Abstract
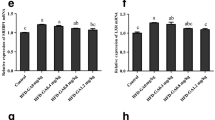
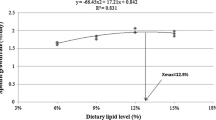
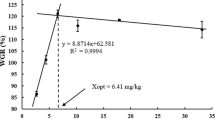
Fatty liver syndrome is a prevalent problem of farmed fish. Conjugated linoleic acid (CLA) has received increased attention recently as a fat-reducing fatty acid to control fat deposition in mammals. Therefore, the aim of the present study was to determine whether dietary CLA can reduce tissue lipid content of darkbarbel catfish (Pelteobagrus vachelli) and whether decreased lipid content is partially due to alterations in lipid metabolism enzyme activities and fatty acid profiles. A 76-day feeding trial was conducted to investigate the effect of dietary CLA on the growth, tissue lipid deposition, and fatty acid composition of darkbarbel catfish. Five diets containing 0 % (control), 0.5 % (CLA0.5), 1 % (CLA1), 2 % (CLA2), and 3 % (CLA3) CLA levels were evaluated. Results showed that fish fed with 2–3 % CLA diets showed a significantly lower specific growth rate and feed conversion efficiency than those fed with the control diet. Dietary CLA decreased the lipid contents in the liver and intraperitoneal fat with the CLA levels from 1 to 3 %. Fish fed with 2–3 % CLA diets showed significantly higher lipoprotein lipase and hepatic triacylglycerol lipase activities in liver than those of fish fed with the control, and fish fed with 1–3 % CLA diets had significantly higher pancreatic triacylglycerol lipase activities in liver than those of fish fed with the control. Dietary CLA was incorporated into liver, intraperitoneal fat, and muscle lipids, with higher percentages observed in liver compared with other tissues. Liver CLA deposition was at the expense of monounsaturated fatty acids (MUFA). In contrast, CLA deposition appeared to be primarily at the expense of MUFA and n-3 polyunsaturated fatty acids (PUFA) in the intraperitoneal fat, whereas in muscle it was at the expense of n-3 PUFA. Our results suggested that CLA at a 1 % dose can reduce liver lipid content without eliciting any negative effect on growth rate in darkbarbel catfish. This lipid-lowering effect could be in part due to an increment in the activity of lipid metabolism enzymes and an extensive interconversion of fatty acids. Although CLA deposition in muscle (0.66–3.19 % of total fatty acids) are higher than presented in natural sources of CLA, EPA (C20:5n-3) in fish muscle appears simultaneously expendable, when the fish fed with 2–3 % CLA.

Similar content being viewed by others
Abbreviations
- HTL:
-
Hepatic triacylglycerol lipase
- HUFA:
-
Total highly unsaturated fatty acids
- IPF:
-
Intraperitoneal fat ratio
- LPL:
-
Lipoprotein lipase
- MUFA:
-
Total monounsaturated fatty acids
- PTL:
-
Pancreatic triacylglycerol lipase
- PUFA:
-
Polyunsaturated fatty acids
- SFA:
-
Total saturated fatty acids
References
AOAC (Association of Official Analytical Chemists) (2003) Official methods of analysis of official analytical chemists international, 17th edn. Association of Official Analytical Chemists, Arlington
Bandarra NM, Nunes ML, Andrade AM, Prates JAM, Pereira S, Monteiro M, Rema P, Valente LMP (2006) Effect of dietary conjugated linoleic acid on muscle, liver and visceral lipid deposition in rainbow trout juveniles (Oncorhynchus mykiss). Aquaculture 254:496–505
Bell JG (1998) Current aspects of lipid nutrition in fish farming. In: Black KD, Pickering AD (eds) Biology of farmed fish. Sheffield Academic, Sheffield, pp 114–145
Berge GM, Ruyter B, Asgard T (2004) Conjugated linoleic acid in diets for juvenile Atlantic salmon (Salmo salar): effects on fish performance, proximate composition, fatty acid and mineral content. Aquaculture 237:365–380
Bradford M (1976) A rapid sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chaiyapechara S, Casten MT, Hardy RW, Dong FW (2003) Fish performance, fillet characteristics, and health assessment index of rainbow trout (Oncorhynchus mykiss) fed diets containing adequate and high concentrations of lipid and vitamin E. Aquaculture 219:715–738
Choi BD, Kang SJ, Ha YL, Ackman RG (1999) Accumulation of conjugated linoleic acid (CLA) in tissues of fish fed diets containing various levels of CLA. In: Xiong YL, Ho CT, Shahidi F (eds) Quality attributes of muscle foods. Kluwer Academic/Plenum Publishers, New York, pp 61–71
Choi JS, Jung MH, Park HS, Song J (2004) Effect of conjugated linoleic acid isomers on insulin resistance and mRNA levels of genes regulating energy metabolism in high-fat-fed rats. Nutrition 20:1008–1017
Christie WW (1982) Lipid analysis, 2nd edn. Pergamon, Oxford, p 207
Cohen P, Miyazaki M, Socci ND, Hagge-Greenberg A, Liedtke W, Soukas AA, Sharma R, Hudgins LC, Ntambi JM, Friedman JM (2002) Role for stearoyl-CoA desaturase-1 in leptin-mediated weight loss. Science 297:240–243
Diez A, Menoyo D, Pérez-Benavente S, Calduch-Giner JA, de Celis SVR, Obach A, Favre-Krey L, Boukouvala E, Leaver MJ, Tocher DR, Pérez-Sanchez J, Krey G, Bautista JM (2007) Conjugated linoleic acid affects lipid composition, metabolism and gene expression in gilthead sea bream (Sparus aurata L.). J Nutr 137:1363–1369
Dong GF, Zou Q, Wang H, Huang F, Liu XC, Chen L, Yang CY, Yang YO (2014a) Conjugated linoleic acid differentially modulates growth, tissue lipid deposition, and gene expression involved in the lipid metabolism of grass carp. Aquaculture 432:181–191
Dong GF, Zhu XM, Ren HX, Nie BY, Chen L, Li H, Yan B (2014b) Effects of oxidized fish oil intake on tissue lipid metabolism and fatty acid composition of channel catfish (Ictalurus punctatus). Aquac Res 45:1867–1880
Du ZY, Clouet P, Zheng WH, Degrace P, Tian LX, Liu YJ (2006) Biochemical hepatic alterations and body lipid composition in the herbivorous grass carp (Ctenopharyngodon idella) fed high-fat diets. Br J Nutr 95:905–915
Fielding BA, Frayn KN (1998) Lipoprotein lipase and the disposition of dietary fatty acids. Br J Nutr 80:495–502
Figueiredo-Silva AC, Rema P, Bandarra NM, Nunes ML, Valente LMP (2005) Effects of dietary conjugated linoleic acid on growth, nutrient utilization, body composition, and hepatic lipogenesis in rainbow trout juveniles (Oncorhynchus mykiss). Aquaculture 248:163–172
Folch J, Lees M, Stanley GHS (1957) A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem 226:497–509
Gao W, Liu YJ, Tian LX, Mai KS, Liang GY, Yang HJ, Huai MY (2011) Protein-sparing capability of dietary lipid in herbivorous and omnivorous freshwater finfish: a comparative case study on grass carp (Ctenopharyngodon idella) and tilapia (Oreochromis niloticus × O. aureus). Aquac Nutr 17:2–12
Gaullier JM, Halse J, Høivik HO, Høye K, Syvertsen C, Nurminiemi M, Hassfeld C, Einerhand A, O’shea M, Gudmundsen O (2007) Six months supplementation with conjugated linoleic acid induces regional-specific fat mass decreases in overweight and obese. Br J Nutr 97:550–560
House RL, Cassady JP, Eisen EJ, Eling TE, Collins JB, Grissom SF, Odle J (2005) Functional genomic characterization of delipidation elicited by trans-10, cis-12-conjugated linoleic acid (t10c12-CLA) in a polygenic obese line of mice. Physiol Genomics 21:351–361
Inoue N, Nagao K, Hirata J, Wang YM, Yanagita T (2004) Conjugated linoleic acid prevents the development of essential hypertension in spontaneously hypertensive rats. Biochem Biophys Res Commun 323:679–684
Kaushik SJ, Luquet P (1984) Relationship between protein intake and voluntary energy intake as affected by body weight with an estimation of maintenance needs in rainbow trout. Z Tierphysiol Tierernahr Futtermittelkd 51:57–69
Kennedy SR, Campbell PJ, Porter A, Tocher DR (2005) Influence of dietary conjugated linoleic acid (CLA) on lipid and fatty acid composition in liver and flesh of Atlantic salmon (Salmo salar). Comp Biochem Physiol 141B:168–178
Kennedy SR, Leaver MJ, Campbell PJ, Zheng X, Dick JR, Tocher DR (2006) Influence of dietary oil content and conjugated linoleic Acid (CLA) on lipid metabolism enzyme activities and gene expression in tissues of Atlantic salmon (Salmo salar L.). Lipids 41:423–436
Kennedy SR, Bickerdike R, Berge RK, Dick JR, Tocher DR (2007) Influence of conjugated linoleic acid (CLA) or tetradecylthioacetic acid (TTA) on growth, lipid composition, fatty acid metabolism and lipid gene expression of rainbow trout (Oncorhynchus mykiss L.). Aquaculture 272:489–501
Leaver MJ, Tocher DR, Obach A, Jensen L, Henderson RJ, Porter AR, Krey G (2006) Effect of dietary conjugated linoleic acid (CLA) on lipid composition, metabolism and gene expression in Atlantic salmon (Salmo salar) tissues. Comp Biochem Physiol 145A:258–267
Lin H, Boylston D, Chang MJ, Luedecke LO, Shultz TD (1995) Survey of the conjugated linoleic acid contents of dairy products. J Dairy Sci 78:2358–2365
Luo Z, Tan XY, Liu CX, Li XD, Liu XJ, Xi WQ (2012) Effect of dietary conjugated linoleic acid levels on growth performance, muscle fatty acid profile, hepatic intermediary metabolism and antioxidant responses in genetically improved farmed Tilapia strain of Nile tilapia Oreochromis niloticus. Aquac Res 43:1392–1403
Makol A, Torrecillas S, Fernandez-Vaquero A, Robaina L, Montero D, Caballero MJ, Tort L, Izquierdo M (2009) Effect of conjugated linoleic acid on dietary lipids utilization, liver morphology and selected immune parameters in sea bass juveniles (Dicentrarchus labrax). Comp Biochem Physiol 154B:179–187
Makol A, Torrecillas S, Caballero MJ, Fernández-Vaquero A, Izquierdo MS (2012) Effect of long term feeding with conjugated linoleic acid (CLA) in growth performance and lipid metabolism of European sea bass (Dicentrarchus labrax). Aquaculture 368–369:129–137
Manning BB, Li MH, Robinson EH, Peterson BC (2006) Enrichment of channel catfish (Ictalurus punctatus) fillets with conjugated linoleic acid and omega-3 fatty acids by dietary manipulation. Aquaculture 261:337–342
Nilsson A, Landin B, Schotz MC (1987) Hydrolysis of chylomicron arachidonate and linoleate ester bonds by lipoprotein lipase and hepatic lipase. J Lipid Res 28:510–517
Ntambi JM, Miyazaki M, Stoehr JP, Lan H, Kendziorski CM, Yandell BS, Song Y, Cohen P, Friedman JM, Attie DA (2002) Loss of stearoyl-CoA desaturase-1 function protects mice against adiposity. Proc Natl Acad Sci USA 99:11482–11486
Pariza MW, Park Y, Cook ME (2001) The biological active isomers of conjugated linoleic acid. Prog Lipid Res 40:283–298
Park Y, Storkson JM, Liu W, Albright KJ, Cook ME, Pariza MW (2004) Structure-activity relationship of conjugated linoleic acid and its cognates in inhibiting heparin-releasable lipoprotein lipase and glycerol release from fully differentiated 3T3-L1 adipocytes. J Nutr Biochem 15:561–568
Ramos A, Bandarra NM, Rema P, Vaz-Pires P, Nunes ML, Andrade AM, Cordeiro AR, Valente LMP (2008) Time course deposition of conjugated linoleic acid in market size rainbow trout (Oncorhynchus mykiss) muscle. Aquaculture 274:366–374
Santos LD, Furuya WM, Silva LCR, Matsushita M, Silva TSC (2011) Dietary conjugated linoleic acid (CLA) for finishing Nile tilapia. Aquac Nutr 17:e70–e81
Sargent JR, Tocher DR, Bell JG (2002) The lipids. In: Halver JE, Hardy RW (eds) Fish nutrition, 3rd edn. Academic Press, San Diego, pp 181–257
Schmid A, Collomb M, Sieber R, Bee G (2006) Conjugated linoleic acid in meat and meat products: a review. Meat Sci 73:29–41
Smith SB, Hively TS, Cortese GM, Han JJ, Chung KY, Casteñada P, Gilbert CD, Adams VL, Mersmann HJ (2002) Conjugated linoleic acid depresses the δ9 desaturase index and stearoyl coenzyme A desaturase enzyme activity in porcine subcutaneous adipose tissue. J Anim Sci 80:2110–2115
Tan XY, Luo Z, Xie P, Li XD, Liu XJ, Xi WQ (2010) Effect of dietary conjugated linoleic acid (CLA) on growth performance, body composition and hepatic intermediary metabolism in juvenile yellow catfish Pelteobagrus fulvidraco. Aquaculture 310:186–191
Tocher DR (2003) Metabolism and functions of lipids and fatty acids in teleost fish. Rev Fish Sci 11:107–184
Twibell RG, Wilson RP (2003) Effects of conjugated linoleic acids and total lipid concentrations on growth responses of juvenile channel catfish, Ictalurus punctatus. Aquaculture 221:621–628
Twibell RG, Watkins BA, Rogers L, Brown PB (2000) Effects of dietary conjugated linoleic acids on hepatic and muscle lipids in hybrid striped bass. Lipids 35:155–161
Twibell RG, Watkins BA, Brown PB (2001) Dietary conjugated linoleic acids and lipid source alter fatty acid composition of juvenile yellow perch, Perca flavescen. J Nutr 131:2322–2328
Valente LMP, Bandarra NM, Figueiredo-Silva AC, Rema P, Vaz-Pires P, Martins S, Pratese JAM, Nunes ML (2007a) Conjugated linoleic acid in diets for large size rainbow trout (Oncorhynchus mykiss): effects on growth, chemical composition and sensory attributes. Br J Nutr 97:289–297
Valente LMP, Bandarra NM, Figueiredo-Silva AC, Cordeiro AR, Simoes RM, Nunes ML (2007b) Influence of conjugated linoleic acid on growth, lipid composition and hepatic lipogenesis in juvenile European sea bass (Dicentrarchus labrax). Aquaculture 267:225–235
Wahle KW, Heys SD, Rotondo D (2004) Conjugated linoleic acids: are they beneficial or detrimental to health? Prog Lipid Res 43:553–587
Wijendran V, Pronczuk A, Bertoli C, Hayes KC (2003) Dietary trans-18:1 raises plasma triglycerides and VLDL cholesterol when replacing either 16:0 or 18:0 in gerbils. J Nutr Biochem 14:584–590
Yasmin A, Takeuchi T (2002) Influence of various dietary levels of conjugated linoleic acid (CLA) on juvenile tilapia Oreochromis niloticus. Fish Sci 68(Suppl.):991–992
Yasmin A, Takeuchi T, Hayashi M, Hirota T, Ishizuka W, Ishida S (2004) Effect of conjugated linoleic and docosahexaenoic acids on growth of juvenile tilapia Oreochromis niloticus. Fish Sci 70:473–481
Acknowledgments
This research was supported by the National Natural Science Foundation of China (Grant No. 31302195), the Natural Science Foundation of Hubei Province of China (Grant No. 2011CDB225), International Science & Technology Cooperation Program of China (Grant No. S2012ZR0135), the special fund for agro-scientific research in the public interest from the Ministry of Agriculture, China (201503242), and Hubei Collaborative Innovation Center for Animal Nutrition and Feed Safety.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dong, GF., Liu, WZ., Wu, LZ. et al. Conjugated linoleic acid alters growth performance, tissue lipid deposition, and fatty acid composition of darkbarbel catfish (Pelteobagrus vachelli). Fish Physiol Biochem 41, 73–89 (2015). https://doi.org/10.1007/s10695-014-0007-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-014-0007-8