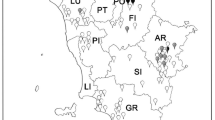Abstract
Sharka, caused by plum pox virus (PPV), is by far the most harmful disease of stone fruits (genus Prunus). Although Italy is a major producer of stone fruits, and sharka has spread in most Italian regions since its first identification in the 1970s, information on the diffusion of the disease in Italy results still scattered and incomplete. To address this knowledge gap, the present review aimed to firstly provide a historical overview of sharka in Italy, summarizing outcomes on the disease achieved since the first identification of its causal agent PPV. Data retrieved from a variety of sources (i.e., published literature, official reports and personal communications) showed that (i) sharka spread across almost the whole Italian peninsula as only Liguria, Umbria and Aosta Valley regions officially result disease-free; (ii) the regions with higher rates of infected districts are not always those more dedicated to the production of stone fruits; (iii) only the three major PPV strains causing sharka, i.e., PPV-Dideron (D), PPV-Marcus (M), and PPV-Recombinant (Rec), have been reported in Italy; and (iv) only apricot, peach and plum (i.e., widespread and economically relevant species) are hosts of PPV, with different scenarios among regions. A focus on Tuscany (Central Italy) showed that, (i) sharka has been reported since the 1990s, but extensive PPV monitoring campaigns were run from 2013 to 2021, especially in nurseries; (ii) higher number of symptomatic and PPV positive samples were recorded in farms, mostly in the Arezzo and Florence districts; and (iii) only PPV-M and PPV-Rec strains seem still present. We believe that the present overview will benefit and encourage both researchers for future investigations on sharka and farmers that have to manage the disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sharka, also known as ‘Plum pox’, is by far the most harmful viral disease of stone fruits worldwide. It is caused by plum pox virus (PPV), a potyvirus in the Potyviridae family, which infects major stone fruit trees of the genus Prunus (Rosaceae family), including apricot (P. armeniaca), peach (P. persica), plum (P. domestica) and cherry (P. avium; García et al., 2014; Sheveleva et al., 2018), as well as other ornamental and wild Prunus species (James & Thompson, 2006; Sebestyen et al., 2008). PPV spread at local scale is mainly caused by aphids (e.g., Myzus persicae, Phorodon humuli and Hyalopterus pruni; Isac et al, 1998), and common herbaceous species (e.g., Bellis perennis, Clematis sp., Convolvulus arvensis, Matricaria chamomilla, Taraxacum officinalis, and Trifolium sp.) may represent important secondary sources of this disease in orchards (Llácer, 2006; Manachini et al., 2007; Moreno et al., 2009). Differently, the long-distance diffusion of PPV has been due to global trade of infected plant material (García et al., 2014).
Identified for the first time in 1918 in Bulgaria (Atanasoff, 1932), PPV is currently widespread in Europe, the Middle East, North and South America, and Asia (EPPO, 2022). The worldwide PPV genetic characterization identified ten strains (Chirkov et al., 2018; James et al., 2013): PPV-Marcus (M), PPV-Dideron (D), PPV-Recombinant (Rec), PPV-El Amar (EA), PPV-Cherry (C), PPV-Winona (W), PPV-Turkey (T), PPV-Cherry Russian (CR), PPV-Ancestor (An), and the recent PPV-Cherry Volga (CV). PPV-M, PPV-D and PPV-Rec have been most widely reported, with PPV-M as the most aggressive virus which is efficiently transmitted by aphids, whereas PPV-D being the most widely distributed, and PPV-Rec (efficiently transmitted by aphids as well) being the recombinant product of PPV-M with PPV-D (García et al, 2014; Sihelská et al., 2017).
Symptoms of sharka may appear on leaves, shoots, barks, petals, fruits and stones (apricot and only a few sensitive plum cultivars; Fig. 1). They are clearly visible on leaves early in the growing season and include mild light-green discoloration, chlorotic spots, bands or rings, vein clearing or yellowing and leaf deformation. Flower symptoms can occur on petals (discoloration), especially in peach cultivars with pink flowers. Infected fruits show chlorotic spots or lightly pigmented yellow rings or line patterns (especially in Japanese plums and peaches). They may become deformed or show irregular shape (especially cherries and plums), and over time may develop brown or necrotic areas under the discolored rings. Furthermore, premature fruit drop was observed in apricot, European plum, sweet and sour cherry. Fruits of early maturing cultivars of all susceptible species usually show more severe symptoms than those of late maturing ones (Revers & García, 2015; Sochor et al., 2012). Thus, although sharka poses no danger to consumers, it can make stone fruits unmarketable.
Symptoms induced by plum pox virus on (i) apricot leaves (A), fruits (B) and stone (C); (ii) peach leaves (D), fruits (E) and petals (F); and (iii) plum leaves (G) and fruit (H); and premature plum fruit drop (I; EPPO, 2023)
As infected trees cannot be treated, but only removed and destroyed, the management of the disease is possible only by prevention, using PPV-free propagation material, contrast to PPV vectors, and field monitoring for PPV presence (García et al., 2014; Labonne & Dallot, 2006; Llácer, 2006). Actually, visual symptoms are not always a reliable indicator of disease as they can occur after several months or years after the tree has been infected, and other viruses can enhance the symptoms (e.g., pseudosharka; Wilhelm & Kunze, 1995). Thus, PPV can be more reliably detected by laboratory analysis of tissue samples or by grafting test material onto highly susceptible hosts and monitoring for the development of symptoms. The impact of sharka disease over 30 years was thus estimated to be over 10,000 million euros, taking into account the economic losses in production and the costs of sanitary control and surveillance, and of eradication programmes run in affected countries worldwide (Cambra et al., 2006a; Rubio et al., 2017).
For these reasons, PPV was ranked as quarantine pest in European Union since 2000 (European Council, 2000), although in 2019 it was regraded to ‘regulated non-quarantine pest’ (RNQP) due to its widespread endemic presence (European Union, 2019). However, the attention paid to PPV challenging stone fruits is still tremendous, as demonstrated by the number of studies and reviews focused on this topic (De Mori et al., 2020; Espinoza et al., 2021; Gürcan et al., 2021; Kil et al., 2021; Maejima et al., 2020; Mori et al., 2021; Okić et al., 2022; Rodamilans et al, 2020; Tamukong et al., 2020).
Europe is a major producer of stone fruits with around 8 million tons produced annually, mostly peaches, plums, cherries and apricots, and Italy is the most ardent contributor, together with Spain. Stone fruit production is of economic importance in Italy, especially of the cultivation of peaches, apricots, plums and cherries (around 1 million, 200,000, 140,000 and 90,000 tons produced in 2021, respectively; FAOSTAT, 2021). However, Italian stone fruit production (as well as other analogous markets worldwide) is continually challenged by sharka, which was firstly identified in Italy in the 1970s (Alber et al., 1974), and has been reported in most Italian regions (Armentano & Bertanza, 2018; Bianco et al., 2005). In Italy, the first regulation about sharka control was established in 1996, and it was based on field monitoring, eradication of infected plants, obligation to use certified propagation material in new orchards, multiplication of only healthy plant material, and destruction of infected mother plants in the nursery (Italian Republic, 1996). In accordance with the European regulation, a new decree was adopted in 2009, which introduced a subdivision among settlement (i.e., infected area where the disease was no longer eradicable), outbreak (i.e., infected area where diseased plants had to be removed, and plants could not be recovered for multiplication), buffer (i.e., not infected area, but close to outbreak and/or settlement zones) and free zones (i.e., not infected area; Italian Republic, 2009). Due to the impossibility of PPV eradication in several Italian areas, some deregulations occurred in 2016 (e.g., reduction of the buffer zone radius from 1 km to 300 m around an infected plant; Italian Republic, 2016) and the decree was finally abrogated in 2021 (Italian Republic, 2021), following the abovementioned PPV downregulation to RNQP by the European Union.
Although several studies and monitoring activities of sharka and its causal agent PPV have been carried out in the last decades, information on the diffusion of the disease in Italy is still scattered and incomplete (e.g., data from specific regions are missing or unavailable). To address this knowledge gap, the present review aimed to firstly provide a historical overview of sharka in Italy, summarizing outcomes on the disease and its causal agent PPV occurred around fifty years ago, also with a further focus on Tuscany (Central Italy). We anticipate that outcomes here reported will be useful for the understanding of genetic diversity and diffusion of sharka in Italy, thus encouraging other research in this area and supporting the challenging contrast to this detrimental disease.
Data recovery
A database reporting peer-reviewed and official information about the diffusion of sharka and its causal agent PPV in Italy was created on May 2023 examining (i) published peer-reviewed literature, (ii) regional laws concerning sharka, reports of sharka monitoring activities and/or informative materials published in websites of regional phytosanitary services (RPS) and other institutions (26 online resources and databases were used, list provided in Supplementary Table S1), and (iii) personal communications obtained from reliable institutions (Table S1).
The published peer-reviewed literature was searched in the Web of Science (Thompson-ISI, Philadelphia, PA, USA), Scopus (Elsevier, Amsterdam, Netherlands), Science Direct (Elsevier) and Google Scholar (Google, Mountain View, CA USA) databases (see websites in Table S1), using multiple combinations of “sharka”, “vaiolatura delle drupacee” (i.e., the common Italian name of sharka), “Plum pox virus”, “PPV”, “PPV-M”, “PPV-Rec”, “PPV-D”, “Prunus”, “peach”, “apricot”, plum” and “Italy” as keywords. Several peer-reviewed papers were dated and not available in digital format, so they were retrieved by the library service of the Department of Agriculture, Food and Environment of the University of Pisa (using also intra-university book lending). The reference lists of any article identified by this literature search were cross-checked in order to include any other relevant reference, finally identifying 28 research papers (Table 1). These articles were used together with 45 official documents and 15 personal communications (Supplementary Table S1).
Recovered data were screened to get the following information for each Italian region: (i) year of the first identification of sharka, (ii) number of districts affected by sharka, (iii) PPV strains reported by enzyme-linked immunosorbent assay (ELISA) or polymerase chain reaction (PCR) methods, and (iv) Prunus hosts of PPV. Furthermore, given the long-lasting collaboration between the Department of Agriculture, Food and Environment (DAFE) of the University of Pisa (i.e., Author institution) and the Regional Phytosanitary Service of Tuscany (RPS-T) more information was recovered for this Region, namely (i) the sites investigated during the PPV monitoring campaigns, (ii) the samples positive to PPV, and (iii) the identified PPV strains.
Sharka in Italy
Sharka spread across almost the whole Italian peninsula, and the regions with higher rates of infected districts are not always those more dedicated to the production of stone fruits
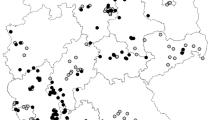
The history of sharka in Italy began about 50 years ago (i.e., 1970s) in north-eastern regions, with the identifications by Alber et al. (1974) in Alto Adige and a few years later by Canova et al. (1977) in nearby Trentino (Fig. 2a). In the 1980s, the disease was then randomly recovered in other northern regions (Emilia-Romagna and Piedmont; Conti et al., 1984; Giunchedi et al., 1983), and later also in central (Lazio; Barba et al., 1985) and southern regions (Apulia, Basilicata and Sicily; Davino et al., 1986; Di Terlizzi et al., 1988). Similarly, in the 1990s, sharka was firstly reported in a further six regions erratically scattered along the country as (following a chronologic order) it was identified in Calabria (Southern Italy; Polizzi & Agosteo, 1992), Abruzzo (Central Italy; Vicchi & Babini, 2008), Tuscany (Central Italy; Ginanni et al., 1993), Friuli-Venezia Giulia (Northern Italy; Pasquini & Barba, 1994), Lombardy (Northern Italy; Vicchi & Babini, 2008), Veneto (Northern Italy; Poggi Pollini et al., 1996) and Campania (Southern Italy; Carella et al., 2002). At the beginning of the new millennium, sharka was also reported in the Marche region (Central Italy; Marche Region, 2018), whereas the most recent first report was by Palmisano et al. (2011) in Molise (Central Italy). The presence of sharka was confirmed also in Sardinia, but no data were retrieved about the year of introduction (Sardinia Region, 2016). Thus, sharka has never been reported in Liguria, Umbria and Aosta Valley (Personal communications), probably due to the scarcity of stone fruit orchards in these regions (ISTAT, 2021). Overall, these outcomes clearly show that sharka spread across almost the whole Italian peninsula from its first identification around 50 years ago, but its scattered diffusion (as well as its presence reported in island regions) suggests that its dissemination occurred not only because of aphid transmission but more importantly by accidental movement of infected plant material.
All districts of Emilia-Romagna (i.e., the region with the second higher production of stone fruits: about 200,000 tons produced annually vs about 71,000 tons as average among Italian regions; ISTAT, 2021) and the Trentino-Alto Adige regions were affected by sharka, as well as 60% of the districts of Campania (i.e., the region with the highest production of stone fruits: about 511,000 tons; ISTAT, 2021). The disease was observed in half of the districts of the Basilicata (i.e., the region with the fourth higher production of stone fruits: 110,000 tons; ISTAT, 2021), Friuli-Venezia Giulia, Apulia and Molise. Lower rates of sharka presence were reported for the other regions, including Sicily (i.e., the region with the third higher production of stone fruits: 140,000 tons; ISTAT, 2021; Fig. 2b). These data indicate that the regions with a larger fraction of infected districts are not always those more dedicated to the production of stone fruits. Evidently, a reliable interpretation of these results has to consider also the extension, district numbers and the production of each region. For example, comparing Campania and Trentino-Alto Adige which have approximately the same territorial area (around 13,600 km2), Campania showed a small fraction of affected districts compared to Trentino-Alto Adige (60 vs 100%). The impact of sharka is larger in Campania being the major stone fruit producer in Italy, while the production in Trentino-Alto Adige is lower (about 4,300 tons produced annually; ISTAT, 2021) as this region is more dedicated to other fruits (i.e., apples, pears and grapevine).
Major PPV strains, i.e., PPV-D, PPV-M and PPV-Rec, have been reported in apricot, peach and plum
Only the three major PPV strains causing sharka, i.e., PPV-D, PPV-M and PPV-Rec, have been reported in Italy (actually, the PPV-C strain was identified once in Apulia in a single sweet cherry tree which was immediately eradicated, and PPV-C was never found again; Crescenzi et al., 1994). The occurrence of the PPV-D, PPV-M and PPV-Rec strains is in accordance with other European regions (Cambra et al., 2006b; Kamenova & Borisova, 2019; Vidal et al., 2020; Zagrai et al., 2009; Fig. 3a). Taking into account the observed presence of these strains, Italian regions could be divided into four groups: (i) Calabria and Lazio with only PPV-D reported, (ii) Abruzzo and Molise with only PPV-M, (iii) Basilicata, Friuli-Venezia-Giulia, Lombardy, Marche, Piedmont, Sicily, Trentino-Alto Adige and Veneto with both PPV-D and PPV-M, and (iv) Campania, Emilia-Romagna, Tuscany and Apulia where all the three strains PPV-D, PPV-M and PPV-Rec have been reported. Thus, PPV-M and PPV-D were the most reported (mostly together) with 14 regions involved, whereas PPV-Rec was observed only in four regions. Actually, the low presence of PPV-Rec could be due to a misidentification of this strain with PPV-M because PPV-Rec was recognized only in 2004 as product of a recombinant event between PPV-D and PPV-M (Glasa et al., 2004). Moreover, the coat proteins targeted by several diagnostic methods are similar for the PPV-M and PPV Rec strains often leading to a wrong diagnosis, especially in the past (James et al., 2013).
PPV was reported only in apricot, peach and plum (other than the single identification in sweet cherry reported above; Crescenzi et al., 1994). All three species were reported as PPV hosts in most of the regions (Fig. 3b), except for Calabria (only apricot), Molise and Sardinia (only peach), Abruzzo and Sicily (only apricot and peach), and Apulia and Lazio (only apricot and plum). Thus, all three species were reported as PPV hosts mostly in northern regions, whereas a larger hostspecificity was observed in central and southern ones. Overall, these outcomes were expected as apricot, peach and plum are widespread and economically relevant species, and for these reasons have been monitored intensively (Umer et al., 2019). However, it is important to highlight that other minor commercial (e.g., almond), ornamental (e.g., cherry laurel) and spontaneous (e.g., blackthorn) Prunus species (García et al., 2014) reported as PPV hosts (and inoculum sources) have not been investigated so far, and further research should be conducted to elucidate their role in the diffusion of sharka in Italy.
A focus on Tuscany
Sharka has been reported in Tuscany since the 1990s, and extensive PPV monitoring campaigns were run from 2013 to 2021, especially in nurseries
Tuscany is one of the major regions of Central Italy in terms of stone fruit production (ISTAT, 2021). As reported above, sharka was here firstly identified in the early 1990s on apricot and plum (Ginanni et al., 1993), but it became a serious threat only with a new outbreak in 2013, when RPS-T reported the disease on plums growing in commercial orchards and a nursery (Tuscany Region, 2013). As a consequence, according to the national legislation (Italian Republic, 2009), a specific Tuscany regional decree aimed to implement sharka monitoring and management methods was established in 2014 (Tuscany Region, 2014) and updated in 2019 (Tuscany Region, 2019), although it was abrogated in 2021 with the Italian reception of EU regulation which downregulated PPV to RNQP (Italian Republic, 2021).
From 2013 to 2021, RPS-T (in collaboration with DAFE) performed sharka monitoring, extensively surveying a total of 1,596 sites characterized by the presence of commercial apricots, peaches, plums, sweet cherries, ornamental myrabolan plums and wild blackthorns. Nurseries were mostly inspected (n = 1217), followed by farms (n = 327) and non-agricultural areas (n = 52; Fig. 4a). The number of surveyed sites increased every year due to the increasing number of monitored nurseries. The number of farms and non-agricultural sites did not change much. Actually, Tuscany is characterized by an ancient nursery tradition, with this business being not only an Italian, but also a European excellence, at both quantitative and qualitative levels, especially for ornamental plants (Lazzerini et al., 2016). This feature, together with the fact that nurseries can play a key role in the spread of plant diseases, being departure and arrival hubs of plant material marketed all around the world (Alonso Chavez et al., 2016; Pisuttu et al., 2023), explains why PPV monitoring was mostly focused in nurseries. During these RPS-T/DAFE surveys, sharka-like symptoms were observed on a total of 5,240 plants (which were collected for diagnostic analyses), among which 3,070, 1,740, and 430 samples were recovered in farms, nurseries and non-agricultural areas, respectively. Peaks in collected samples (n = ca. 700–800) occurred in 2016, 2017, and 2020, whereas lowest amount was sampled in 2013 (n < 200), so showing an irregular trend over the years (Fig. 4b).
Higher number of symptomatic and PPV positive samples were recorded in farms, mostly in Arezzo and Florence districts, and only PPV-M and PPV-Rec strains seem still present in Tuscany
Overall, around 4% of the collected samples tested positive for sharka, with higher numbers of infected samples reported in 2014 and 2018 (n = 41 and 37, respectively), and lowest in 2017 (n = 7), thus not showing a clear trend over the years (Fig. 5). As expected, most of the positive samples were collected in farms (n = 211), as only six and two positive samples were from nurseries and non-agricultural areas, respectively, thus showing no relation between the number of collected samples and those testing positive (this feature being true also taking into account the years of monitoring). The larger number of positive samples in farms was likely due to more favourable conditions for the spread of the disease (e.g., vectors, environmental conditions) and less survey activities, compared to nurseries (although the large number of potential hosts).
Sharka was reported only in six Tuscan districts, mostly and continuously in Arezzo and Florence, whereas more discontinuously in Grosseto and Pistoia, and only once in Lucca and Siena (Fig. 6). Other than the four districts reported above for identifications in farms (Fig. 2b), identifications in nurseries occurred in Pistoia and Siena districts, where no positive samples were reported in other area types. These outcomes showed that most affected districts were those with a higher density of stone fruit farms (ISTAT, 2021).
Although only PPV-D strain was reported in the whole Italy until 1996 (Poggi Pollini et al., 1996), a genetic characterization of PPV strains isolated from stone fruits (apricot, peach, plum) of Tuscany collected in 2020 and 2021 allowed to identify also PPV-M, mostly in peach and less in plum, whereas PPV-Rec was identified only in plum (Pedrelli et al., 2023). This divergence among PPV strain/host relations was already reported (Sihelská et al., 2017), and the lack of PPV-D identification could be due to a PPV-M and PPV Rec more efficient transmission by vectors and a best adaptation in hosts (James et al., 2013).
Conclusions
According to the European regulation that ranked PPV as quarantine pest, sharka was subjected to a national monitoring and eradication programme until 2021, whereas nowadays, with the new European regulation that downregulated PPV to RNQP due to its endemicity, the management of sharka in Italy is demanded of farmers. However, information collected in the last decades on the diffusion of sharka in Italy show scattered results, incomplete and difficult to interprete. This review summarized the the development and spread since the first identification of its causal agent occurred around fifty years ago and summarised the monitoring activity of the last decade in Tuscany. Data retrieved from a variety of sources (e.g., published literature, official documents and personal communications) showed that (i) sharka spread across almost the whole Italian peninsula as only the Liguria, Umbria and Aosta Valley regions remain disease-free, (ii) the regions with higher fractions of infected districts are not always those more dedicated to the production of stone fruits; (iii) only the three major PPV strains causing sharka, i.e., PPV-D, PPV-M and PPV-Rec, have been reported in Italy; and (iv) only apricot, peach and plum (i.e., widespread and economically relevant species) were reported as PPV hosts in Italy (with different rates of occurrence among regions). In addition, the analysis of the data from Tuscany showed that (i) sharka has been reported since the 1990s, but extensive PPV monitoring campaigns were run from 2013 to 2021, especially in nurseries; (ii) higher number of symptomatic and PPV positive samples were recorded in farms, mostly in the Arezzo and Florence districts, important stone fruit growing regions, and (iii) only PPV-M and PPV-Rec strains seem still present. We believe that the present overview will benefit both researchers for future investigations on sharka and farmers that have to manage the disease.
Data availability
The data that support the findings of this study are available in the article and its online supplementary material.
References
Alber, L., Willeit, H., & Stainer, R. (1974). Die Sharkakrankheit eine ernste Gefahr für die Marillen Bestände in Vinschgau. Obstbau Weinbau, 11, 314–315.
Alonso Chavez, V., Parnell, S., & Van Den Bosch, F. (2016). Monitoring invasive pathogens in plant nurseries for early-detection and to minimise the probability of escape. Journal of Theoretical Biology, 407, 290–302. https://doi.org/10.1016/j.jtbi.2016.07.041
Armentano, G., & Bertanza, P. (2018). Sharka, un problema non risolto. Speciale Difesa Drupacee. L’informatore Agrario, 9, 45–47.
Atanasoff, D. (1932). Plum pox. A new virus disease. Annals of the University of Sofia, Faculty of Agriculture and Silviculture, 11, 49–69.
Barba, M., Simeone, M., & Quacquerelli, A. (1985). Indagine sul virus della «sharka» nel Lazio. Rivista Di Ortoflorofrutticoltura Italiana, 69, 145–151.
Bianco, P. A., Fanigliulo, A., Comes, S., Casati, P., Crescenzi, A., & Belli, G. (2005). Characterization of plum pox virus (PPV) isolates associeted with sharka infection in Northern and Southern Italy. Phytopathologia Polonica, 36, 17–24.
Bonfanti, R. (2010). Il plum pox virus (o sharka), agente della vaiolatura delle drupacee. L’informatore Agricolo-L’informateur Agricole, 4, 28–29.
Cambra, M., Capote, N., Cambra, M. A., Llácer, G., Botella, P., & López-Quílez, A. (2006a). Epidemiology of sharka disease in Spain. EPPO Bulletin, 36(2), 271–275. https://doi.org/10.1111/j.1365-2338.2006.00986.x
Cambra, M., Capote, N., Myrta, A., & Llácer, G. (2006b). Plum pox virus and the estimated costs associated with Sharka disease. EPPO Bulletin, 36, 202–204. https://doi.org/10.1111/j.13652338.2006.01027.x
Camele, I., Nuzzacci, N., Avolino, A., Rana, G. L., Lafortezza, R., Marcone, C., & Palumbo, M. (1990). La “Saharka” (=Vaiolatura) delle drupacee su albicocco in Basilicata. L’informatore Agrario, 46, 65–67.
Canova, A., Giunchedi, L., Credi, R., Albertini, A., & Nicolli, C. (1977). La vaiolatura ad anello (sharka o plum pox) su albicocco e susino in Italia. Frutticoltura, 6, 17–23.
Carella, D., Pezzella, M., & Spigno, P. (2002). La Sharka All’attacco. Campania Agricoltura, 3, 15–16.
Chirkov, S., Sheveleva, A., Ivanov, P., & Zakubanskiy, A. (2018). Analysis of genetic diversity of Russian sour cherry plum pox virus isolates provides evidence of a new strain. Plant Disease, 102, 569–575. https://doi.org/10.1094/PDIS-07-17-1104-RE
Conti, M. (1983). Problemi di quarantena vegetale relativi agli insetti vettori di virus e di altri agenti fitopatogeni. Informatore Fitopatologico, 2, 21–25.
Conti, M., Roggero, P., Mancini, G., Lenzi, R., & Accotto, G. P. (1984). Il virus della vaiolatura del susino (PPV) o ‘Sharka’ in Piemonte. Giornate Fitopatologiche, 1, 273–284.
Crescenzi, A., Nuzzaci, M., Levy, L., Piazzolla, P., & Hadidi, A. (1994). Infezioni di sharka su ciliegio dolce in Italia meridionale. Informatore Agrario, 50(34), 73–75.
Crescenzi, A., Nuzzaci, M., Levy, L., Piazzolla, P., & Hadidi, A. (1995). Plum pox virus (PPV) in sweet cherry. Acta Horticolturae, 386, 219–225.
Crescenzi, A., d’Aquino, L., Comes, S., Nuzzaci, M., Piazzolla, P., Boscia, D., & Hadidi, A. (1997a). Characterization of the sweet cherry isolate of plum pox potyvirus. Plant Disease, 81(7), 711–714. https://doi.org/10.1094/pdis.1997.81.7.711
Crescenzi, A., D'Aquino, L., Nuzzaci, M., Ostuni, A., Bavoso, A., Comes, S., De Stradis A., & Piazzolla, P. (1997b). Production of strain specific antibodies against a synthetic polypeptide corresponding to the N-terminal region of the plum pox potyvirus coat protein. 69(1–2), 181–189. https://doi.org/10.1016/s0166-0934(97)00158-4
Davino, M., Areddia, R., & Guo, Z. Y. (1986). Situazione della vaiolatura delle drupacee (Sharka) in Sicilia. Informatore Fitopatologico, 36(7–8), 43–45.
De Mori, G., Savazzini, F., & Geuna, F. (2020). Molecular tools to investigate sharka disease in Prunus species. Applied Plant Biotechnology for Improving Resistance to Biotic Stress, 203–223. https://doi.org/10.1016/b978-0-12-816030-5.00010-0
Di Terlizzi, B., Savino, V., Castellano, M. A., & Martelli, G. P. (1988). Presenza della vaiolatura delle drupacee in Puglia e Basilicata. Informatore Fitopatologico, 38(11), 53–55.
EPPO. (2022). Map with the distribution of the plum pox virus. https://gd.eppo.int/taxon/PPV000/distribution. Accessed 20 Mar 2022
EPPO. (2023). Plum pox virus symptoms photos on apricot, peach and plum. https://gd.eppo.int/taxon/PPV000/photos. Accessed 20 May 2023
Espinoza, C., Bascou, B., Calvayrac, C., & Bertrand, C. (2021). Deciphering Prunus responses to PPV Infection: A way toward the use of metabolomics approach for the diagnostic of sharka disease. Metabolites, 11(7), 465. https://doi.org/10.3390/metabo11070465
European Council. (2000). Protective measures against the introduction into the Community of organisms harmful to plants or plant products and against their spread within the Community. Directive 2000/29/EC 1, 1-112.
European Union. (2019). Regulations-Commission implementing regulation (EU) 2019/2072 of 28 November 2019 establishing uniform conditions for the implementation of Regulation (EU) 2016/2031 of the European Parliament and the Council, as regards protective measures against pests of plants, and repealing Commission Regulation (EC) No 690/2008 and amending Commission Implementing Regulation (EU) 2018/2019. Official Journal of the European Union., 1, 1–279.
FAOSTAT. (2021). Prunus fruit production in Italy. https://fenix.fao.org/faostat/internal/en/#home. Accessed 10 Dec 2021
Fontana, F., & Babini, A. R. (2014). Sharka delle drupacee: test sulle nuove cultivar. Agricoltura - Periodico Della Regione Emilia-Romagna, 10, 66–67.
Frisinghelli, C., Grando, M.S., & Vindimian, M.E. (1996). Individuazione di ceppi di plum pox virus D e M in Trentino. Proceedings in: Convegno annuale SIPaV, Udine 26-27 settembre 1996, C107.
García, J. A., Glasa, M., Cambra, M., & Candresse, T. (2014). Plum pox virus and sharka: A model potyvirus and a major disease. Molecular Plant Pathology, 15, 226–241. https://doi.org/10.1111/mpp.12083
Ginanni, M., Materazzi, A., Mainardi, M., & Triolo, E. (1993). La “vaiolatura” delle drupacee: Indagini sulla presenza in Toscana. Informatore Fitopatologico, 43, 58–62.
Giunchedi, L., Poggi Pollini, C., & Vicchi, V. (1983). Presenza della malattia nota come “vaiolatura” su susino e albicocco in Romagna. Informatore Fitopatologico, 33, 56–60.
Glasa, M., Palkovics, L., Komínek, P., Labonne, G., Pittnerová, S., Kúdela, O., Candresse, T., & Šubr, Z. (2004). Geographically and temporally distant natural recombinant isolates of plum pox virus (PPV) are genetically very similar and form a unique PPV subgroup. Journal of General Virology, 85(9), 2671–2681. https://doi.org/10.1099/vir.0.80206-0
Gürcan, K., Teber, S., Akbulut, M., & Çağlayan, K. (2021). Genetic diversity and a long evolutionary history of plum pox virus strain Rec in Turkey. European Journal of Plant Pathology, 161(2), 453–461. https://doi.org/10.1007/s10658-021-02336-2
Isac, M., Preda, S., & Marcu, M. (1998). Aphid species-vectors of plum pox virus. Acta Virologica, 42(4), 233–234.
ISTAT. (2021). Prunus fruit production in Italian regions. http://dati.istat.it/Index.aspx?QueryId=33705. Accessed 5 Nov 2021
Italian Republic. (1996). Lotta obbligatoria contro il virus della “Vaiolatura delle drupacee”. Decreto ministeriale 29 novembre 1996. Gazzetta Ufficiale Della Repubblica Italiana, 289, 26–27.
Italian Republic. (2009). Lotta obbligatoria per il controllo del virus plum pox virus (PPV), agente della «Vaiolatura delle drupacee» (Sharka). Gazzetta Ufficiale Della Repubblica Italiana, 235, 24–28.
Italian Republic. (2016). Modifiche al decreto 28 luglio 2009, recante: «Lotta obbligatoria per il controllo del virus plum pox virus (PPV), agente della “Vaiolatura delle drupacee” (Sharka). Gazzetta Ufficiale Della Repubblica Italiana, 81, 17–19.
Italian Republic. (2021). Abrogazione di provvedimenti recanti lotte obbligatorie e misure fitosanitarie nazionali. Gazzetta Ufficiale Della Repubblica Italiana, 3, 6–9.
James, D., & Thompson, D. (2006). Hosts and symptoms of plum pox virus: Ornamental and wild Prunus species. EPPO Bulletin, 36, 222–224. https://doi.org/10.1111/j.1365-2338.2006.00976.x
James, D., Varga, A., & Sanderson, D. (2013). Genetic diversity of plum pox virus: Strains, disease and related challenges for control. Canadian Journal of Plant Pathology, 35, 431–441. https://doi.org/10.1080/07060661.2013.828100
Kamenova, I., & Borisova, A. (2019). Update on distribution and genetic variability of plum pox virus strains in Bulgaria. The Plant Pathology Journal, 35, 243–256. https://doi.org/10.5423/ppj.oa.09.2018.0189
Kil, E.-J., Ho, P. T., Fadhila, C., Lal, A., Vo, T. T. B., Kim, M., & Lee, S. (2021). Plum pox virus: Diagnosis and spread inhibition by weed control. Journal of Plant Diseases and Protection, 128(4), 1091–1099. https://doi.org/10.1007/s41348-021-00481-8
Labonne, G., & Dallot, S. (2006). Epidemiology of sharka disease in France. EPPO Bulletin, 36, 267–270. https://doi.org/10.1111/j.1365-2338.2006.00985.x
Lazzerini, G., Lucchetti, S., & Nicese, F. P. (2016). Green House Gases (GHG) emissions from the ornamental plant nursery industry: A Life Cycle Assessment (LCA) approach in a nursery district in central Italy. Journal of Cleaner Production, 112, 4022–4030. https://doi.org/10.1016/j.jclepro.2015.08.065
Llácer, G. (2006). Hosts and symptoms of plum pox virus: Herbaceous hosts. EPPO Bulletin, 36, 227–228. https://doi.org/10.1111/j.1365-2338.2006.00985.x
Maejima, K., Hashimoto, M., Hagiwara-Komoda, Y., Miyazaki, A., Nishikawa, M., Tokuda, R., Kumita, K., Maruyama, N., Namba, S., & Yamaji, Y. (2020). Intra-strain biological and epidemiological characterization of plum pox virus. Molecular Plant Pathology, 21(4), 475–488. https://doi.org/10.1111/mpp.12908
Manachini, B., Casati, P., Cinanni, L., & Bianco, P. (2007). Role of Myzus persicae (Hemiptera: Aphididae) and its secondary hosts in plum pox virus propagation. Journal of Economic Entomology, 100(4), 1047–1052. https://doi.org/10.1603/0022-0493(2007)100[1047:rompha]2.0.co;2
Marche Region (2018). Misure per la lotta obbligatoria al plum pox virus agente della vaiolatura delle drupacee nella Regione Marche – individuazione nuove aree delimitate, prescrizioni fitosanitarie e annullamento DD n. 145 del 28/02/17. Anno 2018. Determina del Dirigente ASSAM n.179/det del 12/04/2018, 1, 1–10.
Moreno, A., Fereresm, A., & Cambra, M. (2009). Quantitative estimation of plum pox virus targets acquired and transmitted by a single Myzus persicae. Archives of Virology, 154, 1391–1399. https://doi.org/10.1007/s00705-009-0450-5
Mori, T., Warner, C., Ohno, S., Mori, K., Tobimatsu, T., & Sera, T. (2021). Genome sequence analysis of new plum pox virus isolates from Japan. BMC Research Notes, 14(1). https://doi.org/10.1186/s13104-021-05683-9
Okić, A., Wetzel, T., Nourinejhad Zarghani, S., Massart, S., Grahić, J., Gaši, F., Konjić, A., & Vončina, D. (2022). Occurrence and distribution patterns of plum tree viruses and genetic diversity of sharka isolates in Bosnia and Herzegovina. Horticulturae, 8(9), 783. https://doi.org/10.3390/horticulturae8090783
Palmisano, F., Tocci, C., Zicca, F., Leuzzi, A., Barbalance, C., Zinni, N., Amato, A., Di Lisa, N., & Boscia, D. (2011). La diffusione della sharka non si ferma: Primi ritrovamenti in Calabria e Molise. Rivista Di Frutticoltura e Di Orticoltura, 73, 28–31.
Pasquini, G., & Barba, M. (1994). Serological characterization of Italian isolates of plum pox potyvirus. EPPO Bulletin, 14, 615–624.
Pedrelli, A., Panattoni, A., & Cotrozzi, L. (2023) First molecular characterization of plum pox virus strains in stone fruits of Tuscany (Central Italy). Journal of Plant Pathology, 105, 1045–1053 Advance online publication. https://doi.org/10.1007/s42161-023-01430-0
Pisuttu, C., Lo Piccolo, E., Paoli, L., Cotrozzi, L., Nali, C., Pellegrini, E., & Lorenzini, G. (2023). Physiochemical responses of Ailanthus altissima under the challenge of Verticillium dahliae: Elucidating the decline of one of the world’s worst invasive alien plant species. Biological Invasions, 25, 61–78. https://doi.org/10.1007/s10530-022-02891-7
Poggi Pollini, C., Bissani, R., Giunchedi, L., Gambin, E., & Goio, P. (1996). Sharka: Reperimento di un pericoloso ceppo del virus in coltivazioni di pesco. L’informatore Agrario, 52, 77–79.
Polizzi, G. C., & Agosteo, G. E. (1992). Presence in Calabria (Southern Italy) of drupaceous pox virus on apricot trees. L’informatore Agrario, 48(1), 68–70.
Refatti, E., Osler, R., Loi, N., & Vindimian, E. (1985). Presenza e diffusione della <sharka> in una zona susinicola del Trentino. Rivista Di Patologia Vegetale, 21, 41–49.
Refatti, E., Osler, R., Loi, N., Tomasi, A., Vindimian, E., & Michelotti, F. (1988). La sharka o vaiolatura delle drupacee. 33(1): 32–37.
Region, Sardinia. (2016). Legge regionale 9 marzo 2015 n. 5, l’articolo 11, comma 1. Legge Regionale 14 maggio 2009 n. 1 (legge finanziaria 2009), articolo 4 comma 22 - indennizzi agli imprenditori agricoli destinatari di provvedimenti ufficiali finalizzati all’eradicazione e a impedire la diffusione di organismi nocivi ai vegetali e ai prodotti vegetali; Bilancio regionale 2016. Determinazione n. 10096/325 del 06 luglio 2016. Bollettino Ufficiale delle Regione Autonoma della Sardegna, 33, 1–21.
Revers, F., & García, J. A. (2015). Molecular biology of Potyviruses. Advances in Virus Research, 101–199. https://doi.org/10.1016/bs.aivir.2014.11.006
Rizza, S., Conti, F., Pasquini, G., & Tessitori, M. (2014). First report of plum pox virus strain M isolates in apricot in Sicily, Italy. Plant Disease, 98, 1591–1592. https://doi.org/10.1094/PDIS-05-14-0458-PDN
Rodamilans, B., Valli, A., & García, J. A. (2020). Molecular Plant-plum pox virus interactions. Molecular Plant-Microbe Interactions, 33(1), 6–17. https://doi.org/10.1094/mpmi-07-19-0189-fi
Rubio, M., Martínez-Gómez, P., Marais, A., Sánchez-Navarro, J. A., Pallás, V., & Candresse, T. (2017). Recent advances and prospects in Prunus virology. Annals of Applied Biology, 171(2), 125–138. https://doi.org/10.1111/aab.12371
Sebestyen, D., Nemeth, M., Hangyal, R., Krizbai, L., Ember, I., Nyerges, K., Kolber, M., Kiss, E., & Bese, G. (2008). Ornamental Prunus species as new natural hosts of plum pox virus and their importance in the spread of the virus in Hungary. Journal of Plant Pathology, 90, S57–S61. https://doi.org/10.4454/jpp.v90i1sup.617
Sheveleva, A., Ivanov, P., Gasanova, T., Osipov, G., & Chirkov, S. (2018). Sequence analysis of plum pox virus strain C isolates from Russia revealed prevalence of the D96E Mutation in the universal epitope and interstrain recombination events. Viruses, 10, 450. https://doi.org/10.3390/v10090450
Sihelská, N., Glasa, M., & Šubr, Z. W. (2017). Host preference of the major strains of plum pox virus - Opinions based on regional and world-wide sequence data. Journal of Integrative Agriculture, 16, 510–515. https://doi.org/10.1016/s2095-3119(16)61356-4
Sochor, J., Babula, P., Adam, V., Krska, B., & Kizek, R. (2012). Sharka: The past, the present and the future. Viruses, 4(11), 2853–2901. https://doi.org/10.3390/v4112853
Tamukong, Y. B., Collum, T. D., Stone, A. L., Kappagantu, M., Sherman, D. J., Rogers, E. E., Dardick, C., & Culver, J. N. (2020). Dynamic changes impact the plum pox virus population structure during leaf and bud development. Virology, 548, 192–199. https://doi.org/10.1016/j.virol.2020.06.014
Tuscany Region. (2013). Lotta obbligatoria contro il PPV virus (sharka delle drupacee). Servizio Fitosanitario Regionale e di vigilanza e controllo agroforestale. Report sull’Attività Realizzata nel, 2013(1), 25–28.
Tuscany Region. (2014). Piano d’azione regionale per contrastare in Toscana la diffusione del virus plum pox virus. - Decreto Dirigenziale n. 1027 del 19/3/2014. Bollettino Ufficiale Della Regione Toscana, 1, 127–137.
Tuscany Region. (2019). Servizio Fitosanitario Regionale-Approvazione delle linee guida per contrastare la diffusione in Toscana di plum pox virus (PPV) agente della vaiolatura delle drupacee - Decreto Dirigenziale n. 3518 del 13/3/2019. Bollettino Ufficiale Della Regione Toscana, 1, 1–5.
Umer, M., Liu, J., You, H., Xu, C., Dong, K., Luo, N., Kong, L., Li, X., Hong, N., Wang, G., Fan, X., Kotta-Loizou, I., & Xu, W. (2019). Genomic, morphological and biological traits of the viruses infecting major fruit trees. Viruses, 11(6), 515. https://doi.org/10.3390/v11060515
Vicchi, V., & Babini, A. R. (2008). Attività delle Regioni per il controllo della malattia. Proceedings in: Workshop SHARKA un problema aperto, Faenza 2 aprile 2008.
Vidal, E., Zagrai, L., Malinowski, T., Soika, G., Warabieda, W., Tasheva-Terzieva, E., Milusheva, S., Zagrai, I., Kamenova, I., Bozhkova, V., Martı́nez, C. E., Cambra-López, M., & Cambra, M. (2020). Statistical model for plum pox virus prediction in Prunus nursery blocks using vector and virus incidence data in four different European ecological areas. Annals of Applied Biology, 177(3), 308–324. https://doi.org/10.1111/aab.12617
Wilhelm, J., & Kunze, L. (1995). Plum pseudopox in german prune after infection with an isolate of apple chlorotic leafspot virus causing plum line pattern. Acta Horticulturae, 386, 122–125. https://doi.org/10.17660/actahortic.1995.386.14
Zagrai, I., Zagrai, L., Ferencz, B., Petricele, I. V., Pamfil, D., Popescu, O., Briciu, A., & Festila, A. (2009). Serological and molecular typing of plum pox virus isolates in the Transylvania, Romania. Notulae botanicae Horti Agrobotanici Cluj-Napoca, 37, 265–272. https://doi.org/10.15835/nbha3713134
Acknowledgements
We thank Luisa Oggiano (Agris, Sardinia); Arturo Caponero (ALSIA, Basilicata); Michele Lugo (Trento district, Trentino); Cosimo Tocci (ARSAC, Calabria); Sandro Nardi (ASSAM, Marche); Cosimo Carmelo Caridi (Dipartimento Tutela dell'Ambiente Settore 9, Calabria); Giulio Franco (ERSA, Friuli-Venezia Giulia); Valerio Mazzoni, Valeria Gualandri, Angela Feltrin (Mach Foundation, Trentino); Giuseppe Malvasi (Regional Phytosanitary Service of Basilicata); Massimiliano Trenta (Phytosanitary Service of Trento district, Trentino); Paola Gotta (Regional Phytosanitary Service of Piedmont); Paolo Fini (Regional Phytosanitary Service of Emilia-Romagna); Mario Saverio Orlando (Regional Phytosanitary Service of Calabria); Francesco Baruzzo, Dott.ssa Veronica Raiola (Regional Phytosanitary Service of Liguria); Domenico Carta Cerrella, Maria Etele Salamone, Filadelfo Conti (Regional Phytosanitary Service of Sicily); Rita Bonfanti (Regional Phytosanitary Service of Valle d’Aosta); Beniamino Cavagna (Regional Phytosanitary Service of Lombardy); Domenico Rizzo (Regional Phytosanitary Service of Tuscany); Domenico D’Ascenzo, Gianluca Massi (Regional Phytosanitary Service of Abruzzo); Anna Percoco (Regional Phytosanitary Service of Apulia). Moreover, we thank Rosanna Bertozzi (SBA, University of Pisa, Tuscany) for her kind help in searching for library material.
Funding
Open access funding provided by Università di Pisa within the CRUI-CARE Agreement. No funding was received to assist with the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Athos Pedrelli: Conceptualization, Data recover, Writing-original draft; Alessandra Panattoni: Conceptualization, Data curation; Lorenzo Cotrozzi: Data curation, Supervision, Writing, Review & editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethics approval
All authors contributed and declare that the manuscript was not submitted or previously published.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pedrelli, A., Panattoni, A. & Cotrozzi, L. The sharka disease on stone fruits in Italy: a review, with a focus on Tuscany. Eur J Plant Pathol 169, 287–300 (2024). https://doi.org/10.1007/s10658-024-02827-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-024-02827-y