Abstract
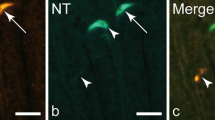
Endocrine cells (ECs) act as a luminal surveillance system responding to either the presence or absence of food in the gut through the secretion of peptide hormones. The aim of this study was to analyze the effects of feeding and fasting on the EC peptide-specific distribution along the intestine of Nile tilapia. We assessed the density of ECs producing gastrin (GAS), cholecystokinin-8 (CCK-8), neuropeptide Y (NPY), and calcitonin gene-related peptide (CGRP) in nine segments of the intestine using immunohistochemistry. Our results show that ECs immunoreactive to CCK-8, GAS, NPY, and CGRP can be found along all the intestinal segments sampled, from the midgut to hindgut, although differences in their distribution along the gut were observed. Regarding nutrient status, we found that the anterior segments of the midgut seem to be the main site responding to luminal changes in Nile tilapia. The NPY+ and CGRP+ EC densities increased in the fasted group, while the amount of CCK-8+ ECs were higher in the fed group. No effects of fasting or feeding were found in the GAS+ EC densities. Changes in ECs density were found only at the anterior segments of the intestine which may be due to the correlation between vagus nerve anatomy, EC location, and peptide turnover. Lastly, ECs may need to be considered an active cell subpopulation that may adapt and respond to different nutrient status as stimuli. Due to the complexity of the enteroendocrine system and its importance in fish nutrition, much remains to be elucidated and it deserves closer attention.




Similar content being viewed by others
Abbreviations
- DNES:
-
Diffuse neuroendocrine system
- GAS:
-
Gastrin
- CCK-8:
-
Cholecystokinin-8
- NPY:
-
Neuropeptide Y
- CGRP:
-
Calcitonin gene-related peptide
- ECs:
-
Endocrine cells
- +ECs mm−2 :
-
Immunoreactive endocrine cells per square millimeter of mucosal epithelium
- S1–S9:
-
First to ninth segment of intestine
References
ACEB (2014) 1o Anuário Brasileiro da Pesca e Aquicultura/1st Brazilian fishery and aquaculture yearbook. Associacao Cultural e Educacional Brasil. ACEB, Brasilia
Ahlman H, Nilsson O (2001) The gut as the largest endocrine organ in the body. Ann Oncol 12:63–68. doi:10.1093/annonc/12.suppl_2.S63
Aldman G, Jönsson AC, Jensen J, Holmgren S (1989) Gastrin/CCK-like peptides in the spiny dogfish, Squalus acanthias; concentrations and actions in the gut. Comp Biochem Physiol Part C Comp 92:103–108. doi:10.1016/0742-8413(89)90210-7
Aldman G, Grove D, Holmgren S (1992) Duodenal acidification and intra-arterial injection of CCK8 increase gallbladder motility in the rainbow trout, Oncorhynchus mykiss. Gen Comp Endocrinol 86:20–25. doi:10.1016/0016-6480(92)90121-Y
Bermúdez R, Vigliano F, Quiroga MI et al (2007) Immunohistochemical study on the neuroendocrine system of the digestive tract of turbot, Scophthalmus maximus (L.), infected by Enteromyxum scophthalmi (Myxozoa). Fish Shellfish Immunol 22:252–263. doi:10.1016/j.fsi.2006.05.006
Bosi G, Di Giancamillo A, Arrighi S, Domeneghini C (2004) An immunohistochemical study on the neuroendocrine system in the alimentary canal of the brown trout, Salmo trutta, L., 1758. Gen Comp Endocrinol 138:166–181. doi:10.1016/j.ygcen.2004.06.003
Çınar K, Şenol N, Özen MR (2006) Immunohistochemical study on distribution of endocrine cells in gastrointestinal tract of flower fish (Pseudophoxinus antalyae). World J Gastroenterol 12:6874–6878
Cummings DE, Overduin J (2007) Review series gastrointestinal regulation of food intake. Health Care (Don Mills) 117:13–23. doi:10.1172/JCI30227.example
Dezfuli BS, Arrighi S, Domeneghini C, Bosi G (2000) Immunohistochemical detection of neuromodulators in the intestine of Salmo trutta L. naturally infected with Cyathocephalus truncatus Pallas (Cestoda). J Fish Dis 23:265–273. doi:10.1046/j.1365-2761.2000.00234.x
Dockray GJ (2009) Cholecystokinin and gut-brain signalling. Regul Pept 155:6–10
Dockray GJ (2010) How the gut sends signals in response to food. Int Dairy J 20:226–230. doi:10.1016/j.idairyj.2009.11.013
Dockray GJ (2013) Enteroendocrine cell signalling via the vagus nerve. Curr Opin Pharmacol 13:954–958. doi:10.1016/j.coph.2013.09.007
Dockray GJ (2014) Gastrointestinal hormones and the dialogue between gut and brain. J Physiol 0:1–15. doi:10.1113/jphysiol.2014.270850
Domeneghini C, Radaelli G, Arrighi S et al (2000) Neurotransmitters and putative neuromodulators in the gut of Anguilla anguilla (L.). Localizations in the enteric nervous and endocrine systems. Eur J Histochem 44:295–306
El-Sayed A-FM (2006) Tilapia culture. CABI Publishing, Wallingford, pp 277
Engelstoft MS, Egerod KL, Holst B, Schwartz TW (2008) A gut feeling for obesity: 7TM sensors on enteroendocrine cells. Cell Metab 8:447–449. doi:10.1016/j.cmet.2008.11.004
FAO (2016) The state of world fisheries and aquaculture: contributing to food security and nutrition for all. Food and Agriculture Organization, FAO, Rome
Gonçalves MAD, Bello NM, Dritz SS et al (2016) An update on modeling dose???Response relationships: accounting for correlated data structure and heterogeneous error variance in linear and nonlinear mixed models. J Anim Sci 94:1940–1950. doi:10.2527/jas2015-0106
Gutierrez-Aguilar R, Woods SC (2011) Nutrition and L and K-enteroendocrine cells. Curr Opin Endocrinol Diabetes Obes 18:35–41. doi:10.1097/MED.0b013e32834190b5
Hartviksen MB, Kamisaka Y, Jordal AEO et al (2009) Distribution of cholecystokinin-immunoreactive cells in the gut of developing atlantic cod Gadus morhua L. larvae fed zooplankton or rotifers. J Fish Biol 75:834–844. doi:10.1111/j.1095-8649.2009.02325.x
HE M, Wang K y, Zhang Y (2009) Immunocytochemical identification and localization of diffuse neuroendocrine system (DNES) cells in gastrointestinal tract of channel catfish (Ictalurus punctatus). Agric Sci China 8:238–243. doi:10.1016/S1671-2927(09)60032-8
Helander HF, Fändriks L (2012) The enteroendocrine “letter cells”—time for a new nomenclature? Scand J Gastroenterol 47:3–12. doi:10.3109/00365521.2011.638391
Hernández DR, Vigliano FA, Sánchez S et al (2012) Neuroendocrine system of the digestive tract in Rhamdia quelen juvenile: an immunohistochemical study. Tissue Cell 44:220–226. doi:10.1016/j.tice.2012.03.005
Hoskins LJ, Volkoff H (2012) The comparative endocrinology of feeding in fish: insights and challenges. Gen Comp Endocrinol 176:327–335. doi:10.1016/j.ygcen.2011.12.025
Hur SW, Lee CH, Lee SH et al (2013) Characterization of cholecystokinin-producing cells and mucus-secreting goblet cells in the blacktip grouper, Epinephelus fasciatus. Tissue Cell 45:153–157. doi:10.1016/j.tice.2012.10.005
Jensen J (2001) Regulatory peptides and control of food intake in non-mammalian vertebrates. Comp Biochem Physiol A Mol Integr Physiol 128(3):471–479. doi:10.1016/S1095-6433(00)00329-9
Ji W, Ping HC, Wei KJ et al (2015) Ghrelin, neuropeptide Y (NPY) and cholecystokinin (CCK) in blunt snout bream (Megalobrama amblycephala): CDNA cloning, tissue distribution and mRNA expression changes responding to fasting and refeeding. Gen Comp Endocrinol 223:108–119. doi:10.1016/j.ygcen.2015.08.009
Jönsson AC, Holmgren S, Holstein B (1987) Gastrin/CCK-like immunoreactivity in endocrine cells and nerves in the gastrointestinal tract of the cod, Gadus morhua, and the effect of peptides of the gastrin/CCK family on cod gastrointestinal smooth muscle. Gen Comp Endocrinol 66:190–202. doi:10.1016/0016-6480(87)90267-X
Kamal M, Kurt A, Brown Michael L (2010) Tilapia: environmental biology and nutritional requirements. South Dakota Coopertaive Ext Serv Fs963-02 7. doi:10.1016/j.aquaculture.2005.04.020
Kamisaka Y, Drivenes O, Kurokawa T et al (2005) Cholecystokinin mRNA in Atlantic herring, Clupea harengus—molecular cloning, characterization, and distribution in the digestive tract during the early life stages. Peptides 26:385–393. doi:10.1016/j.peptides.2004.10.018
Kehoe AS, Volkoff H (2007) Cloning and characterization of neuropeptide Y (NPY) and cocaine and amphetamine regulated transcript (CART) in Atlantic cod (Gadus morhua). Comp Biochem Physiol - A Mol Integr Physiol 146:451–461. doi:10.1016/j.cbpa.2006.12.026
Kumar GL, Rudbeck L (2009) Immunohistochemical staining methods, 5th edn. Dako North America, Carpinteria
Kurokawa T, Suzuki T, Hashimoto H (2003) Identification of gastrin and multiple cholecystokinin genes in teleost. Peptides 24:227–235. doi:10.1016/S0196-9781(03)00034-2
Lee JH, Ku SK, Park KD, Lee HS (2004) Immunohistochemical study of the gastrointestinal endocrine cells in the Korean aucha perch. J Fish Biol 65:170–181. doi:10.1111/j.0022-1112.2004.00442.x
MacDonald E, Volkoff H (2009) Cloning, distribution and effects of season and nutritional status on the expression of neuropeptide Y (NPY), cocaine and amphetamine regulated transcript (CART) and cholecystokinin (CCK) in winter flounder (Pseudopleuronectes americanus). Horm Behav 56:58–65. doi:10.1016/j.yhbeh.2009.03.002
Martinez V, Wang L, Taché Y (2006) Peripheral adrenomedullin inhibits gastric emptying through CGRP8–37-sensitive receptors and prostaglandins pathways in rats. Peptides 27:1376–1382. doi:10.1016/j.peptides.2005.11.003
Martínez-Álvarez RM, Volkoff H, Muñoz-Cueto JA, Delgado MJ (2008) Molecular characterization of calcitonin gene-related peptide (CGRP) related peptides (CGRP, amylin, adrenomedullin and adrenomedullin-2/intermedin) in goldfish (Carassius auratus): cloning and distribution. Peptides 29:1534–1543. doi:10.1016/j.peptides.2008.04.013
Martínez-Alvarez RM, Volkoff H, Muñoz-Cueto JA, Delgado MJ (2009) Effect of calcitonin gene-related peptide (CGRP), adrenomedullin and adrenomedullin-2/intermedin on food intake in goldfish (Carassius auratus). Peptides 30:803–807. doi:10.1016/j.peptides.2008.12.015
May C, Kaestner K (2010) Gut endocrine cell development. Mol Cell Endocrinol 323:70–75. doi:10.1016/j.mce.2009.12.009.Gut
Micale V, Campo S, D’Ascola A et al (2012) Cholecystokinin in White Sea bream: molecular cloning, regional expression, and Immunohistochemical localization in the gut after feeding and fasting. PLoS One. doi:10.1371/journal.pone.0052428
Micale V, Campo S, D’Ascola A et al (2014) Cholecystokinin: how many functions? Observations in seabreams. Gen Comp Endocrinol 205:166–167. doi:10.1016/j.ygcen.2014.02.019
Morrison CM, Wright J (1999) A study of the histology of the digestive tract of the Nile tilapia. J Fish Biol 54:597–606. doi:10.1006/jfbi.1998.0890
Nag K, Kato A, Nakada T et al (2006) Molecular and functional characterization of adrenomedullin receptors in pufferfish. Am J Physiol Regul Integr Comp Physiol 290:R467–R478. doi:10.1152/ajpregu.00507.2005
NRC (2011) Nutrient requirement council: nutrient requirements of fish and shrimp. National Academies Press, Washington
Olsson C (2009) Autonomic innervation of the fish gut. Acta Histochem 111:185–195. doi:10.1016/j.acthis.2008.11.014
Olsson C, Holmgren S (2001) The control of gut motility. Comp Biochem Physiol A Mol Integr Physiol 128:481–503. doi:10.1016/s1095-6433(00)00330-5
Olsson C, Aldman G, Larsson A, Holmgren S (1999) Cholecystokinin affects gastric emptying and stomach motility in the rainbow trout Oncorhynchus mykiss. J Exp Biol 202:161–170
Pan QS, Fang ZP, Zhao YX (2000) Immunocytochemical identification and localization of APUD cells in the gut of seven stomachless teleost fishes. World J Gastroenterol 6:96–101
Pereira RT, Costa LS, Oliveira IRC et al (2015) Relative distribution of gastrin-, CCK-8-, NPY- and CGRP-immunoreactive cells in the digestive tract of dorado (Salminus brasiliensis). Tissue Cell 47:123–131. doi:10.1016/j.tice.2015.01.009
Powley TL, Phillips RJ (2004) Gastric satiation is volumetric, intestinal satiation is nutritive. Physiol Behav 82:69–74. doi:10.1016/j.physbeh.2004.04.037
Riche M, Haley DI, Oetker M et al (2004) Effect of feeding frequency on gastric evacuation and the return of appetite in tilapia Oreochromis niloticus (L.) Aquaculture 234:657–673. doi:10.1016/j.aquaculture.2003.12.012
Rindi G, Leiter AB, Kopin AS et al (2004) The “normal” endocrine cell of the gut: changing concepts and new evidences. Ann N Y Acad Sci 1014:1–12. doi:10.1196/annals.1294.001
Rønnestad I, Kamisaka Y, Conceição LEC et al (2007) Digestive physiology of marine fish larvae: hormonal control and processing capacity for proteins, peptides and amino acids. Aquaculture 268:82–97. doi:10.1016/j.aquaculture.2007.04.031
Schubert ML, Makhlouf GM (1992) Neural, hormonal, and paracrine regulation of gastrin and acid secretion. Yale J Biol Med 65:553–560
Shahbazi F, Karila P, Olsson C et al (1998) Primary structure, distribution, and effects on motility of CGRP in the intestine of the cod Gadus morhua. Am J Physiol-Regul Integr Comp Physiol 275:R19–R28
Steinert RE, Beglinger C (2011) Nutrient sensing in the gut: interactions between chemosensory cells, visceral afferents and the secretion of satiation peptides. Physiol Behav 105:62–70. doi:10.1016/j.physbeh.2011.02.039
Takei Y, Loretz CA (2011) The gastrointestinal tract as an endocrine, paracrine and autocrine organ. In Grosell M, Farrell AP, Brauner CJ (eds) Fish physiology vol 30, the multifunctional gut of fish. Academic Press, San Diego, pp 262–300
Vigliano FA, Muñoz L, Hernández D et al (2011) An immunohistochemical study of the gut neuroendocrine system in juvenile pejerrey Odontesthes bonariensis (Valenciennes). J Fish Biol 78:901–911. doi:10.1111/j.1095-8649.2011.02912.x
Volkoff H (2016) The neuroendocrine regulation of food intake in fish: a review of current knowledge. Front Neurosci 10:1–31. doi:10.3389/fnins.2016.00540
Volkoff H, Hoskins LJ, Tuziak SM (2010) Influence of intrinsic signals and environmental cues on the endocrine control of feeding in fish: potential application in aquaculture. Gen Comp Endocrinol 167:352–359. doi:10.1016/j.ygcen.2009.09.001
Webb KA, Khan IA, Nunez BS et al (2010) Cholecystokinin: molecular cloning and immunohistochemical localization in the gastrointestinal tract of larval red drum, Sciaenops ocellatus (L.) Gen Comp Endocrinol 166:152–159. doi:10.1016/j.ygcen.2009.10.010
Wilson JM, Castro LFC (2011). Morphological diversity of the gastrointestinal tract in fishes. In Grosell M, Farrell AP, Brauner CJ (eds) Fish physiology vol 30, the multifunctional gut of fish. Academic Press, San Diego, pp 2–44
Yokobori E, Azuma M, Nishiguchi R et al (2012) Neuropeptide Y stimulates food intake in the zebrafish, Danio rerio. J Neuroendocrinol 24:766–773. doi:10.1111/j.1365-2826.2012.02281.x
Yuan D, Wang T, Zhou C et al (2014) Leptin and cholecystokinin in Schizothorax prenanti: molecular cloning, tissue expression, and mRNA expression responses to periprandial changes and fasting. Gen Comp Endocrinol 204:13–24. doi:10.1016/j.ygcen.2014.05.013
Zhou Y, Liang XF, Yuan X et al (2013) Neuropeptide Y stimulates food intake and regulates metabolism in grass carp, Ctenopharyngodon idellus. Aquaculture 380–383:52–61. doi:10.1016/j.aquaculture.2012.11.033
Acknowledgements
This work was supported by a cooperative project between Brazil and Argentina (Process No. CAPG030 CAPES). We thank the financial support given to Priscila Vieira Rosa (grants Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, CAPES, Brazil) and to Fabricio A. Vigliano (grants PICT 2014-1858 from Agencia Nacional de Promoción Científica y Tecnológica and 2010-169-14 from Ministerio de Ciencia, Tecnología e Innovación Productiva de Santa Fe). We also thank the Fundação de Amparo à Pesquisa de Minas Gerais, FAPEMIG, for the research funding granted to Priscila Vieira Rosa (PPM 00227/12) and the Ph.D. scholarship from Conselho Nacional de Desenvolvimento Científico e Tecnológico, CNPq, granted to Raquel T. Pereira (No. 153616/2012-1), which made possible the accomplishment of this work. The technical training in gastrointestinal morphology and immunohistochemistry given to Raquel T. Pereira at Cátedra de Histología y Embriología, Facultad de Ciencias Veterinarias, Universidad Nacional de Rosario, Argentina, is also gratefully acknowledged. We thank Vanessa Seiko Sugihara for the schematic drawing of the Nile tilapia digestive tract.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Pereira, R.T., de Freitas, T.R., de Oliveira, I.R.C. et al. Endocrine cells producing peptide hormones in the intestine of Nile tilapia: distribution and effects of feeding and fasting on the cell density. Fish Physiol Biochem 43, 1399–1412 (2017). https://doi.org/10.1007/s10695-017-0380-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-017-0380-1