolsawat L, Guo Z, Yao W, Zhang C, Leng X (2020) Effects of fish meal replaced by fermented soybean meal on growth performance, intestinal histology and microbiota of largemouth bass (Micropterus salmoides). Aquac Nutr 26:1058–1071. https://doi.org/10.1111/anu.13064
Hedrera MI, Galdames JA, Jimenez-Reyes MF, Reyes AE, Avendaño-Herrera R, Romero J, Feijóo CG (2013) Soybean meal induces intestinal inflammation in zebrafish larvae. PLoS ONE 8:e69983. https://doi.org/10.1371/journal.pone.0069983
İnal ME, Kanbak G, Sunal E (2001) Antioxidant enzyme activities and malondialdehyde levels related to aging. Clin Chim Acta 305:75–80. https://doi.org/10.1016/S0009-8981(00)00422-8
Janero DR (1990) Malondialdehyde and thiobarbituric acid-reactivity as diagnostic indices of lipid peroxidation and peroxidative tissue injury. Free Radic Biol Med 9:515–540. https://doi.org/10.1016/0891-5849(90)90131-2
Kaur A, Chen T, Green SJ, Mutlu E, Martin BR, Rumpagaporn P, Patterson JA, Keshavarzian A, Hamaker BR (2019) Physical inaccessibility of a resistant starch shifts mouse gut microbiota to butyrogenic Firmicutes. Mol Nutr Food Res 63:e1801012. https://doi.org/10.1002/mnfr.201801012
Kelly Caleb J, Zheng L, Campbell Eric L, Saeedi B, Scholz Carsten C, Bayless Amanda J, Wilson Kelly E, Glover Louise E, Kominsky Douglas J, Magnuson A, Weir Tiffany L, Ehrentraut Stefan F, Pickel C, Kuhn Kristine A, Lanis Jordi M, Nguyen V, Taylor Cormac T, Colgan Sean P (2015) Crosstalk between microbiota-derived short-chain fatty acids and intestinal epithelial hif augments tissue barrier function. Cell Host Microbe 17:662–671. https://doi.org/10.1016/j.chom.2015.03.005
Kokou F, Sarropoulou E, Cotou E, Rigos G, Henry M, Alexis M, Kentouri M (2015) Effects of fish meal replacement by a soybean protein on growth, histology, selected immune and oxidative status markers of gilthead sea bream, Sparus aurata. J World Aquacult Soc 46:115–128. https://doi.org/10.1111/jwas.12181
Krogdahl BM, RØed, Baeverfjord, (2000) Feeding Atlantic salmon Salmo salar L. soybean products: effects on disease resistance (furunculosis), and lysozyme and IgM levels in the intestinal mucosa. Aquac Nutr 6:77–84. https://doi.org/10.1046/j.1365-2095.2000.00129.x
Krogdahl Å, Bakke-McKellep AM, Baeverfjord G (2003) Effects of graded levels of standard soybean meal on intestinal structure, mucosal enzyme activities, and pancreatic response in Atlantic salmon (Salmo salar L.). Aquac Nutr 9:361–371. https://doi.org/10.1046/j.1365-2095.2003.00264.x
Lee S-M, Mohammadi Azarm H, Chang KH (2016) Effects of dietary inclusion of fermented soybean meal on growth, body composition, antioxidant enzyme activity and disease resistance of rockfish (Sebastes schlegelii). Aquaculture 459:110–116. https://doi.org/10.1016/j.aquaculture.2016.03.036
Leonel AJ, Alvarez-Leite JI (2012) Butyrate: implications for intestinal function. Curr Opin Clin Nutr Metab Care 15:474–479. https://doi.org/10.1097/MCO.0b013e32835665fa
Li XM, Yan QY, Xie SQ, Hu W, Yu YH, Hu ZH (2013) Gut microbiota contributes to the growth of fast-growing transgenic common carp (Cyprinus carpio L.). PLoS One 8:11. https://doi.org/10.1371/journal.pone.0064577
Liu W, Yang Y, Zhang J, Gatlin DM, Ringø E, Zhou Z (2014) Effects of dietary microencapsulated sodium butyrate on growth, intestinal mucosal morphology, immune response and adhesive bacteria in juvenile common carp (Cyprinus carpio) pre-fed with or without oxidised oil. Br J Nutr 112:15–29. https://doi.org/10.1017/S0007114514000610
Liu Y, Chen Z, Dai J, Yang P, Xu W, Ai Q, Zhang W, Zhang Y, Zhang Y, Mai K (2019) Sodium butyrate supplementation in high-soybean meal diets for turbot (Scophthalmus maximus L.): effects on inflammatory status, mucosal barriers and microbiota in the intestine. Fish Shellfish Immunol 88:65–75. https://doi.org/10.1016/j.fsi.2019.02.064
Macfarlane GT, Macfarlane S (2012) Bacteria, colonic fermentation, and gastrointestinal health. J AOAC Int 95:50–60. https://doi.org/10.5740/jaoacint.SGE_Macfarlane
Macfarlane S, Steed H, Macfarlane GT (2009) Intestinal bacteria and inflammatory bowel disease. Crit Rev Clin Lab Sci 46:25–54. https://doi.org/10.1080/10408360802485792
Merrifield D, Olsen R, Myklebust R, Ringø E (2011) Dietary effect of soybean (Glycine max) products on gut histology and microbiota of fish. In: El-Shemy H (ed) Soybean and Nutrition. InTech, Rijeka, Croatia, pp 231–250. https://doi.org/10.5772/20101
Nadal AL, Ikeda-Ohtsubo W, Sipkema D, Peggs D, McGurk C, Forlenza M, Wiegertjes GF, Brugman S (2020) Feed, microbiota, and gut immunity: using the zebrafish model to understand fish health. Front Immunol 11:15. https://doi.org/10.3389/fimmu.2020.00114
Ng W-K, Koh C-B (2017) The utilization and mode of action of organic acids in the feeds of cultured aquatic animals. Rev Aquac 9:342–368. https://doi.org/10.1111/raq.12141
Olsen RL, Hasan MR (2012) A limited supply of fishmeal: impact on future increases in global aquaculture production. Trends Food Sci Technol 27:120–128. https://doi.org/10.1016/j.tifs.2012.06.003
Owen MAG, Waines P, Bradley G, Davies SJ (2006) The effect of dietary supplementation of sodium butyrate on the growth and microflora of Clarias gariepinus (Burchell 1822). In: XII International Symposium on Fish Nutrition and Feeding, May 28–June 1. Biarritz, France, p 149 [Abstract]
Paglia DE, Valentine WN (1967) Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med 70:158–169
Pervin MA, Jahan H, Akter R, Omri A, Hossain Z (2020) Appraisal of different levels of soybean meal in diets on growth, digestive enzyme activity, antioxidation, and gut histology of tilapia (Oreochromis niloticus). Fish Physiol Biochem 46:1397–1407. https://doi.org/10.1007/s10695-020-00798-5
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT–PCR. Nucleic Acids Res 29:e45. https://doi.org/10.1093/nar/29.9.e45
Razin S, Yogev D, Naot Y (1998) Molecular biology and pathogenicity of mycoplasmas. Microbiol Mol Biol Rev 62:1094–1156. https://doi.org/10.1128/mmbr.62.4.1094-1156.1998
Roediger WE (1980) Role of anaerobic bacteria in the metabolic welfare of the colonic mucosa in man. Gut 21:793–798. https://doi.org/10.1136/gut.21.9.793
Shah YM (2016) The role of hypoxia in intestinal inflammation. Mol Cellular Pediatr 3:1. https://doi.org/10.1186/s40348-016-0030-1
Shao T, Zhao C, Li F, Gu Z, Liu L, Zhang L, Wang Y, He L, Liu Y, Liu Q, Chen Y, Donde H, Wang R, Jala VR, Barve S, Chen S-Y, Zhang X, Chen Y, McClain CJ, Feng W (2018) Intestinal HIF-1α deletion exacerbates alcoholic liver disease by inducing intestinal dysbiosis and barrier dysfunction. J Hepatol 69:886–895. https://doi.org/10.1016/j.jhep.2018.05.021
Tacon AG, Metian MR, Tacon MAG, Hasan MR, Metian M (2011) Demand and supply of feed ingredients for farmed fish and crustaceans: trends and prospects. Food and Agriculture Organization of the United Nations (FAO), Rome
Tan C, Zhou H, Wang X, Mai K, He G (2019) Resveratrol attenuates oxidative stress and inflammatory response in turbot fed with soybean meal based diet. Fish Shellfish Immunol 91:130–135. https://doi.org/10.1016/j.fsi.2019.05.030
Thomas F, Hehemann J-H, Rebuffet E, Czjzek M, Michel G (2011) Environmental and gut Bacteroidetes: the food connection. Front Microbiol 2:93. https://doi.org/10.3389/fmicb.2011.00093
Tian L, Zhou X-Q, Jiang W-D, Liu Y, Wu P, Jiang J, Kuang S-Y, Tang L, Tang W-N, Zhang Y-A, Xie F, Feng L (2017) Sodium butyrate improved intestinal immune function associated with NF-κB and p38MAPK signalling pathways in young grass carp (Ctenopharyngodon idella). Fish Shellfish Immunol 66:548–563. https://doi.org/10.1016/j.fsi.2017.05.049
Tidwell JH, Coyle SD, Woods TA (2000) Species profile largemouth bass. Southern Regional Aquaculture Center, Stoneville, Mississippi
Tran NT, Li Z, Wang S, Zheng H, Aweya JJ, Wen X, Li S (2018) Progress and perspectives of short-chain fatty acids in aquaculture. Rev Aquac 12:283–298. https://doi.org/10.1111/raq.12317
Urán PA, Gonçalves AA, Taverne-Thiele JJ, Schrama JW, Verreth JAJ, Rombout JHWM (2008) Soybean meal induces intestinal inflammation in common carp (Cyprinus carpio L.). Fish Shellfish Immunol 25:751–760. https://doi.org/10.1016/j.fsi.2008.02.013
van den Ingh TSGAM, Krogdahl Å, Olli JJ, Hendriks HGCJM, Koninkx JGJF (1991) Effects of soybean-containing diets on the proximal and distal intestine in Atlantic salmon (Salmo salar): a morphological study. Aquaculture 94:297–305. https://doi.org/10.1016/0044-8486(91)90174-6
Volatiana JA, Sagada G, Xu B, Zhang J, Ng W-K, Shao Q (2020) Effects of butyrate glycerides supplementation in high soybean meal diet on growth performance, intestinal morphology and antioxidative status of juvenile black sea bream, Acanthopagrus schlegelii. Aquac Nutr 26:15–25. https://doi.org/10.1111/anu.12984
Wang AR, Ran C, Ringø E, Zhou ZG (2018) Progress in fish gastrointestinal microbiota research. Rev Aquac 10:626–640. https://doi.org/10.1111/raq.12191
Wilson AJ, Gibson PR (1997) Short-chain fatty acids promote the migration of colonic epithelial cells in vitro. Gastroenterology 113:487–496. https://doi.org/10.1053/gast.1997.v113.pm9247468
Wu W, Xiao Z, An W, Dong Y, Zhang B (2018) Dietary sodium butyrate improves intestinal development and function by modulating the microbial community in broilers. PLoS ONE 13:e0197762. https://doi.org/10.1371/journal.pone.0197762
Yin J, Zhou C, Yang K, Ren Y, Qiu Y, Xu P, Xiao W, Yang H (2020) Mutual regulation between butyrate and hypoxia-inducible factor-1α in epithelial cell promotes expression of tight junction proteins. Cell Bio Int 44:1405–1414. https://doi.org/10.1002/cbin.11336
Yuan J, Ni M, Liu M, Wang H, Zhang C, Mi G, Gu Z (2019) Analysis of the growth performances, muscle quality, blood biochemistry and antioxidant status of Micropterus salmoides farmed in in-pond raceway systems versus usual-pond systems. Aquaculture 511:734241. https://doi.org/10.1016/j.aquaculture.2019.734241
Zhang C, Rahimnejad S, Wang Y-r, Lu K, Song K, Wang L, Mai K (2018) Substituting fish meal with soybean meal in diets for Japanese seabass (Lateolabrax japonicus): effects on growth, digestive enzymes activity, gut histology, and expression of gut inflammatory and transporter genes. Aquaculture 483:173–182. https://doi.org/10.1016/j.aquaculture.2017.10.029
Zhang J, Song L, Wang Y, Liu C, Zhang L, Zhu S, Liu S, Duan L (2019) Beneficial effect of butyrate-producing Lachnospiraceae on stress-induced visceral hypersensitivity in rats. J Gastroen Hepatol 34:1368–1376. https://doi.org/10.1111/jgh.14536
Zhang J, Zhong L, Chi S, Chu W, Liu Y, Hu Y (2020) Sodium butyrate supplementation in high-soybean meal diets for juvenile rice field eel (Monopterus albus): effects on growth, immune response and intestinal health. Aquaculture 520:734952. https://doi.org/10.1016/j.aquaculture.2020.734952
Zhang JX, Guo LY, Feng L, Jiang WD, Kuang SY, Liu Y, Hu K, Jiang J, Li SH, Tang L, Zhou XQ (2013) Soybean beta-conglycinin induces inflammation and oxidation and causes dysfunction of intestinal digestion and absorption in fish. PLoS ONE 8:12. https://doi.org/10.1371/journal.pone.0058115
Zheng L, Kelly CJ, Colgan SP (2015) Physiologic hypoxia and oxygen homeostasis in the healthy intestine. A review in the theme: cellular responses to hypoxia. Am J Physiol Cell Physiol 309:C350–C360. https://doi.org/10.1152/ajpcell.00191.2015
Acknowledgements
Great thanks were given to Dongying Bai and technician Shitu Tan for their technical assistance in the experiment.
Funding
This work was supported by the Doctoral Scientific Research Foundation of Henan University of Science and Technology (13,480,088), the National College Students Innovation and Entrepreneurship Training Program (202,110,464,058), and the Key Research and Development and Promotion of Special (Science and Technology) Project of Henan Province (212,102,110,368).
Author information
Authors and Affiliations
Contributions
Kuo Chang carried out the experiment, wrote the manuscript, and contributed to the biochemical analysis. Jialong Chen and Xiaoyu Zhao assisted in the biochemical analysis. Shiyang Gao designed the experiment and revised the manuscript. Weijun Chen contributed to the study conception and design, revised the manuscript, and acquired the funding.
Corresponding author
Ethics declarations
Ethics approval
The experimental protocol and procedures were approved by the Institutional Animal Care and Use Committee, Henan University of Science and Technology.
Consent to participate
Not applicable.
Consent for publication
All authors review and approve the manuscript for publication.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's nte
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chen, W., Chang, K., Chen, J. et al. Dietary sodium butyrate supplementation attenuates intestinal inflammatory response and improves gut microbiota composition in largemouth bass (Micropterus salmoides) fed with a high soybean meal diet. Fish Physiol Biochem 47, 1805–1819 (2021). https://doi.org/10.1007/s10695-021-01004-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-021-01004-w