Abstract
A low level of available potassium (K) in soils is a major factor restricting crop growth and production. In earlier studies we found that Tibetan wild barley has a wider variation in low K stress tolerance than cultivated barley. In this study, we performed a genome-wide association analysis of 179 Tibetan wild barley (Hordeum vulgare L.) accessions exposed to low K stress and investigated the associations of K uptake rate (KUR), K translocation rate (KTR) and other K metabolism traits with 11,013 diversity arrays technology markers. The results showed that there were significant differences in tissue K concentration and content, KUR and KTR among the wild barley accessions analyzed. Scores for all traits showed a normal distribution. Three significant quantitative trait loci (QTLs) for KUR and KTR were identified and located on 6H and 1H. These QTLs were found to be associated with the cation/H(+) antiporter and cyclic nucleotide gated channel. A number of unique candidate genes associated with K-related signals, including K channels, K transporters and ethylene-related genes, were also identified. The K channels and K transporters involved in K uptake and translocation, respectively, were closely associated with KUR in response to low K stress. The present study highlights the potential of Tibetan wild barley to provide elite candidate genes for the improvement of K use efficiency of barley.





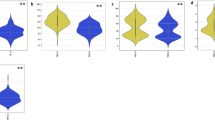
Arrows The point indicated by the arrow represents the significant association signals

Arrows The point indicated by the arrow represents the significant association signals



Similar content being viewed by others
References
Amtmann A, Troufflard S, Armengaud P (2008) The effect of potassium nutrition on pest and disease resistance in plants. Physiol Plant 133:682–691
Balagué C, Lin B, Alcon C, Flottes G, Malmström S, Köhler C, Neuhaus G, Pelletier G, Gaymard F, Roby D (2003) HLM1, an essential signalling component in the hypersensitive response, is a member of the cyclic nucleotide-gated channel ion channel family. Plant Cell 15:365–379
Britto DT, Kronzucker HJ (2008) Cellular mechanisms of potassium transport in plants. Physiol Plant 133:637–650
Cai SG, Wu DZ, Jabeen Z, Huang YQ, Huang YC, Zhnag GP (2013) Genome-wide association analysis of aluminum tolerance in cultivated and Tibetan wild barley. PLoS ONE 8(7):e69776. https://doi.org/10.1371/journal.pone.0069776
Ceccarelli S, Grando S, Van Leur JAG (1995) Barley landraces of the fertile crescent offer new breeding options for stress environments. Diversity 11:112–113
Cellier F, Conejero G, Ricaud L, Luu DT, Lepetit M, Gosti F, Casse F (2004) Characterization of AtCHX17, a member of the cation/H+ exchangers, CHX family, from Arabidopsis thaliana suggests a role in K+ homeostasis. Plant J 39:834–846
Chan CWM, Schorrak LM, Smith RK, Bent AF, Sussman MR (2003) A cyclic nucleotide-gated ion channel, CNGC2, is crucial for plant development and adaptation to calcium stress. Plant Physiol 132:728–731
Chanroj S, Lu Y, Padmanaban S, Nanatani K, Uozumi N, Rao R, Sze H (2011) Plant-specific cation/H+ exchanger 17 and its homologs are endomembrane K+ transporters with roles in protein sorting. J Biol Chem 286(39):33931–33941
Christopher DA, Borsics T, Yuen CYL, Ullmer W, Andème- Ondzighi C, Andres MA, Kang B-H, Staehelin LA (2007) The cyclic nucleotide gated cation channel AtCNGC10 traffics from the ER via Golgi vesicles to the plasma membrane of Arabidopsis root and leaf cells. BMC Plant Biol 7:48
Coskun D, Britto DT, Kronzucker HJ (2014) The physiology of channel-mediated K+ acquisition in roots of higher plants. Physiol Plant 151:305–312
Dai F, Nevo E, Wu DZ, Comadran J, Zhou MX, Qiu L, Chen ZH, Beiles A, Chen GX, Zhang GP (2012) Tibet is one of the centers of domestication of cultivated barley. Proc Natl Acad Sci USA 109:16969–16973
Deeken R, Ivashikina N, Czirjak T, Philippar K, Becker D, Ache P, Hedrich R (2003) Tumour development in Arabidopsis thaliana involves the Shaker-like K+ channels AKT1 and AKT2/3. Plant J 34:778–787
Food and Agriculture Organization of the United Nations (FAO) (2017) World fertilizer trends and outlook to 2020. FAO, Rome
Fuchs I, Stolzle S, Ivashikina N, Hedrich R (2005) Rice K+ uptake channel OsAKT1 is sensitive to salt stress. Planta 221:212–221
Gajdanowicz P, Michard E, Sandmann M, Rocha M, Correa LGG et al (2011) Potassium (K+) gradients serve as a mobile energy source in plant vascular tissues. Proc Natl Acad Sci USA 108:864–869
Garcia-Oliveira AL, Tan L, Fu Y, Sun C (2009) Genetic identification of quantitative trait loci for contents of mineral nutrients in rice grain. J Integr Plant Biol 51:84–92
Gierth M, Mäser P, Schroeder JI (2005) The potassium transporter AtHAK5 functions in K+ deprivation-induced high-affinity K+ uptake and AKT1 K+ channel contribution to K+ uptake kinetics in Arabidopsis roots. Plant Physiol 137:1105–1114
Glass ADM, Perley JE (1980) Varietal differences in potassium uptake by barley. Plant Physiol 65:160–164
Gobert A, Park G, Amtmann A, Sanders D, Maathuis FJM (2006) Arabidopsis thaliana cyclic nucleotide gated channel 3 forms a non-selective ion transporter involved in germination and cation transport. J Exp Bot 57:791–800
Grotewold E, Chappell J, Kellogg EA (2015) The shifting genomic landscape in plant genes. In: Grotewold E, Chappell J, Kellogg EA (eds) Plant genomes and genetics. Wiley, Chichester, pp 17–44
Guo KM, Babourina O, Christopher DA, Borsics T, Rengel Z (2008) The cyclic nucleotide-gated channel, AtCNGC10, influences salt tolerance in Arabidopsis. Physiol Plant 134:499–507
Hartje S, Zimmermann S, Klonus D, Mueller-Roeber B (2000) Functional characterisation of LKT1, a K + uptake channel from tomato root hairs, and comparison with the closely related potato inwardly rectifying K + channel SKT1 after expression in Xenopus oocytes. Planta 210:723–731
Hogh-Jensen H, Pedersen MB (2003) Morphological plasticity by crop plants and their potassium use efficiency. J Plant Nutr 26:969–984
Hong JP, Takeshi Y, Kondou Y, Schachtman DP, Matsui M, Shin R (2013) Identification and characterization of transcription factors regulating Arabidopsis HAK5. Plant Cell Physiol 54:1478–1490
Hubisz MJ, Falush D, Stephens M, Pritchard JK (2009) Inferring weak population structure with the assistance of sample group information. Mol Ecol Resour 9:1322–1332
Jung JY, Shin R, Schachtman DP (2009) Ethylene mediates response and tolerance to potassium deprivation in Arabidopsis. Plant Cell 21:607–621
Kellermeier F, Chardon F, Amtmann A (2013) Natural variation of Arabidopsis root architecture reveals complementing adaptive strategies to potassium starvation. Plant Physiol 161:1421–1432
Kochian LV, Lucas WJ (1988) Potassium transport in roots. Adv Bot Res 15:136–151
Kong FM, Guo Y, Liang X, Wu CH, Wang YY, Zhao Y, Li SS (2013) Potassium (K) effects and QTL mapping for K efficiency traits at seedling and adult stages in wheat. Plant Soil 373:877–892
Korte A, Farlow A (2013) The advantages and limitations of trait analysis with GWAS: a review. Plant Methods 9:29
Lacombe B, Pilot G, Michard E, Gaymard F, Sentenac H, Thibaud JB (2000) A shaker-like K+ channel with weak rectification is expressed in both source and sink phloem tissues of Arabidopsis. Plant Cell 12:837–851
Leng Q, Mercier RW, Hua B-G, Fromm H, Berkowitz GA (2002) Electrophysiological analysis of cloned cyclic nucleotide-gated ion channels. Plant Physiol 128:400–410
Li XL, Borsics T, Harrington HM, Christopher DA (2005) Arabidopsis AtCNGC10 rescues potassium channel mutants of E. coli, yeast and Arabidopsis and is regulated by calcium/calmodulin and cyclic GMP in E. coli. Funct Plant Biol 7:643–653
Lin HX, Zhu MZ, Yano M, Gao JP, Liang ZW, Su WA, Hu XH, Ren ZH, Chao DY (2004) QTLs for Na+ and K+ uptake of the shoots and roots controlling rice salt tolerance. Theor Appl Genet 108:253–260
Liu K, Li L, Luan S (2006) Intracellular K+ sensing of SKOR, a Shaker-type K+ channel from Arabidopsis. Plant J 46:260–268
Lu YX, Chanroj S, Zulkifli L, Johnson MA, Uozumi N, Cheung A, Sze H (2011) Pollen tubes lacking a pair of K+ transporters fail to target ovules in Arabidopsis. Plant Cell 23:81–93
Marten I, Hoth S, Deeken R, Ache P, Ketchum KA, Hoshi T, Hedrich R (1999) AKT3, a phloem-localized K+ channel, is blocked by protons. Proc Natl Acad Sci USA 96:7581–7586
Nieves-Cordones M, Miller AJ, Aleman F, Martinez V, Rubio F (2008) A putative role for the plasma membrane potential in the control of the expression of the gene encoding the tomato high-affinity potassium transporter HAK5. Plant Mol Biol 68:521–532
Padmanaban S, Chnroj S, Kwak JM, Li X, Ward J et al (2007) Participation of endomembrane cation/H+ exchanger AtCHX20 in osmoregulation of guard cells. Plant Physiol 44:82–93
Palmgren MG (2001) Plant plasma membrane H+-ATPases: powerhouses for nutrient uptake. Annu Rev Plant Physiol Plant Mol Biol 52:817–845
Pandit A, Rai V, Bal S, Sinha S, Kumar V, Chauhan M, Gautam RK, Singh R, Sharma PC, Singh AK, Gaikwad K, Sharma TR, Mohapatra T, Singh NK (2010) Combining QTL mapping and transcriptome profiling of bulked RILs for identification of functional polymorphism for salt tolerance genes in rice (Oryza sativa L.). Mol Genet Genomics 284:121–136
Patishtan J, Hartley TN, Fonseca de Carvalho R, Maathuis FJM (2018) Genome-wide association studies to identify rice salt-tolerance markers. Plant Cell Environ 41(5):970–982
Pettersson S, Jensén P (1983) Variation among species and varieties in uptake and utilization of potassium. Plant Soil 72:231–237
Philippar K, Fuchs I, Lüthen H, Hoth S, Bauer CS, Haga K, Thiel G, Ljung K, Sandberg G, Böttger M, Becker D, Hedrich R (1999) Auxin-induced K+ channel expression represents an essential step in coleoptile growth and gravitropism. Proc Natl Acad Sci USA 96:12186–12191
Prinzenberg AE, Barbier H, Salt DE, Stich B, Reymond M (2010) Relationships between growth, growth response to nutrient supply, and ion content using a recombinant inbred line population in Arabidopsis. Plant Physiol 154:1361–1371
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
Pyo YJ, Gierth M, Schroeder JI, Cho MH (2010) High-affinity K+ transport in Arabidopsis: AtHAK5 and AKT1 are vital for seedling establishment and postgermination growth under low-potassium conditions. Plant Physiol 153:863–875
Qiu L, Ali S, Cai SG, Dai F, Jin XL, Wu FB, Zhang GP (2011) Evaluation of salinity tolerance and analysis of allelic function of HvHKT1 and HvHKT2 in Tibetan wild barley. Theor Appl Genet 122:695–703
Rengel Z, Damon PM (2008) Crops and genotypes differ in efficiency of potassium uptake and use. Physiol Plant 133:624–636
Römheld V, Kirkby EA (2010) Research on potassium in agriculture: needs and prospects. Plant Soil 335:155–180
Russell J, Booth A, Fuller J, Harrower B, Hedley P, Machray G, Powell W (2004) A comparison of sequence-based polymorphism and haplotype content in transcribed and anonymous regions of the barley genome. Genome 47:389–398
Shin R (2014) Strategies for improving potassium use efficiency in plants. Mol Cells 37:575–584
Shin R, Schachtman DP (2004) Hydrogen peroxide mediates plant root cell response to nutrient deprivation. Proc Natl Acad Sci USA 101:8827–8832
Si L, Chen J, Huang X, Gong H et al (2016) OsSPL13 controls grain size in cultivated rice. Nat Genet 48:447–456
Song CP, Guo Y, Qiu Q, Lambert G, Galbraith DW, Jagendorf A, Zhu JK (2004) A probable Na+(K+)/H+ exchanger on the chloroplast envelope functions in pH homeostasis and chloroplast development in Arabidopsis thaliana. Proc Natl Acad Sci USA 101:10211–10216
Spalding EP, Hirsch RE, Lewis DR, Qi Z, Sussman MR (1999) Potassium uptake supporting plant growth in the absence of AKT1 channel activity. J Gen Physiol 113:909–918
Tom NH, Alice ST, Frans JMM (2019) A role for the OsHKT 2; 1 sodium transporter in potassium use efficiency in rice. J Exp Bot. https://doi.org/10.1093/jxb/erz113
Venema K, Belver A, Marin-Manzano MC, Rodriguez- Rosales MP, Donaire JP (2003) A novel intracellular K+/H+ antiporter related to Na+/H+ antiporters is important for K+ ion homeostasis in plants. J Biol Chem 278:22453–22459
Wang Y, Wu WH (2013) Potassium Transport and Signaling in Higher Plants. Annu Rev Plant Biol 64:451–476
Wu P, Ni JJ, Luo AC (1998) QTLs underlying rice tolerance to low-potassium stress in rice seedlings. Crop Sci 38:1458–1462
Wu DZ, Sato K, Ma JF (2015) Genome-wide association mapping of cadmium accumulation in different organs of barley. New Phytol 208:817–829
Ye ZL, Zeng JB, Li X, Zeng FR, Zhang GP (2017) Physiological characterizations of three barley genotypes in response to low potassium stress. Acta Physiol Plant 39:232
Yokoi S, Quintero FJ, Cubero B, Ruiz MT, Bressan RA, Hasegawa PM, Pardo JM (2002) Differential expression and function of Arabidopsis thaliana NHX Na+/H+ antiporters in the salt stress response. Plant J 30:529–539
Yu J, Pressoir G, Briggs WH, Vroh Bi I, Yamasaki M, Doebley JF, McMullen MD, Gaut BS, Nielsen DM, Holland JB, Kresovich S, Buckler ES (2006) A unified mixed-model method for association mapping that accounts for multiple levels of relatedness. Nat Genet 38:203–208
Zhang G, Chen J, Eshetu AT (1999) Genotypic variation for potassium uptake and utilization efficiency in wheat. Nutr Cycl Agroecosyst 54:41–48
Zhao J, Cheng NH, Motes CM, Blancaflor EB, Moore M et al (2008) AtCHX13 is a plasma membrane K+ transporter. Plant Physiol 148:796–807
Zhao J, Sun HY, Dai HX, Zhang GP, Wu FB (2010) Difference in response to drought stress among Tibet wild barley genotypes. Euphytica 172:395–403
Zhao Y, Li X, Zhang S, Wang J, Yang X, Tian J, Hai Y, Yang X (2014) Mapping QTLs for potassium-deficiency tolerance at the seedling stage in wheat (Triticum aestivum L.). Euphytica 198:185–198
Acknowledgements
We thank Prof. Dongfa Sun (Huazhong Agricultural University, China) for providing the Tibetan wild barley accessions.
Funding
This study was supported by National Natural Science Foundation of China (31620103912 by Guoping Zhang, 31771685 by Jianbin Zeng), China Agriculture Research System (CARS-05 by Guoping Zhang) and Jiangsu Collaborative Innovation Center for Modern Crop Production (JCIC-MCP by Guoping Zhang). These funding bodies provided the financial support in carrying out the experiments, sample and data analysis, and MS writing.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by ZY, JZ, LY, LL and GZ. The first draft of the manuscript was written by ZY, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary material 1: Table S1
All phenotypic traits data of each wild barley accessions (XLSX 96 kb)
Supplementary material 2: Table S2
The definitions and abbreviations of phenotypic traits (XLSX 13 kb)
Supplementary material 3: Table S3
The Q matrix of 179 barley accessions (XLSX 15 kb)
Supplementary material 4: Table S4
The kinship matrix of 179 barley accessions (XLSX 383 kb)
Supplementary material 5: Table S5–S8
Table S5–S8 (DOCX 22 kb)
Supplementary material 6: Table S9
SNPs and candidate gene from GWAS in KUR and KTR (XLSX 15 kb)
Supplementary material 7: Table S10
Outline of 32 SNPs relative information (XLSX 22 kb)
Supplementary material 8: Table S11
Summary of three genes from KUR and KTR (XLSX 12 kb)
Supplementary material 9: Table S12
Transcripts of three genes of Hordeum vulgare L. (XLSX 13 kb)
Supplementary material 10: Table S13
Amino acid sequences of three gene of Hordeum vulgare L. (XLSX 14 kb)
Supplementary material 11: Table S14
Summary of AP2-like ethylene-responsive transcription factors participated in ethylene biosynthesis (DOCX 16 kb)
Supplementary material 12: Figure S1–S5
Figure S1–S5 (PPTX 328 kb)
Rights and permissions
About this article
Cite this article
Ye, Z., Zeng, J., Ye, L. et al. Genome-wide association analysis of potassium uptake and translocation rates under low K stress in Tibetan wild barley. Euphytica 216, 35 (2020). https://doi.org/10.1007/s10681-020-2556-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10681-020-2556-5